Abstract
Hypoxia in coastal seas is a severe threat to marine ecosystems, with the Baltic Sea exhibiting the largest hypoxic areas worldwide. While perennial oxygen deficiency in the deep basins is a component of environmental assessments, seasonal oxygen deficiency in shallow areas is not routinely assessed. Current measurements alone cannot provide the spatio-temporal resolution needed for highly dynamic seasonal oxygen deficiency, making estimations on its duration and extent uncertain. Utilizing long-term 3D model simulations with a horizontal resolution of 3 nautical miles, we analyzed the development of seasonal oxygen deficiency in the western Baltic Sea. Different metrics (near-bottom area, water volume, duration, and frequency) and critical oxygen concentrations were analyzed for exemplary sub-basins as defined by the Helsinki Commission. Our results indicate that the extent of seasonal oxygen deficiency has continually increased in the second half of the twentieth century until the end of the 1980s and slightly decreased in the last two decades. In the 1950s, the spatial extent of oxygen deficiency was still at a low plateau before increasing, indicating that this period could be suitable as a reference period representing a good status, including naturally occurring oxygen deficiency. Overall, seasonal oxygen deficiency is a suitable indicator for describing the ecological status of the western Baltic Sea. For an application as eutrophication indicator in shallow areas, a sufficient spatio-temporal resolution of seasonal oxygen deficiency is needed which can be gained by a combination of model simulations and measurements. Further analysis is needed to integrate in situ measurements and model results to obtain the most reliable approach.
Similar content being viewed by others
1 Introduction
The Baltic Sea exhibits the worldwide largest hypoxic areas (oxygen concentration < 2 mg/l; [1] and its frequency and extent continually increased over the last century due to excess nutrient inputs [2]. Therefore, monitoring of hypoxia is of high importance and the development of an oxygen indicator current subject of the work of commissions on the protection of the Baltic Sea.
Due to limited exchange with salty North Sea water, a vertical salinity stratification is characteristic for the intracontinental Baltic Sea [3]. Due to restricted ventilation with oxygen-rich surface waters, the Baltic Sea exhibits naturally occurring hypoxic areas [4]. But with its catchment area about four times larger than its surface area, the Baltic Sea is specifically influenced by eutrophication-induced hypoxia through excess nutrient inputs (nitrogen and phosphorus) from land and the atmosphere [5, 6]. Although hypoxia-stimulating nutrient inputs have decreased since the 1980s [6], significant improvements in oxygen concentrations are not yet evident [7,8,9]. The reasons for this are the long water residence time of about 30 years [1] and feedback loops (phosphorus release under anoxic conditions,vicious circle [10]). The effects of climate change can further enhance hypoxic conditions by increasing temperature and stratification as well as salinity changes, resulting in lower solubility of oxygen and reduced vertical mixing [11,12,13].
For the Baltic Sea, three types of hypoxia can be distinguished based on a temporal aspect: perennial hypoxia in the deep open and central part, seasonal hypoxic events during summer and autumn, and episodic hypoxic events at many shallow coastal sites [1]. Perennial hypoxia occurs in areas with a permanent halocline, where vertical mixing with oxygen-rich surface water is limited. It is suggested that perennial hypoxia in the Baltic Sea has reached its maximum areal extent [7, 8]. In contrast, seasonal and episodic hypoxia is increasingly observed in many places [14, 15]. Especially in the western Baltic Sea, phenomena of seasonal hypoxia were observed only occasionally until the 1970s, before annual observations of seasonal hypoxia were made, for example, in Kiel Bay and Bay of Mecklenburg [16]. Seasonal stratification of the water column due to decreased bottom water transport and seasonal temperature increase favors the occurrence of seasonal hypoxia [14]. In addition, specific local conditions, such as low oxygen input from the air, complex bottom topography, and rapid oxygen consumption by biological processes, are favorable for episodic hypoxic events to occur [1, 17].
Eutrophication and oxygen deficiency as an indirect effect are the most challenging environmental problems of the Baltic Sea. Negative impacts of hypoxia include altered distributions and abundances of animal populations and key changes in benthic community structures [18, 19], whereas most prominent are mass mortality events of fishes caused by hypoxia [16, 20]. In the Baltic Sea, not only spawning success of cod can be impaired by hypoxia [21]. Moreover, about 30% of total secondary production is missing due to persistent hypoxic zones [22] and therewith a potential benthic food energy for fisheries is lost.
To counteract associated negative effects for the marine ecosystem, regional and (inter)national conventions (Helsinki Convention (HELCOM)) and legislations (EU Water Framework Directive (WFD, 2000/60/EC); Marine Strategy Framework Directive (MSFD, 2008/56/EC)) implemented actions against eutrophication and require their member states to reach a Good Environmental (or Ecological) Status (GES). As an indirect effect of eutrophication, dissolved oxygen in the bottom of the water column is included as a primary and thereby mandatory criterion to assess under Descriptor 5 “eutrophication” of the MSFD. For the Baltic Sea Action Plan, the achievement of “natural oxygen levels” is one of the ecological objectives in the eutrophication segment [ 23, 24].
More specifically, “oxygen debt” is implemented by HELCOM as core indicator to evaluate the average oxygen debt below the halocline [25], thus assessing perennial hypoxia in the deep basins. Due to increasing occurrences of seasonal hypoxic events [14], HELCOM already stated the need for an additional shallow water oxygen indicator, assessing oxygen deficiency in areas without a permanent halocline [26, 27]. Currently, the shallow water oxygen indicator has “pre-core” status but was not applied in the last holistic HELCOM assessment HOLAS II and the report on the state of the Baltic Sea due to the lack of common target values [5, 28].
Several Baltic States already implemented monitoring approaches to assess oxygen deficiency in their water bodies as demanded by EU Directives (MSFD, WFD). In contrast to the oxygen debt indicator, various oxygen thresholds from < 2 to < 6 mg/l are used to describe the boundary between a good and moderate condition for the shallow water oxygen indicator [16, 29]. Volume and/or area are mostly used as metrics, whereas single-point oxygen measurements are extrapolated into space to evaluate oxygen deficiency. Currently lacking in the assessment of Baltic States, but of extreme importance to benthic communities, is the duration and frequency of hypoxia [19]. For example, in Chesapeake Bay duration of hypoxia has already been used as metric to quantify the bay-wide hypoxic areas [30].
Although the frequency and spatial coverage of oxygen measurements has improved since the 1960s [31], the assessment of highly dynamic seasonal oxygen deficiency stays uncertain due to the spatio-temporal limits of single-point measurements. Thus, the development of an adequate and comparable shallow water oxygen indicator, which targets seasonal oxygen deficiency, is still ongoing. As time and resources to conduct extensive cruises are limited, additional methods to assess seasonal oxygen deficiency are needed. Here, numerical models can help to provide high-resolution information on the frequency, the duration, and both the horizontal and vertical extent of oxygen deficiency areas. The German national working group explicitly mentioned hydrodynamic modeling as an important approach to gain information on the temporal and spatial extent of oxygen deficiency to support the MSFD criterion “dissolved oxygen in the bottom of the water column” (Commission Decision 2017/848/EU) in the future [16].
In our study, we used the coupled hydrodynamic-biogeochemical model MOM-ERGOM to gain knowledge on the spatio-temporal variability of seasonal oxygen deficiency in the western Baltic Sea and to assess various oxygen metrics.
Specifically, we aim (I) to evaluate the model quality by comparing model data against in situ measurements for representative stations, (II) to analyze the spatio-temporal variability of oxygen concentrations in the western Baltic to draw conclusions for a more efficient oxygen monitoring, (III) to assess various oxygen metrics derivable from model simulation products to be used as oxygen indicators, and (IV) to analyze time-series of oxygen concentrations as a basis for the determination of reference values and related thresholds.
