Abstract
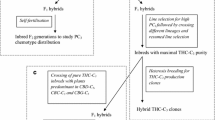
This paper aims to clarify the genetic mechanism that is responsible for the accumulation of cannabigerol (CBG) in certain phenotypes of Cannabis sativa L. CBG is the direct precursor of the cannabinoids CBD, THC and CBC. Plants strongly predominant in CBG have been found in different fibre hemp accessions. Inbred offspring derived from one such individual were crossed with true breeding THC predominant- and CBD predominant plants, respectively. The segregations in the cross progenies indicate that CBG accumulation is due to the homozygous presence of a minimally functional allele, tentatively called B0, at the single locus B that normally controls the conversion of CBG into THC (allele BT) and/or CBD (allele BD). The fact that CBG accumulating plants have so far been found in European fibre hemp populations that are generally composed of BD/BD plants, and the observation that the here investigated B0 allele possesses a residual ability to convert small amounts of CBG into CBD, make it plausible that this B0 is a mutation of normally functional BD. Therefore, B0 is considered as a member of the BD allelic series encoding a CBD synthase isoform with greatly weakened substrate affinity and/or catalytic capacity.
Similar content being viewed by others
References
Adams, R., M. Hunt & J.H. Clark, 1940. Structure of cannabidiol, a product isolated from the marihuana extract of Minnesota wild hemp. J Am Chem Soc 62: 196–200.
Fellermeier, M. & M.H. Zenk, 1998. Prenylation of olivetolate by a hemp transferase yields cannabigerolic acid, the precursor of tetrahydrocannabinol. FEBS Lett 427: 283–285.
Fournier, G., C. Richez-Dumanois, J. Duvezin, J.-P. Mathieu & M. Paris, 1987. Identification of a new chemotype in Cannabis sativa: Cannabigerol-dominant plants, biogenetic and agronomic prospects. Planta Med 53: 277–280.
Gaoni, Y. & R. Mechoulam, 1964a. Isolation, structure and partial synthesis of an active constituent of hashish. J Am Chem Soc 86: 1646–1647.
Gaoni, Y. & R. Mechoulam, 1964b. The structure and synthesis of cannabigerol a new hashish constituent. Proc Chem Soc, March: 82.
Gaoni, Y. & R. Mechoulam, 1966. Cannabichromene, a new active principle in hashish. Chem Commun 1: 20–21.
Hillig, K., 2002. Letter to the editor. J Ind Hemp 7: 5–6.
Hillig, K. & E.P.M. de Meijer, 2004. Letter to the editor ‘On Cannabis’. Econ Bot 58: 328.
Hillig, K.W. & P.G. Mahlberg, 2004. A chemotaxonomic analysis of cannabinoid variation in Cannabis (Cannabaceae). Am J Bot 91: 966–975.
Mandolino, G., 2004. Again on the nature of inheritance of chemotype. Letter to the editor. J Ind Hemp 9: 5–7.
Mechoulam, R. & Y. Shvo, 1963. Hashish-I, the structure of cannabidiol. Tetrahedron 19: 2073–2078.
de Meijer, E.P.M., M. Bagatta, A. Carboni, P. Crucitti, V.M.C. Moliterni, P. Ranalli & G. Mandolino, 2003. The inheritance of chemical phenotype in Cannabis sativa L. Genetics 163: 335–346.
de Meijer, E.P.M., 2004. The breeding of Cannabis cultivars for pharmaceutical end-uses. In: G.W. Guy, B.A. Whittle & P.J. Robson (Eds.), The medicinal uses of Cannabis and cannabinoids, pp. 55–69. Pharmaceutical Press, London.
Morimoto, S., K. Komatsu, F. Taura & Y. Shoyama, 1997. Enzymological evidence for cannabichromenic acid biosynthesis. J Nat Prod 60: 854–857.
Morimoto, S., K. Komatsu, F. Taura & Y. Shoyama, 1998. Purification and characterization of cannabichromenic acid synthase from Cannabis sativa. Phytochemistry 49: 1525– 1529.
Shoyama, Y., H. Hirano & I. Nishioka, 1984. Biosynthesis of propyl cannabinoid acid and its biosynthetic relationship with pentyl and methyl cannabinoid acids. Phytochemistry 23: 1909– 1912.
Small, E. & D. Marcus, 2003. Tetrahydrocannabinol levels in hemp (Cannabis sativa) germplasm resources. Econ Bot 57: 545–558.
Taura, F., S. Morimoto, Y. Shoyama & R. Mechoulam, 1995. First direct evidence for the mechanism of delta-1-tetrahydrocannabinolic acid biosynthesis. J Am Chem Soc 38: 9766–9767.
Taura, F., S. Morimoto & Y. Shoyama, 1996. Purification and characterization of cannabidiolic-acid synthase from Cannabis sativa L. J Biol Chem 271: 17411–17416.
Virovets, V.G., 1996. Selection for non-psychoactive hemp varieties (Cannabis sativa L.) in the CIS (former USSR). J Int Hemp Assoc 3: 13–15.
Vree, T.B., D.D. Breimer, C.A.M. van Ginneken & J.M. van Rossum, 1971. Identification of the methyl and propyl homologues of CBD, THC and CBN in hashish by a new method of combined gas chromatography-mass spectrometry. Acta Pharm Suedica 8: 683–684.
de Zeeuw, R.A., J. Wijsbek, D.D. Breimer, T.B. Vree, C.A. Van Ginneken & J.M. van Rossum, 1972. Cannabinoids with a propyl side chain in Cannabis. Occurrence and chromatographic behaviour. Science 175: 778–779.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Meijer, E.P.M., Hammond, K.M. The inheritance of chemical phenotype in Cannabis sativa L. (II): Cannabigerol predominant plants. Euphytica 145, 189–198 (2005). https://doi.org/10.1007/s10681-005-1164-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10681-005-1164-8