Abstract
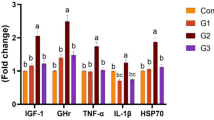
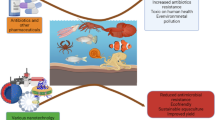
There is an urgent requirement for the improvement of the white leg shrimp, Litopenaeus vannamei; health-related indices; and immunity due to emerging diseases. Recently, probiotics have been playing an important role in L. vannamei health management. Therefore, the current pond trial was to evaluate the probiotic proficiency of commercial probiotic products of THIONIL (THIO) on the enhancement of the water, soil, growth, digestibility, survival, immune-related indices, and susceptibility of L. vannamei to infection. The study was carried out in the major shrimp culturing regions of Kavali, Nellore (Andhra Pradesh), and Ponneri (Tamil Nadu), India. Six groups (lacks/ha) of the experimental L. vannamei were allocated, including a control group (THIO 0%—untreated) and groups containing 2%, 4%, 6%, 8%, and 10% of THIO that were encapsulated with commercial feed (CP Aqua). Bioassays were performed on PLs/ shrimp at various days interval of 0, 5, 25, 50, 100, and 123th to assess productivity, anti-vibrio activity, and digestive enzyme for digestibility, histological and immunological indices, and cytotoxicity in Artemia nauplii. Significant differences were observed in the increased growth (35.71 ± 3.24 g/shrimp) and digestive parameters in 10% THIO-fed shrimp. Although in contrast to the control group, the other THIO-fed prawn groups also displayed appreciable development. The findings showed that, in comparison to the control, the gill, hepatopancreas, and stomach had reduced tissue damage with 10% THIO. Furthermore, Vibrio parahaemolyticus (0.008 × 104 cfu/g) and Vibrio harveyi (0.051 × 105 cfu/g) (vibriosis) were potentially resistant to the 10% THIO-fed group. In addition, THIO-fed prawns (10%) showed significant improvements in immune-related expresses (proPO, SOD, and SOA) in comparison to the control. In conclusion, the findings showed that the THIO treatment prawns significantly improved the quality of their water (pH, ammonia, nitrogen dioxide, hydrogen sulfide, and DO) and soil (Pb, Cr, Hg, Mg, Cu, Fe, and Ni), increased and demonstrated protection against vibrio infections.
Graphical Abstract










Similar content being viewed by others
Data availability
Data will be provided under request.
References
Amoah, K., Huang, Q. C., Tan, B. P., Zhang, S., Chi, S. Y., Yang, Q. H., Liu, H. Y., & Dong, X. H. (2019). Dietary supplementation of probiotic Bacillus coagulans ATCC 7050, improves the growth performance, intestinal morphology, microflora, immune response, and disease confrontation of Pacific white shrimp, Litopenaeus vannamei. Fish & Shellfish Immunology, 87, 796–808. https://doi.org/10.1016/j.fsi.2019.02.029
Amparyup, P., Charoensapsri, W., & Tassanakajon, A. (2013). Prophenoloxidase system and its role in shrimp immune responses against major pathogens. Fish & Shellfish Immunology, 34(4), 990–1001. https://doi.org/10.1016/j.fsi.2012.08.019
Arias-Moscoso, J. L., Espinoza-Barrón, L. G., Miranda-Baeza, A., Rivas-Vega, M. E., & Nieves-Soto, M. (2018). Effect of commercial probiotics addition in a biofloc shrimp farm during the nursery phase in zero water exchange. Aquaculture Reports, 11, 47–52. https://doi.org/10.1016/j.aqrep.2018.06.001
Beauchamp, C., & Fridovich, I. (1971). Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Analytical Biochem, 44(1), 276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Boonyaratpalin, M., Supamattaya, K., Verakunpiriya, V., & Suprasert, D. (2001). Effects of aflatoxin B1 on growth performance, blood components, immune function and histopathological changes in black tiger shrimp (Penaeus monodon Fabricius). Aquacul Res, 32, 388–398. https://doi.org/10.1046/j.1355-557x.2001.00046.x