2 Material and Methods
2.1 The Ecosystem Model Approach
The model simulations in the Baltic Sea (Fig. 1) were performed with the integrated biogeochemical model ERGOM (www.ergom.net) which is coupled to a 3D circulation model (MOM) [32, 33]. The model has previously been applied in the Baltic Sea to analyze the extent of hypoxic areas in conjunction with saltwater inflows from the North Sea, nutrient input scenarios as well as climate change scenarios [34,35,36,37]. The biogeochemical model simulates the marine nitrogen and phosphorus cycle: the three nutrients dissolved in water, ammonium, nitrate, and phosphate, which are the basis for primary production realized by three functional phytoplankton groups (large cells, small cells, and cyanobacteria). Grazing pressure on the phytoplankton is applied in the model via a dynamically developing bulk zooplankton variable. Dead organic material is considered by a detritus state variable.
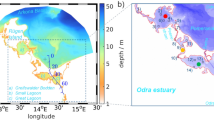
The Baltic Sea in northwestern Europe with sub-basin divisions according to HELCOM (black lines) and our study area including the sub-basins “Kiel Bay,” “Bay of Mecklenburg,” “Arkona Basin,” and “Pomeranian Bay” located in the shallow western Baltic Sea including measuring stations used for model validation (red dots)
During sinking, part of the detritus is mineralized again into dissolved ammonium and phosphate. The portion that reaches the sea bottom accumulates and is partly buried permanently, or alternatively mineralized or resuspended when the velocity of near-bottom currents is sufficiently high. Coupled to the nitrogen and phosphorus cycle is a carbon cycle as described in Kuznetsov and Neumann [38]. Under oxic conditions, part of the mineralized phosphate is bound by iron oxides and is retained in the sediment. When conditions become anoxic, it is released to the water column. Oxygen development is coupled to biogeochemical processes via stoichiometric ratios, with oxygen levels in turn controlling processes such as denitrification and nitrification.
The physical part of the model is based on the circulation model MOM (version 5.1; [39, 40]). It has been adapted to the Baltic Sea with an open boundary condition to the North Sea and riverine freshwater input. To estimate ice cover thickness and extent, the MOM model is complemented with a sea ice model [41]. The horizontal resolution of the model grid is three nautical miles, while vertically the model is resolved into 152 layers, with layer thicknesses from 0.5 to 2 m. The model domain and bathymetry are shown in Fig. 1.
Atmospheric forcing is based on a dynamical downscaling provided by the coastDat data set [42, 43] with a grid resolution of about 25 × 25 km. Nutrient loads to the Baltic Sea due to riverine discharge have been compiled based on data from HELCOM assessments (e.g., [44, 45]) and prior to 1995 based on Gustafsson et al. [46]. Atmospheric deposition of nitrogen inputs was provided by EMEP [47, 48] and prior to 1995 by Ruoho-Airola et al. [49].
2.2 Model Performance
As the model system ERGOM-MOM is largely validated against recent observations, focusing on key parameters and stations in the Baltic Sea [37, 50,51,52,53], the presented study is focused on the oxygen dynamics in the western Baltic Sea. Therefore, we compared the model data with in situ measurements for representative stations in the western Baltic Sea. Observational data was provided by local authorities (State Agency for Agriculture, Environment and Rural Areas (LLUR) and State Agency for Environment, Nature Conservation and Geology Mecklenburg-Vorpommern (LUNG)) and enhanced by data from the Leibniz Institute for Baltic Sea Research Warnemünde (https://odin2.io-warnemuende.de/) and data from the ICES oceanographic database (https://www.ices.dk/data/data-portals/Pages/ocean.aspx). Observations were checked for plausibility and only the deepest layer was selected (except for the vertical profile comparison).
To analyze the seasonal cycle, four representative stations in the western Baltic Sea were selected (Fig. 1), representing mostly the deeper basins (Kiel Bay, Bay of Mecklenburg, and Arkona Basin). While these stations are dominated by muddy sediments [54], for comparison a sandy station (O9, west of Hiddensee) was additionally selected. To compare the model behavior, observations from the last years (2010–2019) were condensed to multi-annual monthly means to verify that the model is capable to reproduce the seasonal cycle. To analyze the spatial agreement, the averaged annual minima from the period 2010 to 2019 of the model results and observations were compared.
2.3 Model Data Analysis
The analysis of the model results was focused on the western Baltic Sea sub-basins depicted in Fig. 1, following the HELCOM assessment units expanded by the newly defined Pomeranian Bay unit. A long-term model run from 1950 to 2019, with daily oxygen concentrations as output, served as basis for all analysis. The initial conditions of 1950 were taken from an earlier simulation starting in the 1850s, implying a model spin-up time of 100 years. The period from 2011 to 2016 which coincides with the latest HELCOM assessment period (HOLAS II) was selected to assess the spatio-temporal variability of oxygen concentrations (II) as well as to assess the various oxygen metrics (III). For the assessment of long-term changes in oxygen concentrations (IV), the full dataset from 1950 to 2019 was investigated.
For the analysis of oxygen deficiency of the near-bottom area (II), we utilized the model results approximately 2.5 m above the bottom. Extracted daily oxygen values were averaged either monthly, yearly, or over assessment periods of 6 years. For the calculation of the standard deviation, hydrogen sulfide (as negative oxygen equivalents) was considered by subtracting twice the concentration of H2S from the oxygen values.
For the analysis of the oxygen metrics (III), mean values were not based on daily oxygen concentrations but on whether oxygen concentrations did fall below critical levels (2, 4, or 6 mg/l oxygen). In the following, all situations below 2 mg/l will be referred to as hypoxia and all situations below 6 mg/l to 2 mg/l will be referred to as oxygen deficiency. For the metric “oxygen depleted area,” the sum of all horizontal model grid cells 2.5 m above the bottom with oxygen values below critical levels was calculated. For the “oxygen depleted volume,” the size of all horizontal and vertical model grid cells (starting from about 2.5 m above the bottom) with oxygen values below critical levels were summed up.
To analyze the “frequency of oxygen deficiency,” the annual average number of days with oxygen concentrations below critical levels in the near-bottom area was calculated and subsequently averaged over the assessment period. For the “duration of oxygen deficiency,” we averaged the number of occurrences of consecutive days (> 2, > 7, > 14, or > 21 days) within a year for oxygen concentrations below the specific critical oxygen levels. Analyzed oxygen concentration-period combinations were chosen based on the comprehensive study by Vaquer-Sunyer and Duarte [55] providing information on lethal and sublethal oxygen concentrations and periods for benthic organisms.
The area of the near-bottom water layer and water volume used for calculations were computed from the model domain with the values for each sub-basin shown in Table 1.
Data aggregation and analysis was performed using CDO (version 1.9.8) and R (version 3.6.3 (2020–02-29)) with the tmap package (version 3.3) to produce all maps.
3 Results
3.1 Model Performance
The model performance was evaluated focusing on the seasonal cycle (Fig. 2) and the spatial fit with low-oxygen areas (Fig. 3).
Comparison of multi-annual monthly means of modeled oxygen concentrations from the deepest vertical layer (orange) and 2.5 m above bottom (purple) with observed near-bottom data (gray and black) for four stations: a Kiel Bay, b Bay of Mecklenburg, c Station O9 in the south-western Arkona Basin, and d Station TF113 in the central Arkona Basin in the western Baltic Sea (see Fig. 1). Single measurements are indicated by gray points and aggregated to monthly means (black squares). Observations and model results were taken from the period 2010 to 2019
The mean absolute error of the modeled oxygen concentrations over a year is comparably lower for the sandy station O9 (0.14) than for the central station in the Arkona Basin (1.75), Kiel Bay (1.51), and Bay of Mecklenburg (1.92; Fig. 2). For the latter two stations, the model shows considerably lower values especially from October to February (mean absolute error of 3.54 and 3.85, respectively) while the spring and summer months are well represented. In particular, the increase in oxygen concentrations after October is comparatively not fast enough. This underestimation of the bottom oxygen concentrations in Kiel Bay and Bay of Mecklenburg is accompanied by a too strong vertical stratification, which can be seen by a comparison of modeled and observed salinities (SI 1).
