Abstract
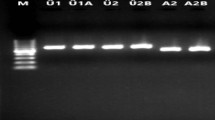
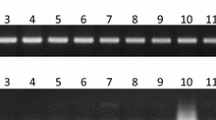
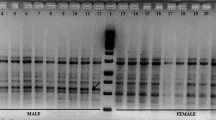
Sex identification in Pistacia species during the long juvenile stage is an economically desirable objective. Due to the lack of morphological methods to identify sex at this stage, the application of molecular markers is expected to facilitate breeding programs. The aim of our study was to identify a marker closely linked to sex loci in Pistacia atlantica Desf subsp. mutica, P. khinjuk, and P. vera subsp. Sarakhs. Samples were collected from both male and female plants of each species, and their band patterns were analyzed according to the presence or absence of specific bands. Thirty random amplified polymorphic DNA (RAPD) primers and a pair of sequence characterized amplified region (SCAR) primers were tested as potential markers of sex in wild Pistacia species. Among the RAPD primers, only BC1200 was found to amplify a specific sex band present in female plants. Based on our analysis of all individual samples, a fragment of approximately 300 bp was amplified in female trees but absent in male ones. Although sex determination mechanisms in Pistacia are still unknown, they may be controlled by a single locus that acts as a trigger. The SCAR technique has proved to be a reliable technique in gender determination of pistachio genotypes at the seedling phenophase. This method could reduce both the time and costs associated with breeding programs.


Similar content being viewed by others
References
Ainsworth C (2000) Boys and girls come out to play: the molecular biology of dioecious plants. Ann Bot 86:211–221. doi:10.1006/anbo.2000.1201
Alstrom-Rapaport C, Lascoux M, Wang YC, Roberts G, Tuskan GA (1998) Identification of a RAPD marker linked to sex determination in the basket willow, Salix viminalis L. J Hered 89:44–49. doi:10.1093/jhered/89.1.44
Bedoya GC, Nunez V (2007) A SCAR marker for the sex types determination in Colombian genotypes of Carica papaya. Euphytica 153:215–220. doi:10.1007/s10681-006-9256-7
Durand R, Durand B (1990) Sexual determination and sexual differentiation. Crit Rev Plant Sci 9:295–316
Gao WJ, Li RL, Li F, Deng CHL, Li SP (2007) Identification of two markers linked to the sex locus in dioecious Asparagus officinalis plants. Russ J Plant Physiol 54:816–821. doi:10.1134/S1021443707060143
Hormaza JI, Dollo L, Polito VS (1994) Identification of a RAPD marker linked to sex determination in Pistacia vera using bulked segregant analysis. Theor Appl Genet 89:9–13. doi:10.1007/BF00226975
Irish E, Nelson T (1989) Sex determination in monoecious and dioecious plants. Plant Cell 1:737–744
Jiang C, Sink KC (1997) RAPD and SCAR markers linked to the sex expression locus M in Asparagus. Euphytica 94:329–333
Jiang L, You RL, Li MX, Shi C (2003) Identification of a sex-associated RAPD marker in Ginkgo biloba. Acta Bot Sin 45:742–747
Korpelainen H, Bisang I, Hedenas L, Kolehmainen J (2008) The first sex-specific molecular marker for mosses discovered in Pseudocalliergon trifarium. J Hered 99:581–587. doi:10.1093/jhered/esn036
Matsunaga S (2006) Sex chromosome-linked genes in plant. Gene Genet Syst 81:219–226
Noroozi S, Baghizadeh A, Javaran MJ (2009) The genetic diversity of Iranian pistachio (Pistacia vera L.) cultivars revealed by ISSR markers. Biol Diversity Conserv 2/2:50–56
Paran I, Michelmore RW (1993) Development of reliable PCR-based markers linked to downy mildew resistance genes in lettuce. Theor Appl Genet 85:985–993. doi:10.1007/BF00215038
Parasnis AS, Ramakrishna W, Chowdari KV, Gupta VS, Pranjekar K (1999) Microsatellite (GATA)n reveals sex-specific differences in papaya. Theor Appl Genet 99:1047–1052. doi:10.1007/s001220051413
Peng J, Richards D, Hartley N, Murphy G, Devos K, Flintham J, Beales J, Fish L, Worland A (1999) ‘Green revolution’ genes encode mutant gibberellin response modulators. Nature 400:256–261
Perl-Treves R (1999) Male to female conversion along the cucumber shoot: approaches to studying sex genes and floral development in Cucumis sativus. In: Ainsworth C (ed) Sex determination in plants. Bios Scientific, Oxford, pp 189–216
Prakash S, Vanstaden S (2006) Sex identification in Encephalartos natalensis (Dyer and Verdoorn) using RAPD markers. Euphytica 152:197–200. doi:10.1007/s10681-006-9198-0
Raus FC, Fairbanks JD, Evans PR, Stutz CH, Anderson RW, Raus MP (1998) Male-specific DNA in the dioecious species Atriplex garrettii (Chenopodiaceae). Am J Bot 85:162–167
Shirkot P, Sharma DR, Mohapatra T (2002) Molecular identification of sex in Actinidia deliciosa var. deliciosa by RAPD markers. Sci Hortic 94:33–39
Sim MC, Lim PE, Gan SY, Phang SM (2007) Identification of random amplified polymorphic DNA (RAPD) marker for differentiating male from female and sporophytic thalli of Gracilaria changii (Rhodophyta). J Appl Phycol 19:763–769. doi:10.1007/s1081-007-9224-1
Stehlik I, Blattner FR (2004) Sex-specific SCAR markers in the dioecious plant Rumex nivalis (Polygonaceae) and implication for the evolution of sex chromosome. Theor Appl Genet 108:238–242. doi:10.1007/s00122-003-1425-7
Terauchi R, Kahl G (1999) Mapping of the Dioscorea tokoro genome: AFLP markers linked to sex. Genome 42:752–762
Urasaki N, Tokumoto M, Tarora K, Ban Y, Kayano T, Tanaka H, Oku H, Chinen I (2002) A male and hermaphrodite specific RAPD marker for papaya (Carica papaya L). Theor Appl Genet 104:281–285
Yakubov B, Barazani O, Golan-Goldhirsh A (2004) Combination of SCAR primers and Touchdown-PCR for sex identification in Pistacia vera L. Sci Hortic 103:473–478. doi:10.1016/j.scienta.2004.06.008
Zhang YH, Distilo VS, Rehman F, Avery A, Mulcahy D (1998) Y chromosome specific markers and the evolution of dioecy in the genus Silene. Genome 41:141–147
Acknowledgments
The authors thank H. Mirshahi for her assistance with various aspect of this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Esfandiyari, B., Davarynejad, G.H., Shahriari, F. et al. Data to the sex determination in Pistacia species using molecular markers. Euphytica 185, 227–231 (2012). https://doi.org/10.1007/s10681-011-0527-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-011-0527-6