Bullerwell, C. N., Collins, S. A., Lall, S. P., & Anderson, D. M. (2016). Growth performance, proximate and histological analysis of rainbow trout fed diets containing Camelina sativa seeds, meal (high-oil and solvent-extracted) and oil. Aquacul, 452, 342–350. https://doi.org/10.1016/j.aquaculture.2015.11.008
Chupawa, P., Inchuen, S., Jaisut, D., Ronsse, F., & Duangkhamchan, W. (2023). Effects of stepwise microwave heating and expanded bed height control on the performance of combined fluidized bed/microwave drying for preparing instant brown rice. Food and Bioproc Technol, 16(1), 199–215. https://doi.org/10.1007/s11947-022-02933-x
Dowarah, R., Verma, A. K., Agarwal, N., Singh, P., & Singh, B. R. (2018). Selection and characterization of probiotic lactic acid bacteria and its impact on growth, nutrient digestibility, health and antioxidant status in weaned piglets. PLoS ONE, 13(3), e0192978. https://doi.org/10.1371/journal.pone.0192978
Downs, C. A., Fauth, J. E., & Woodley, C. M. (2001). Assessing the health of grass shrimp (Palaeomonetes pugio) exposed to natural and anthropogenic stressors: A molecular biomarker system. Marine Biotechnol, 3, 380–397. https://doi.org/10.1007/s10126-001-0008-3
Edition, F. (2011). Guidelines for drinking-water quality. WHO Chronicle, 38(4), 104–108.
El-Dakar, A., & Gohar, Y. M. (2004). Use of Bacillus subtilis in microparticulate diets for producing biosecure Peneaus japonicus postlarvae. J Animal and Poul Prod, 29(12), 6853–6871. https://doi.org/10.21608/JAPPMU.2004.239471
Einar Ringø (2020) Probiotics in shellfish aquaculture. Aquacul Fisheri 5(1):1-27https://doi.org/10.1016/j.aaf.2019.12.001
El-Dakar, A. Y., Shalaby, S. M., & Saoud, I. P. (2007). Assessing the use of a dietary probiotic/prebiotic as an enhancer of spinefoot rabbitfish Siganus rivulatus survival and growth. Aquacul Nut, 13(6), 407–412. https://doi.org/10.1111/j.1365-2095.2007.00491.x
Farzanfar, A. (2006). the use of probiotics in shrimp aquaculture. FEMS Immunol Medi Microbiol, 48(2), 149–158. https://doi.org/10.1111/j.1574-695X.2006.00116.x
Franco, R., Martín, L., Arenal, A., Santiesteban, D., Sotolongo, J., Cabrera, H., Mejías, J., Rodríguez, G., Moreno, A. G., Pimentel, E., & Castillo, N. M. (2017). Evaluation of two probiotics used during farm production of white shrimp Litopenaeus vannamei (Crustacea: Decapoda). Aquaculture Research, 48(4), 1936–1950. https://doi.org/10.1111/are.13031
Fuller, R. (1989). Probiotics in man and animals. J Appl Bact, 66, 365–378. https://doi.org/10.1111/j.1365-2672.1989.tb05105.x
Gatesoupe, F. J. (1999). The use of probiotics in aquaculture. Aquaculture, 180, 147–165. https://doi.org/10.1016/S0044-8486(99)00187-8
Ghosh, A. K., Bir, J., Azad, M. A. K., Hasanuzzaman, A. F. M., Islam, M. S., & Huq, K. A. (2016). Impact of commercial probiotics application on growth and production of giant fresh water prawn (Macrobrachium Rosenbergii De Man, 1879). Aquacul Rep, 4, 112–117. https://doi.org/10.1016/j.aqrep.2016.08.001
Goh, J. X. H., Tan, L. T. H., Law, J. W. F., Ser, H. L., Khaw, K. Y., Letchumanan, V., Lee, L. H., & Goh, B. H. (2022). Harnessing the potentialities of probiotics, prebiotics, synbiotics, paraprobiotics, and postbiotics for shrimp farming. Reviews in Aquaculture, 14(3), 1478–1557. https://doi.org/10.1111/raq.12659
Gopal, S., Otta, S. K., Kumar, S., Karunasagar, I., Nishibuchi, M., & Karunasagar, I. (2005). The occurrence of Vibrio species in tropical shrimp culture environments; implications for food safety. International Journal of Food Microbiology, 102, 151–159. https://doi.org/10.1016/j.ijfoodmicro.2004.12.011