Since measurements directly above the seafloor are not possible, and rather taken within a range of about 1 to 4 m above the seafloor, we also compared the observations with the values in the model layer 2.5 m above the seafloor (purple dots in Fig. 2). Here, the fit between modeled and observed oxygen concentration improved substantially for the stations in the Kiel Bay and Bay of Mecklenburg (Fig. 2a, b). Although for stratified stations some differences between observed and modeled data still exist in the autumn and winter months, the model results 2.5 m above the bottom capture the seasonal oxygen minima better. For our analysis of the bottom water layer, we thus utilized this model layer which is in the following referred to as “near-bottom layer.”
Next, we compared the minima oxygen values from 2010 to 2019 of the modeled near-bottom oxygen concentrations with the deviation to observed values for stations in the entire western Baltic Sea (Fig. 3). The average annual modeled near-bottom oxygen minima are mostly in good agreement with the average of the annual minima from the observations (Fig. 3). A reasonable good correlation between modeled and observed values (r = 0.71, R2 = 0.5) indicates that the model system is well able to reproduce the spatial gradients in the western Baltic Sea. Higher modeled oxygen concentrations compared to the measurements occur mainly along the coastline. In some near-shore areas (like Bay of Wismar and the southern coast of Funen), the model tends to overestimate the oxygen concentrations substantially, as the steep bathymetry gradients are not resolved. In contrast, too low oxygen concentrations are mainly predicted by the model in the deep basins and the western part of Pomeranian Bay. Apart from that, in other areas the differences between model values and observations seem to vary strongly although some measuring stations are quite near to each other as, for example, in the Arkona Basin (Fig. 3). This is maybe caused by unevenly distributed observations in time among measuring stations, as data from some measuring stations is less often available. For example, if no observations for the time period with the lowest oxygen concentrations are available, the annual minima are hardly comparable between the monitoring stations.
3.2 Analysis of Spatial and Seasonal Oxygen Deficiency
According to model results, there is a large difference of the extent of areas affected by oxygen deficiency among the assessment units (Fig. 4a and Table 2). For the period 2011–2016, averaged annual dissolved oxygen concentrations do not show hypoxia in the western Baltic Sea (Fig. 4a), but seasonally hypoxia occurs from July to September (Fig. 5). Considering oxygen deficiency, the Bay of Mecklenburg exhibited the largest areas with critical oxygen concentrations below 6 mg/l with on average 45% of the near-bottom water layer, followed by Kiel Bay with on average 11% (Fig. 4a). In the Arkona Basin and the Pomeranian Bay, the average annual oxygen concentrations in the near-bottom water layer were above critical levels (Fig. 4a). In most areas of the western Baltic Sea, the annual average variability of oxygen concentration is 0 to 4 mg/l with highest variability observed in the south-western part of Bay of Mecklenburg (Fig. 4b).
Modeled a average annual oxygen concentrations (DO [mg/l]) and b standard deviation (STD [mg/l]) in the near-bottom water layer in the period 2011 to 2016 in the western Baltic Sea. Black lines indicate sub-basin divisions according to HELCOM (KB, Kiel Bay; BM, Bay of Mecklenburg; AB, Arkona Basin; PB, Pomeranian Bay). Country borders in gray
Modeled average monthly oxygen concentrations (DO [mg/l]) in the near-bottom water layer from April to September for the period 2011 to 2016 in the western Baltic Sea (for standard deviation see SI 3). Black lines indicate sub-basin divisions according to HELCOM (KB, Kiel Bay; BM, Bay of Mecklenburg; AB, Arkona Basin; PB, Pomeranian Bay). Country borders in gray
Comparing average annual oxygen concentrations among single years (SI 7), the differences were not as pronounced as between the individual months (Fig. 5); thus, seasonal variability seems to be more important for an assessment than inter-annual variability. Moreover, the lower model fit in the months of October through March is also reflected by a higher variability of oxygen concentrations in these months (SI 3). Therefore, only the results for the months April to September are shown in Fig. 5 and Table 2 and are considered for the seasonal analysis below.
Generally, the spatial extent and time-span of oxygen deficiency differed among sub-basins (Figs. 5 and 6, Table 2). To account for naturally occurring hypoxia and oxygen deficiency in the area, we defined critical periods exemplary as periods in which at least 10% of the near-bottom area of a sub-basin exhibits concentrations below a critical oxygen level. Accordingly, critical periods of hypoxia can be observed in the Bay of Mecklenburg (17 to 21% during August and September) and the Arkona Basin (10% in September). According to model results, hypoxia was also observed in a small area during the summer in the Kiel Bay (3 to 7%) and the Pomeranian Bay (1 to 2%; Fig. 5, Table 2).
Bar charts showing the modeled average monthly oxygen depleted water volume as percentage for the period from 2011 to 2016 where oxygen concentrations were below 2, 4, and 6 mg/l (purple coloring) separately for the four sub-basins of the western Baltic Sea. The black bars indicate the 95% confidence interval
For oxygen deficiency, critical periods are highly variable among the sub-basins ranging from about 1 to 5 months (Fig. 5, Table 2). Likewise, the spatial extent exhibits a high variability among the sub-basins from about 10 to 74% (Fig. 5, Table 2). For example, the critical period in the Bay of Mecklenburg lasts 5 months and affects an area from 16 to 74%, whereas in the Pomeranian Bay an area of 10% is affected for only 1 month (Fig. 5, Table 2). Kiel Bay exhibits a near-bottom area between 17 and 74% affected by oxygen concentrations below 6 mg/l, whereas the period is 1 month less as compared to the Bay of Mecklenburg. In the Arkona Basin an average area from 16 to 36% is affected within a period of 3 months.
The high spatio-temporal variability between assessment units is also seen in the analysis of the water volume affected by hypoxia and oxygen deficiency (Fig. 6). Within the period from April to September, the affected water volume by hypoxia reaches less than 2% in Kiel Bay, the Arkona Basin, and in the Pomeranian Bay. Only in the Bay of Mecklenburg the monthly average volume of water reaches up to 7% in September (Fig. 6). Looking at the monthly average volume of water where oxygen concentration has dropped below 6 mg/l, less than 10% of the water volume in the Arkona Basin and Pomeranian Bay is on average affected over the year. Again, Bay of Mecklenburg is the most affected, with water volumes from 3 to 27% experiencing oxygen deficiency, followed closely by the Kiel Bay where water volumes from 2 to 22% experience oxygen deficiency during the period from April to September (Fig. 6).
3.3 Assessment of Ecological Relevant Oxygen Indicator Metrics
To evaluate various oxygen metrics that can be derived from model simulation products and be used as oxygen indicators, we further looked more closely at the temporal component in addition to intensity and extent. Considering the duration and recurrence of oxygen deficiency situations is a critical step toward developing metrics relevant to the ecological health of benthic communities, as duration is most important along with the intensity of oxygen deficiency.
Hypoxic conditions in the near-bottom water do frequently (at least half a year ± 1 to 2 months) occur within 5% of the Bay of Mecklenburg. About 25% of the area shows hypoxia from a quarter to half a year and 50% of the area experiences hypoxic conditions for less than 2 months (Fig. 7). Within the Arkona Basin 91% and in the Pomeranian Bay even the whole area is less than twice a month affected by hypoxic conditions (Fig. 7).
Maps showing modeled annual average (top left) and standard deviation (top right) of days with near-bottom oxygen concentrations below 2 mg/l for the western Baltic Sea in the period from 2011 to 2016. The corresponding average annual extent of the near-bottom water layer is shown as percentage (bottom)
If a threshold of 6 mg/l is considered, the near-bottom water layer experiencing oxygen deficiency of at least half a year rises to 43% in the Bay of Mecklenburg (SI 4). In addition, 13% of the near-bottom water layer in Kiel Bay is also affected by critical oxygen levels below 6 mg/l at this frequency (SI 4), while in the Arkona Basin 22% of the near-bottom water layer are affected from a quarter to half a year (SI 4). Additional information on the frequency of situations below critical oxygen levels of 4 and 6 mg/l is provided within the supplementary information.
The largest share of areas and highest incidences where hypoxia occurs for more than 2 as well as 7 consecutive days are in the Bay of Mecklenburg and Arkona Basin (for up to 6 times per year and 2 to 4 times a year, respectively), followed by Kiel and Pomeranian Bay (Fig. 8). Hypoxic conditions for more than 14 consecutive days occur on average about 1 to 2 times a year for noticeable areas in the Arkona Basin and Bay of Mecklenburg, the latter showing areas with incidences of up to 4 times (Fig. 8). However, the pattern of recurrences behaves differently within the single assessment units. While the average number of days per year with hypoxia is high in the deeper parts (Fig. 7), the recurrence is low, indicating that only a few, but long-lasting hypoxic events occur. This changes at the borders, e.g., of Bay of Mecklenburg, where the number of incidences of at least 2 days with oxygen conditions below 2 mg/l is very high (Fig. 8), although the average number of days per year below the oxygen threshold is quite low. This indicates that the oxygen conditions are much more dynamic in these intermediate waters.
Maps showing the modeled frequency of occurrence of hypoxic situations (dissolved oxygen < 2 mg/l) in the near-bottom water layer for specific time periods in the western Baltic Sea. White areas indicate areas where no hypoxic situations occurred during the periods studied. Black lines indicate sub-basin divisions according to HELCOM (KB, Kiel Bay; BM, Bay of Mecklenburg; AB, Arkona Basin; PB, Pomeranian Bay). Country borders in gray
Finally, looking at the occurrence of critical oxygen concentrations below 4 and 6 mg/l for certain periods of time, we further see that the oxygen-deficient near-bottom areas increase, but not the incidences (SI 5). When different thresholds are applied, the areas affected by hypoxia and oxygen deficiency remain similar in the Bay of Mecklenburg but increase clearly recognizable in the Kiel Bay and Arkona Basin (Figs. 8 and SI 5).
3.4 Long-Term Analysis of Oxygen Deficiency
Looking at previous hypothetical assessment periods, our model results show that averaged annual dissolved oxygen concentrations do not show hypoxia in the western Baltic Sea (Fig. 9). However, the near-bottom area and water volume affected by hypoxia can be identified when evaluating the near-bottom water layer and water volume with oxygen concentrations below 2 mg/l over this period (Fig. 10). A general increase in hypoxic near-bottom water layer as well as water volume can especially be seen for the Kiel Bay and Bay of Mecklenburg until the 1980s before conditions improved at the end of the 1980s (Fig. 10). If taking the running mean of the previous hypothetical assessment periods of 6 years, hypoxic conditions affected on average about 0.4% of the near-bottom water layer of the Kiel Bay and about 7% of the Bay of Mecklenburg in the 1950s (Fig. 10a). The maximum extent was reached in the 1980s (about 12% and 26%, respectively), before the situation started to improve for the two sub-basins (Fig. 10a). A similar pattern can be observed for the hypoxic water volume (Fig. 10b), which had a share of about 0.04% and 0.9% in the Kiel Bay and Bay of Mecklenburg, respectively, in the 1950s. The maximum extent was equally reached in the 1980s, when about 1.4% (Kiel Bay) and 4.1% (Bay of Mecklenburg) of the water volume were affected by hypoxia.
Modeled average annual dissolved oxygen concentrations (DO) in the near-bottom water layer for assessment periods of 6 years from 1951 to 2019 in the western Baltic Sea. Black lines indicate sub-basin divisions according to HELCOM (KB, Kiel Bay; BM, Bay of Mecklenburg; AB, Arkona Basin; PB, Pomeranian Bay). Country borders in gray
Looking at oxygen deficiency situations in previous hypothetical assessment periods, our model results show that only the Bay of Mecklenburg exhibited near-bottom areas with average annual oxygen concentrations below 6 mg/l in the 1950s. This area, which accounted for about 25% of the near-bottom water layer in the 1950s, doubled to about 51% over the next 50 years before declining over the last two assessment periods, reaching a share of about 38% in the 2016 to 2019 period (Fig. 9, SI 6). A similar trend can be observed for the Kiel Bay, with maximum values in the 1980s (28%) and 2000s (24%). Overall, there seems to be no clear trend for the last two assessment periods (Fig. 9). The situation seemed to improve slightly for some areas (Kiel Bay, Bay of Mecklenburg), whereas it worsens in others (Arkona Basin, Pomeranian Bay). In general, average annual oxygen concentrations did not fall below critical levels in the Pomeranian Bay (Fig. 9, SI 6). Nevertheless, one has to consider that those values are rather conservative estimates, as the annual average, including non-critical periods, was taken.
4 Discussion
4.1 Model Performance and Potential Improvements of ERGOM-MOM
The results of the model evaluation indicate that the model is well able to reproduce the seasonal cycle sufficiently for our analysis (Fig. 2). The spatial gradients in the western Baltic Sea are also well simulated (Fig. 3). Despite a general good fit, there are several shortcomings of the model results. The autumn reoxygenation of the bottom waters takes place too slow, resulting in a period with substantially too low oxygen concentrations between October and February.
Comparing the model results with salinity measurements (SI 1) points to an overestimation of the stratification in the model, what might result in a too weak vertical transport of oxygen during late autumn and winter months. These too high salinities occur purely in the western-most basins of the utilized model setup, which on the other hand is well able to simulate the large-scale salinity dynamics and major Baltic inflows [37].
Another critical aspect in the model simulation might be that the seasonal cycle of the oxygen consumption in the bottom water and sediments is disturbed. The modeled oxygen concentrations are too high in spring and too low in autumn. This aspect can be improved in future by incorporating a more sophisticated sediment module [56]. This may help to improve the modeled oxygen dynamics at the water sediment interface, which vary spatially and temporally in the western Baltic Sea [57]. It would even allow to incorporate more information (like the sediment type), processes (like community bioturbation potential; [58]), or oxygen producers (like eelgrass, [59]), so that the total oxygen fluxes and the seasonal cycle may get improved in future.
Further, the model does not reflect the measured oxygen data everywhere in the western Baltic Sea, especially in coastal areas. This is probably caused by the complicated topography of the western Baltic Sea, where fine structures are not represented by the current horizontal model resolution of three nautical miles. Using a finer horizontal resolution (e.g., 1 NM) is therefore expected to improve both these fine structures and the inflows of high saline waters into the western Baltic Sea.
On the other hand, observing near-bottom oxygen minima is difficult as the area with very low oxygen concentrations is limited to a thin layer above the bottom (sometimes related to the “fluffy layer”; [60]), which is barely accessible with in situ measurements. This fact is also reflected in the definition of near-bottom oxygen by HELCOM. Near-bottom oxygen is defined as the oxygen concentration at lowest possible sampling depth using CTD and/or water sampling bottles which is 0.5 to 1 m above the bottom but can be up to 4 m in deep basins [16]. This makes a direct comparison difficult and may explain why the measured oxygen concentrations are mostly above the model results, especially in the mud-dominated areas, like Bay of Mecklenburg.
4.2 Uncertainties and Limitations
Like any other biogeochemical model, ERGOM-MOM is only a simplification of the real world. Despite its shortcomings and possible future improvements, the current set-up allows a consistent analysis of oxygen deficiency in the western Baltic Sea and to provide insights into its spatio-temporal dynamics. Since the objective of this study is to evaluate how the model simulation results can be used for a better monitoring (aim II) and indicator development (aim III), uncertainties in absolute values are of minor importance, although they should be acknowledged here.
Model underestimations of the oxygen concentration result in an overestimation of critical oxygen situations in time and space. Thereby, underestimations which are below the considered critical oxygen levels are problematic in our context and thus absolute values of oxygen metrics for the Kiel Bay and the Bay of Mecklenburg should be considered with care (Fig. 2). As the bias between modeled and observed values seems to be non-uniform across stations (Fig. 2), a quantitative assessment is difficult and requires closer consideration in future studies.
The strongest differences in near-bottom oxygen concentrations appear mainly along the coasts. Since seasonal oxygen deficiencies occur in deeper areas, the derived oxygen indicator metrics (aim III) are only slightly affected in these areas. In addition, uncertainty resulting from a temporal shift between modeled and observed data (SI 2) is of little importance when considered on an annual basis as we did for the metrics. The overestimation of vertical stratification in late autumn/winter is more problematic, which leads to a general underestimation of predicted annual average oxygen concentrations as well as calculated annual average values for the assessed oxygen indicator metrics. A very positive aspect of the model results is that the oxygen concentration calculated by the model for the near-bottom layer agrees very well with the absolute minimum values of the measurements during summer and autumn (Fig. 2). These values are thus directly usable and of high practical relevance.