Gullian, M., Thompson, F., & Rodriguez, J. (2004). Selection of probiotic bacteria and study of their immunostimulatory effect in Penaeus vannamei. Aquacul, 233, 1–14. https://doi.org/10.1016/j.aquaculture.2003.09.013
Hernández-López, J., Gollas-Galván, T., & Vargas-Albores, F. (1996). Activation of the prophenoloxidase system of the brown shrimp Penaeus californiensis Holmes. Comparative Biochemistry and Physiology Part c: Toxicology & Pharmacology, 113, 61–66. https://doi.org/10.1016/0742-8413(95)02033-0
Hill, C., Guarner, F., Reid, G., Gibson, G. R., Merenstein, D. J., Pot, B., Morelli, L., Canani, R. B., Flint, H. J., Salminen, S., & Calder, P. C. (2014). The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nature Reviews Gastroenterol Hepatol, 11, 506–514. https://doi.org/10.1038/nrgastro.2014.66
Hogg N (1998) December. Free radicals in disease. In Seminars in reproductive endocrinology 16: 241-248. Copyright© 1998 by Thieme Medical Publishers, Inc.https://doi.org/10.1055/s-2007-1016284
Hoseinifar SH, Sun YZ, Wang A, Zhou Z (2018) Probiotics as means of diseases control in aquaculture, a review of current knowledge and future perspectives. Front in Microbiol 2429. https://doi.org/10.3389/fmicb.2018.02429.
Johnson PT (1980) Histology of the blue crab, Callinectes sapidus: A model for the Decapoda. (No Title). https://lccn.loc.gov/80013083.
Koppenol, W. H., Moreno, J. J., Pryor, W. A., Ischiropoulos, H., & Beckman, J. S. (1992). Peroxynitrite, a cloaked oxidant formed by nitric oxide and superoxide. Chemical Research in Toxicology, 5(6), 834–842. https://doi.org/10.1021/tx00030a017
Krishna, N. M., & Philip, L. (2005). Thiobacillus denitrificans immobilized biotrickling filter for NO 2 removal. Clean Technol Envi Pol, 7, 285–293. https://doi.org/10.1007/s10098-005-0003-x
Kumar, V., Roy, S., Meena, D. K., & Sarkar, U. K. (2016). Application of probiotics in shrimp aquaculture: Importance, mechanisms of action, and methods of administration. Rev Fisheri Sci Aquacul, 24, 342–368. https://doi.org/10.1080/23308249.2016.1193841
Lee, S. Y., & Lee, B. H. (1990). Esterolytic and lipolytic activities of Lactobacillus casei-subsp-Casei LLG. Journal of Food Science, 55(1), 119–122. https://doi.org/10.1111/j.1365-2621.1990.tb06031.x
Li, Y., Liu, H., Dai, X., Li, J., & Ding, F. (2018). Effects of dietary inulin and mannan oligosaccharide on immune related genes expression and disease resistance of Pacific white shrimp, Litopenaeus vannamei. Fish & Shellfish Immunology, 76, 78–92. https://doi.org/10.1016/j.fsi.2018.02.034
Martinez-Porchas, M., & Martinez-Cordova, L. R. (2012). World aquaculture: Environmental impacts and troubleshooting alternatives. The Sci World J, 389623, 1–9. https://doi.org/10.1100/2012/389623
Mustafa Kamal, A. H., Hishamuddin, O., & Boyd, C. E. (2018). Physical and chemical characteristics of soil from tiger shrimp aquaculture ponds at Malacca Malaysia. J App Aquacul, 30(1), 47–62. https://doi.org/10.1080/10454438.2017.1406417
Neves, C. A., Santos, E. A., & Bainy, A. C. D. (2000). Reduced superoxide dismutase activity in Palaemonetes argentinus (Decapoda, Palemonidae) infected by Probopyrus ringueleti (Isopoda, Bopyridae). Dis of Aquatic Organ, 39(2), 155–158. https://doi.org/10.3354/dao03915
Oliveira, A. S., Weinberg, Z. G., Ogunade, I. M., Cervantes, A. A., Arriola, K. G., Jiang, Y., Kim, D., Li, X., Gonçalves, M. C., Vyas, D., & Adesogan, A. T. (2017). Meta-analysis of effects of inoculation with homofermentative and facultative heterofermentative lactic acid bacteria on silage fermentation, aerobic stability, and the performance of dairy cows. Journal of Dairy Science, 100(6), 4587–4603. https://doi.org/10.3168/jds.2016-11815