Since the over- or underestimation of modeled oxygen concentrations occurs throughout the simulation period, the relative changes between years and thus the long-term trends derived from the model remain unaffected (Figs. 9, 10), allowing conclusions to be drawn about suitable reference periods for deriving target values. However, because the absolute values of the calculated metrics are overestimated by the model, our current approach is not yet suitable for deriving reference and related target values (aim IV).
4.3 Added Value of Model Results for an Oxygen Monitoring
The non-permanently stratified shallow near-shore areas of the western Baltic Sea are subject to completely different oxygen dynamics than those of the deep basins of the Baltic Sea exhibiting permanent oxygen deficiency. Our model results of the western Baltic Sea emphasize the high variability of oxygen concentrations on both, the spatial as well as the temporal scale.
To assess the status or trends of hypoxia or oxygen deficiency, a representative spatial coverage of measurements is required. The bottom structure of the western Baltic Sea exhibits fine-scale structures such as channels, local sills, and basins, which favors an uneven distribution of the specifically denser, oxygen-depleted bottom water. Especially in these areas, better spatial coverage of measurements would be necessary to perform reliable interpolations for area calculations. For example, in the water bodies managed by Germany one measuring station covers on average an area of approximately 300 km2 (about 52 measuring stations are currently located within an assessment area of 15,518 km2; [16]). With our model a ten times higher resolution (about 30 km2) is achieved. For volume calculations, the situation is aggravated by the fact that even less data from vertical profiles is available. Despite current model limitations, model results are available in daily resolution over the entire water depth, allowing to better reproduce the 3-dimensional spreading of oxygen deficiency areas than it could be achieved by interpolations of single-point measurements, even if a vertical profile is fully resolved for the latter (SI 2).
In addition to sufficient spatial coverage, a high temporal resolution is important, especially for the development of biologically important metrics such as consecutive days and frequency of hypoxic and oxygen deficiency situations. With regard to observational data only in Sweden the entire year is currently considered for monitoring purposes. The German and Danish oxygen assessment includes the months from July to November, Poland from June to September, and Estonia, for example, assess the period from May to October. The peak months August and September of hypoxia [14] are covered by all approaches but critical periods can range from June to December [26]. Moreover, if the hypoxic period of an area is shorter than the defined critical period, the hypoxic situation in that area is likely to be underestimated in an assessment that aggregates data over time. Hence, seasonal differences among assessment units should be considered in an oxygen assessment. Due to the high effort involved, current monitoring methods can only provide sporadic daily measurements over weeks, months, or years. In order to get information about whether hypoxic or oxygen deficiency situations can become dangerous for organisms, daily resolved data at least for the critical period is required.
For the oxygen-depleted near-bottom water layer, we pragmatically defined the critical period for hypoxic and oxygen deficiency situations when at least 10% of the area reached values below the established oxygen threshold. However, if reference values and related thresholds are defined in future, this value can be adapted accordingly. Other definitions are also applied to account for differences in seasonality. For example, Bever et al. [30] calculated the duration of hypoxia as the time during which the hypoxic volume is greater than 2 km3. Conley et al. [14] used the notion of a seasonal window, where seasonal trends in bottom oxygen are decoupled from seasonal trends in surface water. A definition that considers stratification behavior seems appropriate, since oxygen exchange processes between the warm surface water and the cold bottom water are reduced with the onset of seasonal stratification.
4.4 Assessment of Ecological Relevant Oxygen Indicator Metrics
Information on the oxygen depleted area or water volume is already helpful to estimate the extent and thus affected habitats by oxygen deficiency. Besides, the application of different critical thresholds is useful to make comprehensive evaluations on the associated negative consequences for fisheries as well as benthic communities. The selected critical thresholds of 2, 4, and 6 mg/l all have their justification. Whereas oxygen values below 2 mg/l have direct negative impacts on fishes, oxygen levels below 4 mg/l negatively affect a large number of benthic organisms [55]. For example, in Germany, in seasonally stratified sea areas, the threshold is set at 4 mg/l oxygen, which roughly corresponds to the precautionary oxygen level of 4.6 mg/l suggested by Vaquer-Sunyer and Duarte [55]. At this level, most of the population is expected to be maintained and catastrophic mortality events avoided. Taking further into account that measurements are not directly on or in the sediment, the target values describing a good oxygen status should be set not lower than 4 mg/l. In addition, the threshold of 6 mg/l is applied in areas where a good vertical mixing is assumed, i.e., where the depth is not sufficient for the formation of a stable thermohaline stratification during summer months [16, 61]. The reason for this is that low oxygen concentrations are very rare in these water bodies and values dropping below this threshold should already be seen as a warning signal [27]. Given a projected warmer Baltic in the future and that oxygen solubility, supply, and demand are temperature dependent, the application of a critical level of 6 mg/l for precautionary purposes is indeed reasonable. With reliable data on benthic communities and habitats, another approach could be the application of area specific thresholds depending on the occurrence of predominating benthic species and the oxygen concentrations required for their healthy life cycle.
In order to evaluate the consequences of oxygen deficiency on the ecological status of a water body as a whole, the duration and frequency must be considered in addition to the extent and intensity. Our analysis of the frequency of days with oxygen concentrations below critical levels in the near-bottom water layer (Figs. 7 and SI 4) already allows to draw conclusions about vulnerable areas. Conley et al. [14] followed a comparable approach utilizing observational data. Based on the frequency of profiles where oxygen concentrations were below 2 mg/l, the study distinguished between episodic, seasonal, and persistent hypoxia. According to this approach, for the assessment period of 2011–2016 near-bottom areas with episodic hypoxia (< 2 mg/l) would be present in Kiel Bay, the Arkona Basin, and the Pomeranian Bay. Only in the Bay of Mecklenburg about 30% of the area would exhibit seasonal hypoxia (Fig. 7). Besides the Pomeranian Bay, seasonal oxygen deficiency (< 6 mg/l) occurs accordingly in all of the investigated assessment units (SI 4).
Although such analysis on the frequency helps to classify the severity of the phenomena in an area, more important for the assessment of adverse effects on biodiversity is the analysis of time-spans where oxygen levels dropped below critical levels. In Kiel Bay, for example, large near-bottom areas experience hypoxia for more than 2 days about 2 to 6 times per year according to our model results (Fig. 8). Large areas of the Bay of Mecklenburg even exhibit hypoxia for more than 2 weeks about 2 to 4 times per year. It is known that hypoxia lasting several weeks can cause a collapse of benthic communities due to oxygen deficiency or the release of toxic hydrogen sulfide from sediments [18, 62]. There are also existing studies where the effects of the duration of hypoxia on benthic communities in the Baltic Sea have been investigated. For example, a high bacterial diversity dropped after 48 days of oxygen deficiency and a regime shift from aerobic to obligate anaerobic bacterial taxa was observed within a field experiment in the northern Baltic Sea [63]. Another in situ experiment in this area focusing on macrobenthos showed that the benthic fauna became severely stressed already after only 3 days of hypoxia, but it was also acknowledged that the macrozoobenthos recovers quickly after a short duration of hypoxia [64]. After 7 days of exposure to hypoxia, the authors observed patterns in the communities indicating a loss in its adaptive capacity. Altered or lost ecosystem functionality was predicted to occur after a duration between 7 and 48 days of hypoxia [64]. In Danish coastal waters, changes in benthic communities were investigated after a severe hypoxic event in 2002 [65]. Here, a decrease in species diversity was evident after only 1 week of hypoxia. The gradual decrease in species diversity during the first 2 to 4 weeks of hypoxia exposure indicated that the benthic community underwent significant changes before hypoxia led to community collapse after 4 to 6 weeks [65]. That also short reoccurring periods of hypoxia can negatively influence benthic communities was further shown by a study of Villnäs et al. [66] which observed a gradual deterioration of macrobenthic functions such as secondary biomass production and bioturbation with reoccurring pulse disturbances lasting 3 days.
The field experiments support the ecological relevance of our chosen time periods for hypoxic situations (2, 7, 14 days) based on laboratory experiments and representing lethal times under acute hypoxia for a range of organisms [55]. To the best of our knowledge, no literature is currently available on the impact of higher critical oxygen levels on organism lethality, so we took a pragmatic approach and extended the time periods accordingly. However, whether these are applicable to make a boundary between good, i.e., high biodiversity and intact ecosystem services, and poor environmental status needs further investigation.
4.5 Long-Term Analysis of Oxygen Deficiency
The long-term analysis of the percentage near-bottom water layer (and the water volume) affected by hypoxic conditions indicates for the Kiel Bay and Bay of Mecklenburg that the affected areas were at similar levels from about 1950 to 1960, despite annual fluctuations, before steadily increasing until the end of 1980s (Fig. 10). Our results agree with national data of Germany where seasonal hypoxia has been observed almost annually since the 1980s in the Kiel Bay and Bay of Mecklenburg [16]. A report on an overall significant long-term increase of hypoxia measured from 1955 to 2009 at coastal sites in the Baltic Sea further supports our findings [14]. The observed trend reversal from the twenty-first century onwards should nevertheless be viewed critically considering current model limitations and can hardly be supported by observations due to the insufficient spatial and temporal resolution of the monitoring. The development of Baltic Sea hypoxia is often only assessed for the central parts [7]. The recent reanalysis of Kõuts et al. [8] may hint to a slight decrease of the area with a higher risk of hypoxia in the Bay of Mecklenburg, although trends differ between the available datasets [9]. On the other hand, the trend reversal contradicts recent reports of increasing oxygen consumption which in turn would lead to higher risks of hypoxia [67]. Moreover, looking at the time-series of the annual average oxygen concentrations for the Arkona Basin (Fig. 9) there seems to be no improvement. Thus, further research is needed to clarify if improvements in the hypoxic near-bottom water layer and water volume are a result of nutrient input reductions as agreed to in 1988 [68]. In addition, the effects of climate change on oxygen concentrations will also need to be studied in the future, which could counteract potential improvements. A proposed warmer Baltic Sea in the future is expected to favor hypoxic conditions due to increased water temperature, microbial activity, and river runoff [12, 69].
Nevertheless, our long-term analysis shows that the relatively stable situation in the 1950s could indicate a situation with non-eutrophication influenced oxygen concentrations and its natural fluctuations. In order to have a comparable approach as for the HELCOM oxygen debt indicator, a somewhat more distant period for the analysis would have been desirable. In the current discussions of the HELCOM working group on the oxygen indicator, later periods are also considered as reference periods, e.g., for the Bothnian Sea and Bothnian Bay (personal communication). In addition, other authors point to a still good ecological status in the 1950s and early 1960s for nutrients and chlorophyll-a [1, 33].
If the current model limitations mentioned above can be overcome in further model runs, then the analysis of past conditions from model runs will help to derive water quality targets for the oxygen indicator. Thereby, our current approach did not consider future climate scenarios, which would be of high importance considering the proposal of water quality targets for environmental assessments.
5 Conclusion
Our analyses show that seasonal oxygen deficiency and the related metrics are indeed a suitable indicator for describing the ecological status of the western Baltic Sea, where seasonal and episodic rather than perennial hypoxia and oxygen deficiency occurs. Likewise to nutrients and chlorophyll-a, the situation in the 1950s may be regarded as “good status,” including naturally occurring oxygen deficits in the western Baltic Sea. Oxygen deficiency has a seasonal and a spatial dimension, which both should be considered in defining a suitable indicator. The use of model simulations allows us to add these dimensions. By providing further information on the duration and recurrence interval of oxygen deficiency, the proposed metrics serve as an interface to the most important ecological drivers for benthic biology, so that they can be related to the Baltic Sea Action Plan eutrophication goal of “natural distribution and occurrences of plants and animals.” However, model simulations always have some shortcomings and should not be used alone to derive thresholds. Instead, an integrated approach is needed to combine model results with observations in a suitable way so that seasonal oxygen deficiency can be used as a meaningful eutrophication indicator in future.
Data Availability
The simulation datasets analyzed during the current study are available in the IOW THREDDS repository, https://thredds-iow.io-warnemuende.de/thredds/catalogs/projects/integral/catalog_integral.html. Other datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Carstensen, J., & Conley, D. J. (2019). Baltic Sea hypoxia takes many shapes and sizes. Limnology and Oceanography Bulletin, 28, 125–129. https://doi.org/10.1002/lob.10350
Meier, H. E. M., Eilola, K., Almroth-Rosell, E., et al. (2019). Disentangling the impact of nutrient load and climate changes on Baltic Sea hypoxia and eutrophication since 1850. Climate Dynamics, 53, 1145–1166. https://doi.org/10.1007/s00382-018-4296-y
Reissmann, J. H., Burchard, H., Feistel, R., et al. (2009). Progress in Oceanography Vertical mixing in the Baltic Sea and consequences for eutrophication – A review. Progress in Oceanography, 82, 47–80. https://doi.org/10.1016/j.pocean.2007.10.004
Carstensen, J., Andersen, J. H., Gustafsson, B. G., & Conley, D. J. (2014). Deoxygenation of the Baltic Sea During the Last Century, 111, 5628–5633. https://doi.org/10.1073/pnas.1323156111
HELCOM. (2018a). State of the Baltic Sea – Second HELCOM holistic assessment 2011–2016. Baltic Sea Environment Proceedings, 155. Retrieved May 19, 2022, from https://helcom.fi/baltic-sea-trends/holistic-assessments/state-of-the-baltic-sea-2018/reports-and-materials/
HELCOM. (2018b). HELCOM thematic assessment of eutrophication 2011–2016. Baltic Sea Environment Proceedings no. 156. Available at: https://helcom.fi/baltic-sea-trends/holistic-assessments/state-of-the-baltic-sea-2018/reports-and-materials/
Almroth-Rosell, E., Wåhlström, I., Hansson, M., et al. (2021). A regime shift toward a more anoxic environment in a eutrophic sea in Northern Europe. Frontiers in Marine Science. https://doi.org/10.3389/fmars.2021.799936
Kõuts, M., Maljutenko, I., Elken, J., et al. (2021). Recent regime of persistent hypoxia in the Baltic Sea. Environmental Research Communications, 3, 075004. https://doi.org/10.1088/2515-7620/AC0CC4
Krapf, K., Naumann, M., Dutheil, C., & Meier, H. E. M. (2022). Investigating hypoxic and euxinic area changes based on various datasets from the Baltic Sea. Frontiers in Marine Science. https://doi.org/10.3389/fmars.2022.823476
Vahtera, E., Conley, D. J., Gustafsson, B. G., et al. (2007). Internal ecosystem feedbacks enhance nitrogen-fixing cyanobacteria blooms and complicate management in the Baltic Sea. Ambio, 36, 186–194. https://doi.org/10.1579/0044-7447(2007)36[186:IEFENC]2.0.CO;2
Carstensen, J., Conley, D. J., Bonsdorff, E., et al. (2014). Hypoxia in the Baltic Sea: Biogeochemical cycles, benthic fauna, and management. Ambio, 43, 26–36. https://doi.org/10.1007/s13280-013-0474-7
HELCOM. (2021a). Climate change in the Baltic Sea. 2021 fact sheet. Baltic Sea environment proceedings n°180. HELCOM/Baltic Earth 2021. Retrieved May 19, 2022, from https://helcom.fi/baltic-sea-climate-change-fact-sheet-new-publication-shows-latest-scientific-knowledge-on-climate-change-in-the-baltic-sea/
Liblik, T., & Lips, U. (2019). Stratification has strengthened in the Baltic Sea – An analysis of 35 years of observational data. Frontiers in Earth Science, 7, 1–15. https://doi.org/10.3389/feart.2019.00174
Conley, D. J., Carstensen, J., Aigars, J., et al. (2011). Hypoxia is increasing in the coastal zone of the Baltic Sea. Environmental Science and Technology, 45, 6777–6783. https://doi.org/10.1021/es201212r
Reusch, T. B. H., Dierking, J., Andersson, H. C., et al. (2018). The Baltic Sea as a time machine for the future coastal ocean. Science Advances. https://doi.org/10.1126/sciadv.aar8195
BLANO. (2018). Zustand der deutschen Ostseegewässer 2018. Aktualisierung der Anfangsbewertung nach § 45c, der Beschreibung des guten Zustands der Meeresgewässer nach § 45d und der Festlegung von Zielen nach § 45e des Wasserhaushaltsgesetzes zur Umsetzung der Meeresstrategie-Rahmenrichtlinie. Anlage 1: Ergänzende nationale Indikatorblätter. Retrieved August 29, 2022, from https://www.meeresschutz.info/berichte-art-8-10.html?file=files/meeresschutz/berichte/%0bart8910/zyklus18/Zustandsbericht_Ostsee_2018.pdf.