Paez-Osuna, F., & Ruiz-Fernandez, C. (1995). Trace metals in the Mexican shrimp Penaeus vannamei from estuarine and marine environments. Environmental Pollution, 87(2), 243–247. https://doi.org/10.1016/0269-7491(94)P2612-D
Pato, M. L., & Duque, A. S. (2023). Traditional agri-food products and sustainability–A fruitful relationship for the development of rural areas in Portugal. Open Agriculture, 8(1), 20220157. https://doi.org/10.1515/opag-2022-0157
Pessione, E., Pessione, A., Lamberti, C., Coïsson, D. J., Riedel, K., Mazzoli, R., Bonetta, S., Eberl, L., & Giunta, C. (2009). First evidence of a membrane-bound, tyramine and β-phenylethylamine producing, tyrosine decarboxylase in Enterococcus faecalis: A two-dimensional electrophoresis proteomic study. Proteomics, 9(10), 2695–2710. https://doi.org/10.1002/pmic.200800780
Piper CS (2019) Soil and plant analysis. Scientific Publishers.
Praepanitchai, O. A., Noomhorm, A., & Anal, A. K. (2019). Survival and behavior of encapsulated probiotics (Lactobacillus plantarum) in calcium-alginate-soy protein isolate-based hydrogel beads in different processing conditions (pH and temperature) and in pasteurized mango juice. BioMed Research International, 2019, 9768152. https://doi.org/10.1155/2019/9768152
Putra, I., Rusliadi, R., Fauzi, M., Tang, U. M., & Muchlisin, Z. A. (2017). Growth performance and feed utilization of African catfish Clarias gariepinus fed a commercial diet and reared in the biofloc system enhanced with probiotic. F1000 Research, 6. https://doi.org/10.12688/f1000research.12438.1
Quiao-Won, M. E., & Teves, F. G. (2018). Characteristics of kombucha fermentation from different substrates and cytotoxicity of tea broth. Sustainable Food Production, 4(1), 11–19.
Ramos, O. Y., Basualdo, M., Libonatti, C., & Vega, M. F. (2020). Current status and application of lactic acid bacteria in animal production systems with a focus on bacteria from honey bee colonies. J App Microbiol, 128(5), 1248–1260. https://doi.org/10.1111/jam.14469
Reilly, C. (1980). Metal contamination of food (pp. 1–5). Applied Science Publishers. London.
Rengpipat, S., Rukpratanporn, S., Piyatiratitivorakul, S., & Menasaveta, P. (2000). Immunity enhancement in black tiger shrimp (Penaeus monodon) by a probiont bacterium (Bacillus S11). Aquacul, 191(4), 271–288. https://doi.org/10.1016/S0044-8486(00)00440-3
Sadat Hoseini Madani, N., Adorian, T. J., Ghafari Farsani, H., & Hoseinifar, S. H. (2018). The effects of dietary probiotic Bacilli (Bacillus subtilis and Bacillus licheniformis) on growth performance, feed efficiency, body composition and immune parameters of whiteleg shrimp (Litopenaeus vannamei) postlarvae. Aquacul Res, 49(5), 1926–1933. https://doi.org/10.1111/are.13648
Sahul Hameed, A. S., Abdul Majeed, S., Vimal, S., Madan, N., Rajkumar, T., Santhoshkumar, S., & Sivakumar, S. (2017). Studies on the occurrence of infectious myonecrosis virus in pond-reared Litopenaeus vannamei (Boone, 1931) in India. Journal of Fish Diseases, 40(12), 1823–1830. https://doi.org/10.1111/jfd.12655
Shao, J., Liu, M., Wang, B., Jiang, K., Wang, M., & Wang, L. (2017). Evaluation of biofloc meal as an ingredient in diets for white shrimp Litopenaeus vannamei under practical conditions: Effect on growth performance, digestive enzymes and TOR signaling pathway. Aquacul, 479, 516–521. https://doi.org/10.1016/j.aquaculture.2017.06.034
Shen, Y., Zhang, H., Cheng, L., Wang, L., Qian, H., & Qi, X. (2016). In vitro and in vivo antioxidant activity of polyphenols extracted from black highland barley. Food Chemistry, 194, 1003–1012. https://doi.org/10.1016/j.foodchem.2015.08.083
Smith, V. J., Brown, J. H., & Hauton, C. (2003). Immunostimulation in crustaceans: Does it really protect against infection? Fish & Shellfish Immunology, 15(1), 71–90. https://doi.org/10.1016/S1050-4648(02)00140-7