Virtanen, E. A., Norkko, A., Nyström Sandman, A., & Viitasalo, M. (2019). Identifying areas prone to coastal hypoxia – The role of topography. Biogeosciences, 16, 3183–3195. https://doi.org/10.5194/bg-16-3183-2019
Diaz, R. J., & Rosenberg, R. (1995). Marine benthic hypoxia: A review of its ecological effects and the behavioural responses of benthic macrofauna. Oceanography and Marine Biology an Annual Review, 33(33), 245–303., 33(33), 245–303.
Zettler, M. L., Friedland, R., Gogina, M., & Darr, A. (2017). Variation in benthic long-term data of transitional waters: Is interpretation more than speculation? PLoS ONE, 12, e0175746. https://doi.org/10.1371/journal.pone.0175746
Garlo, E. V., Milstein, C. B., & Jahn, A. E. (1979). Impact of hypoxic conditions in the vicinity of Little Egg Inlet, New Jersey in summer 1976. Estuarine and Coastal Marine Science, 8, 421–432. https://doi.org/10.1016/0302-3524(79)90059-8
Cardinale, M., & Modin, J. (1999). Changes in size-at-maturity of Baltic cod (Gadus morhua) during a period of large variations in stock size and environmental conditions. Fisheries Research, 41, 285–295. https://doi.org/10.1016/S0165-7836(99)00021-1
Diaz, R. J., & Rosenberg, R. (2008). Spreading dead zones and consequences for marine ecosystems. Science, (80-)321:926–929. https://doi.org/10.1126/science.1156401
HELCOM. (2021b). Baltic Sea Action Plan – 2021 update. Adopted on 20 October 2021 in Lübeck, Germany by the HELCOM Ministerial Meeting. Available at: https://helcom.fi/baltic-sea-action-plan
HELCOM. (2007). HELCOM Baltic Sea Action Plan. Adopted on 15 November 2007 in Krakow, Poland by the HELCOM Extraordinary Ministerial Meeting. Retrieved May 19, 2022, from https://www.helcom.fi/wp-content/uploads/2019/08/BSAP_Final.pdf
HELCOM. (2018c). HELCOM core indicator report – Oxygen debt. Retrieved May 19, 2022, from https://helcom.fi/baltic-sea-trends/indicators/
HELCOM. (2020). HELCOM indicator on near-bottom oxygen concentrations in shallow waters - Towards a Baltic Sea wide application IN-EUTROPHICATION 16–2020. Retrieved May 19, 2022, from https://portal.helcom.fi/meetings/IN-EUTROPHICATION%2016-2020-694/MeetingDocuments/Forms/AllItems.aspx
HELCOM. (2021c). German national indicator on near-bottom oxygen concentrations. IN-EUTROPHICATION 19A-2021. Retrieved August 29, 2022, from https://portal.helcom.fi/meetings/IN-EUTROPHICATION%2019A-2021-887/MeetingDocuments/2-1%20German%20national%20indicator%20on%20near-bottom%20oxygen%20concentrations.pdf
HELCOM. (2016). Shallow-water oxygen candidate indicator – Draft description of methodology. State & Conservation 5–2016. Available at: https://portal.helcom.fi/meetings/STATE%20-%20CONSERVATION%205-2016-363/MeetingDocuments/4J-31%20Shallow-water-oxygen%20candidate%20indicator%20-%20draft%20description%20of%20methdology.pdf
Sveegaard, S. (Ed.). (2020). Danish scientific appraisal of HELCOM’s and OSPAR’s indicators in relation to the monitoring programme under the Marine Strategy Framework Directive – Status 2020. Aarhus University, DCE – Danish Centre for Environment and Energy, 114 pp. Technical report no. 168. Retrieved August 29, 2022, from http://dce2.au.dk/pub/TR168.pdf
Bever, A. J., Friedrichs, M. A. M., Friedrichs, C. T., et al. (2013). Combining observations and numerical model results to improve estimates of hypoxic volume within the Chesapeake Bay, USA. The Journal of Geophysical Research, 118, 4924–4944. https://doi.org/10.1002/jgrc.20331
Conley, D. J., Bj̈orck S, Bonsdorff E, et al. (2009). Hypoxia-related processes in the Baltic Sea. Environmental Science and Technology, 43, 3412–3420. https://doi.org/10.1021/es802762a
Neumann, T., Koponen, S., Attila, J., et al. (2021). Optical model for the Baltic Sea with an explicit CDOM state variable: A case study with model ERGOM (version 1.2). Geoscientific Model Development, 14, 5049–5062. https://doi.org/10.5194/gmd-14-5049-2021
Neumann, T., & Schernewski, G. (2008). Eutrophication in the Baltic Sea and shifts in nitrogen fixation analyzed with a 3D ecosystem model. Journal of Marine Systems, 74, 592–602. https://doi.org/10.1016/j.jmarsys.2008.05.003
Friedland, R., Neumann, T., & Schernewski, G. (2012). Climate change and the Baltic Sea action plan: Model simulations on the future of the western Baltic Sea. Journal of Marine Systems, 105–108, 175–186. https://doi.org/10.1016/j.jmarsys.2012.08.002
Meier, H. E. M., Andersson, H. C., Arheimer, B., et al. (2012). Comparing reconstructed past variations and future projections of the Baltic Sea ecosystem—First results from multi-model ensemble simulations. Environmental Research Letters, 7, 034005. https://doi.org/10.1088/1748-9326/7/3/034005
Meier, H. E. M., Andersson, H. C., Eilola, K., et al. (2011). Hypoxia in future climates: A model ensemble study for the Baltic Sea. Geophysical Research Letters, 38:n/a-n/a. https://doi.org/10.1029/2011GL049929
Neumann, T., Radtke, H., & Seifert, T. (2017). On the importance of major Baltic inflows for oxygenation of the central Baltic Sea. Journal of Geophysical Research Ocean, 122, 1090–1101. https://doi.org/10.1002/2016JC012525
Kuznetsov, I., & Neumann, T. (2013). Simulation of carbon dynamics in the Baltic Sea with a 3D model. Journal of Marine Systems, 111–112, 167–174. https://doi.org/10.1016/j.jmarsys.2012.10.011
Griffies, S. M., Harrison, M. J., Pacanowski, R. C., & Rosati, A. (2004). A technical guide to MOM4. GFDL Ocean Group technical report no. 5. NOAA/Geophysical Fluid Dynamics Laboratory. Version prepared on August 23, 2004. Available online at: www.gfdl.noaa.gov
Griffies, S. M. (2012). Elements of the Modular Ocean Model (MOM) - 2012 release with updates. GFDL Ocean Group technical report no. 7. NOAA/Geophysical Fluid Dynamics Laboratory.