Song, Y. L., & Hsieh, Y. T. (1994). Immunostimulation of tiger shrimp (Penaeus monodon) hemocytes for generation of microbicidal substances: Analysis of reactive oxygen species. Developmental and Comparative Immunology, 18(3), 201–209. https://doi.org/10.1016/0145-305X(94)90012-4
Tan, L. V., Tran, T., & Loc, H. H. (2020). Soil and water quality indicators of diversified farming systems in a saline region of the Mekong Delta Vietnam. Agricul, 10(2), 38.
Thamizhvanan, S., Sivakumar, S., Santhosh Kumar, S., Vinoth Kumar, D., Suryakodi, S., Balaji, K., Rajkumar, T., Vimal, S., Abdul Majeed, S., Taju, G., & Sahul Hameed, A. S. (2019). Multiple infections caused by white spot syndrome virus and Enterocytozoon hepatopenaei in pond-reared Penaeus vannamei in India and multiplex PCR for their simultaneous detection. Journal of Fish Diseases, 42(3), 447–454. https://doi.org/10.1111/jfd.12956
Thompson, F. L., Iida, T., & Swings, J. (2004). Biodiversity of vibrios. Microbiology and Molecular Biology Reviews, 68(3), 403–431. https://doi.org/10.1128/MMBR.68.3.403-431.2004
Tsai, C. Y., Chi, C. C., & Liu, C. H. (2019). The growth and apparent digestibility of white shrimp, Litopenaeus vannamei, are increased with the probiotic Bacillus Subtilis. Aquacul Res, 50(5), 1475–1481. https://doi.org/10.1111/are.14022
Tseng, D. Y., Ho, P. L., Huang, S. Y., Cheng, S. C., Shiu, Y. L., Chiu, C. S., & Liu, C. H. (2009). Enhancement of immunity and disease resistance in the white shrimp, Litopenaeus vannamei, by the probiotic, Bacillus subtilis E20. Fish & Shellfish Immunology, 26(2), 339–344. https://doi.org/10.1016/j.fsi.2008.12.003
Vanbelle, M., Teller, E., & Focant, M. (1990). Probiotics in animal nutrition: A review. Arch Animal Nut, 40(7), 543–567. https://doi.org/10.1080/17450399009428406
Vijayan, K. K., Singh, I. B., Jayaprakash, N. S., Alavandi, S. V., Pai, S. S., Preetha, R., Rajan, J. J. S., & Santiago, T. C. (2006). A brackishwater isolate of Pseudomonas PS-102, a potential antagonistic bacterium against pathogenic vibrios in penaeid and non-penaeid rearing systems. Aquacul, 251, 192–200. https://doi.org/10.1016/j.aquaculture.2005.10.010
Vogt, G. (1993). Differentiation of B-cells in the hepatopancreas of the prawn Penaeus monodon. Acta Zoolog, 74(1), 51–60. https://doi.org/10.1111/j.1463-6395.1993.tb01220.x
Wanna, W., Aucharean, C., Kaitimonchai, P., & Jaengkhao, W. (2023). Effect of dietary Pediococcus pentosaceus MR001 on intestinal bacterial diversity and white spot syndrome virus protection in Pacific white shrimp. Aquac Rep, 30, 101570. https://doi.org/10.1016/j.aqrep.2023.101570
Wang, J., Ji, H., Wang, S., Liu, H., Zhang, W., Zhang, D., & Wang, Y. (2018). Probiotic Lactobacillus plantarum promotes intestinal barrier function by strengthening the epithelium and modulating gut microbiota. Fron Microbiol, 9, 1953. https://doi.org/10.3389/fmicb.2018.01953
Wang, Y. C., Hu, S. Y., Chiu, C. S., & Liu, C. H. (2019). Multiple-strain probiotics appear to be more effective in improving the growth performance and health status of white shrimp, Litopenaeus vannamei, than single probiotic strains. Fish & Shellfish Immunology, 84, 1050–1058. https://doi.org/10.1016/j.fsi.2018.11.017
Widanarni W, Yuhana M, Muhammad A (2014) Bacillus NP5 improves growth performance and resistance against infectious myonecrosis virus in white shrimp (Litopenaeus vannamei)(Bacillus NP5 Meningkatkan Pertumbuhan dan Ketahanan Terhadap Infeksi Virus Myonecrosis pada Udang Putih (L. vannamei). ILMU KELAUTAN: Indonesian J Marine Sci 19(4):211–218. . https://doi.org/10.14710/ik.ijms.19.4.211-218.