Winton, M. (2000). A reformulated three-layer sea ice model. The Journal of Atmospheric and Oceanic Technology, 17, 525–531. https://doi.org/10.1175/1520-0426(2000)017%3c0525:ARTLSI%3e2.0.CO;2
Weisse, R., von Storch, H., Callies, U., et al. (2009). Regional meteorological–marine reanalyses and climate change projections. Bulletin of the American Meteorological Society, 90, 849–860. https://doi.org/10.1175/2008BAMS2713.1
Geyer, B., & Rockel, B. (2013). coastDat-2 COSMO-CLM atmospheric reconstruction. World Data Center for Climate (WDCC) at DKRZ. Available at: https://doi.org/10.1594/WDCC/coastDat-2_COSMO-CLM
HELCOM. (2015). Updated fifth Baltic Sea pollution load compilation (PLC-5.5). Baltic Sea environment proceedings no. 14. Retrieved May 19, 2022, from https://helcom.fi/baltic-sea-trends/pollution-load-compilations/
HELCOM (2021d) Input of nutrients by the seven biggest rivers in the Baltic Sea region 1995-2017. Baltic Sea Environment Proceedings No. 178. Available at: https://helcom.fi/wp-content/uploads/2021/09/The-seven-biggest-rivers-in-the-Baltic-Sea-Region.pdf
Gustafsson, B. G., Schenk, F., Blenckner, T., et al. (2012). Reconstructing the development of Baltic Sea eutrophication 1850–2006. Ambio, 41, 534–548. https://doi.org/10.1007/s13280-012-0318-x
Gauss, M., Bartnicki, J., & Klein, H. (2019). Atmospheric nitrogen deposition to the Baltic Sea, Oslo. Retrieved May 19, 2022, from https://emep.int/publ/helcom/2018/index.html.
Gauss, M., Bartnicki, J., Jalkanen, J., et al. (2021). Airborne nitrogen deposition to the Baltic Sea: Past trends, source allocation and future projections. Atmospheric Environment, 253, 118377. https://doi.org/10.1016/j.atmosenv.2021.118377
Ruoho-Airola, T., Eilola, K., Savchuk, O. P., et al. (2012). Atmospheric nutrient input to the Baltic Sea from 1850 to 2006: A reconstruction from modeling results and historical data. Ambio, 41, 549–557. https://doi.org/10.1007/s13280-012-0319-9
Eilola, K., Gustafsson, B. G., Kuznetsov, I., et al. (2011). Evaluation of biogeochemical cycles in an ensemble of three state-of-the-art numerical models of the Baltic Sea. Journal of Marine Systems, 88, 267–284. https://doi.org/10.1016/j.jmarsys.2011.05.004
Meier, H. E. M., Edman, M. K., Eilola, K. J., et al. (2018). Assessment of eutrophication abatement scenarios for the Baltic Sea by multi-model ensemble simulations. Frontiers in Marine Science, 5, 1–24. https://doi.org/10.3389/fmars.2018.00440
Neumann, T., Siegel, H., & Gerth, M. (2015). A new radiation model for Baltic Sea ecosystem modelling. Journal of Marine Systems, 152, 83–91. https://doi.org/10.1016/j.jmarsys.2015.08.001
Neumann, T., Siegel, H., Moros, M., et al. (2020). Ventilation of the northern Baltic Sea. Ocean Science, 16, 767–780. https://doi.org/10.5194/os-16-767-2020
Leipe, T., Naumann, M., Tauber, F., et al. (2017). Regional distribution patterns of chemical parameters in surface sediments of the south-western Baltic Sea and their possible causes. Geo-Marine Letters, 37, 593–606. https://doi.org/10.1007/s00367-017-0514-6
Vaquer-Sunyer, R., & Duarte, C. M. (2008). Thresholds of hypoxia for marine biodiversity. Proceedings of the National Academy of Sciences, 105, 15452–15457. https://doi.org/10.1073/pnas.0803833105
Radtke, H., Lipka, M., Bunke, D., et al. (2019). Ecological ReGional Ocean Model with vertically resolved sediments (ERGOM SED 1.0): Coupling benthic and pelagic biogeochemistry of the south-western Baltic Sea. Geosciences Model Development, 12, 275–320. https://doi.org/10.5194/gmd-12-275-2019
Lipka, M., Woelfel, J., Gogina, M., et al. (2018). Solute reservoirs reflect variability of early diagenetic processes in temperate brackish surface sediments. Frontiers in Marine Science, 9, 1–20. https://doi.org/10.3389/fmars.2018.00413
Gogina, M., Lipka, M., Woelfel, J., et al. (2018). In search of a field-based relationship between benthic macrofauna and biogeochemistry in a modern brackish coastal sea. Frontiers in Marine Science, 5, 1–18. https://doi.org/10.3389/fmars.2018.00489
Schubert, P. R., Hukriede, W., Karez, R., et al. (2015). Mapping and modeling eelgrass Zostera marina distribution in the western Baltic Sea. Marine Ecology Progress Series, 522, 79–95. https://doi.org/10.3354/meps11133
Löffler, A., Leipe, T., & Emeis, K. C. (2000). The “fluffy layer” in the Pomeranian Bight (western Baltic Sea): Geochemistry, mineralogy and environmental aspects. Meyniana, 52, 85–86.
LLUR. (2020). LLUR-Bericht zu den Sauerstoffuntersuchungen in der Westlichen Ostsee im Sept. 2020. Retrieved November 14, 2022, from https://www.schleswig-holstein.de/DE/Fachinhalte/M/meeresschutz/Downloads/Bericht_LLUR_Sauerstoff_2020.pdf
Janas, U., Wocial, J., & Szaniawska, A. (2004). Seasonal and annual changes in the macrozoobenthic populations of the Gulf of Gdańsk with respect to hypoxia and hydrogen sulphide. Oceanologia, 46, 85–102.
Sinkko, H., Hepolehto, I., Lyra, C., et al. (2019). Increasing oxygen deficiency changes rare and moderately abundant bacterial communities in coastal soft sediments. Science and Reports, 9, 1–14. https://doi.org/10.1038/s41598-019-51432-1
Villnäs, A., Norkko, J., Lukkari, K., et al. (2012). Consequences of increasing hypoxic disturbance on benthic communities and ecosystem functioning. PLoS ONE. https://doi.org/10.1371/journal.pone.0044920
Conley, D. J., Carstensen, J., Ærtebjerg, G., et al. (2007). Long-term changes and impacts of hypoxia in Danish coastal waters. Ecological Applications, 17, 165–184. https://doi.org/10.1890/05-0766.1
Villnäs, A., Norkko, J., Hietanen, S., et al. (2013). The role of recurrent disturbances for ecosystem multifunctionality. Ecology, 94, 2275–2287. https://doi.org/10.1890/12-1716.1
Meier, H. E. M., Väli, G., Naumann, M., et al. (2018). Recently accelerated oxygen consumption rates amplify deoxygenation in the Baltic Sea. The Journal of Geophysical Research Ocean, 123, 3227–3240. https://doi.org/10.1029/2017JC013686
HELCOM. (1988). Declaration on the protection of the marine environment of the Baltic Sea area. Adopted on 15 February 1988 in Helsinki, Finland by the Ministers responsible for the environmental protection in the Baltic Sea States. Retrieved May 19, 2022, from https://helcom.fi/helcom-at-work/ministerial-meetings/1988-helsinki/
Meier, H. E. M., Hordoir, R., Andersson, H. C., et al. (2012). Modeling the combined impact of changing climate and changing nutrient loads on the Baltic Sea environment in an ensemble of transient simulations for 1961–2099. Climate Dynamics, 39, 2421–2441. https://doi.org/10.1007/s00382-012-1339-7
Acknowledgements
We like to thank Marina Carstens (LM-MV), Mario von Weber (LUNG-MV), Hannah Lutterbeck (LLUR-SH), Clarissa Vock, and the HELCOM IN-EUTRO working group for providing data and information.
Funding
Open Access funding enabled and organized by Projekt DEAL. This research was funded by the German Environment Agency (UBA) project “The Baltic Sea Action Plan—Modelling of Water Quality Indicators” (Grant number 3720252020). Supercomputing power was provided by HLRN (North-German Supercomputing Alliance).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Model simulations were carried out by TN. Data collection and analysis were performed by SP and RF. The first draft of the manuscript was written by SP and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Piehl, S., Friedland, R., Heyden, B. et al. Modeling of Water Quality Indicators in the Western Baltic Sea: Seasonal Oxygen Deficiency. Environ Model Assess 28, 429–446 (2023). https://doi.org/10.1007/s10666-022-09866-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10666-022-09866-x