Xie, Z., Wang, F., Liu, H., Guo, S., Zhu, A., & Niu, H. (2010). Gelatin-walled microencapsulated diet for larval shrimp (Penaeus japonicus Bate) manufactured using the fluidized bed coating process. Aquacul Res, 42(1), 65–73. https://doi.org/10.1111/j.1365-2109.2010.02557.x
Yan, D. C., Liu, H. L., Sun, H. S., & Wang, Y. Y. (2014). Investigation of possible presence of infectious myonecrosis virus in shrimp in C hina. Journal of Fish Diseases, 37(7), 679–682. https://doi.org/10.1111/jfd.12151
Yu, Q., Fu, Z., Huang, M., Xu, C., Wang, X., Qin, J. G., Chen, L., Han, F., & Li, E. (2021). Growth, physiological, biochemical, and molecular responses of Pacific white shrimp Litopenaeus vannamei fed different levels of dietary selenium. Aquacul, 535, 736393. https://doi.org/10.1016/j.aquaculture.2021.736393
Zokaeifar, H., Balcázar, J. L., Saad, C. R., Kamarudin, M. S., Sijam, K., Arshad, A., & Nejat, N. (2012). Effects of Bacillus subtilis on the growth performance, digestive enzymes, immune gene expression and disease resistance of white shrimp Litopenaeus Vannamei. Fish Shellfish Immunol, 33(4), 683–689.
Funding
The authors acknowledge the grant support from UGC DR. D. S. Kothari Postdoctoral Fellowship (Normal Fellowship): No. F. 4–2/2006 (BSR)/BL/19–20/0298 New Delhi, The Management of Poseidon Biotech, Chennai and Manonmaniam Sundaranar University, Tirunelveli, Tamil Nadu, India for their constant support. The authors express their sincere appreciation to the Researchers Supporting Project Number (RSPD2023R696), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Rajkumar Thamarai: work design, writing review, editing, conceptualization, supervision. Thiyagarajan Sivaprakasam: data curation, supervision, project administration. Chinnaperumal Kamaraj: conceptualization, investigation, data curation, methodology, software, formal analysis, writing—review and editing. Dhatchayani Harikrishnan: formal analysis. Jawahar sri Prakash Thiyagarajan: documentation, formal analysis. Jeyashree Swaminathan: formal analysis. Koteeswaran Mani: formal analysis. Rajkumar Prabhakaran: data curation and analysis. Vimal Sukumar: formal analysis. Shine Kadaikunnan: critical review. Kalidass Subramaniam: critical review. All authors have read, understood, and have complied as applicable with the statement on “Ethical responsibilities of Authors” as found in the Instructions for Authors and are aware that with minor exceptions, no changes can be made to authorship once the paper is submitted.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent for publication
All authors gave final approval for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thamarai, R., Sivaprakasam, T., Kamaraj, C. et al. Impact of THIONIL® on reducing water and soil micro-pollutant and enhancement of growth, survival, anti-vibrio, immunity and health indices of Litopenaeus vannamei in pond trial. Environ Monit Assess 196, 93 (2024). https://doi.org/10.1007/s10661-023-12261-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-023-12261-y