Abstract
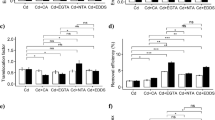
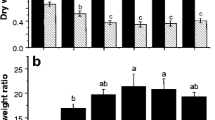
Chemically assisted phytoremediation is suggested as an effective approach to amplify the metal-remediating potential of hyperaccumulators. The current study assessed the efficiency of two biodegradable chelants (S,S-ethylenediamine disuccinic acid, EDDS; nitrilotriacetic acid, NTA) in enhancing the remediation of Cd by Coronopus didymus (Brassicaceae). C. didymus growing in Cd-contaminated soil (35–175 mg kg−1 soil) showed increased growth and biomass due to the hormesis effect, and chelant supplementation further increased growth, biomass, and Cd accumulation. A significant interaction with chelants and different Cd concentrations was observed, except for Cd content in roots and Cd content in leaves, which exhibited a non-significant interaction with chelant addition. The effect of the NTA amendment on the root dry biomass and shoot dry biomass was more pronounced than EDDS at all the Cd treatments. Upon addition of EDDS and NTA, bio-concentration factor values were enhanced by ~184–205 and ~ 199–208, respectively. The tolerance index of root and shoot increased over the control upon the addition of chelants, with NTA being better than EDDS. With chelant supplementation, bio-accumulation coefficient values were in the order Cd35 + NTA (~163%) > Cd105 + NTA (~137%) > Cd35 + EDDS (~89%) > Cd175 + NTA (~85%) > Cd105 + EDDS (~62%) > Cd175 + EDDS (~40%). The translocation factor correlated positively (r ≥ 0.8) with tolerance index and Cd accumulation in different plant parts. The study demonstrated that chelant supplementation enhanced Cd-remediation efficiency in C. didymus as depicted by improved plant growth and metal accumulation, and NTA was more effective than EDDS in reclaiming Cd.



Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Aghelan, N., Sobhanardakani, S., Cheraghi, M., Lorestani, B., & Merrikhpour, H. (2021). Evaluation of some chelating agents on phytoremediation efficiency of Amaranthus caudatus L. and Tagetes patula L. in soils contaminated with lead. Journal of Environmental Health Science and Engineering, 19, 503–514. https://doi.org/10.1007/s40201-021-00623-y
Bech, J., Poschenrieder, C., Llugany, M., Barceló, J., Tume, P., Tobias, F. J., Barranzuela, J. L., & Vásquez, E. R. (1997). Arsenic and heavy metal contamination of soil and vegetation around a copper mine in northern Peru. Science of the Total Environment, 203, 83–91. https://doi.org/10.1016/S0048-9697(97)00136-8
Bursztyn Fuentes, A. L., José, C., de Los Ríos, A., do Carmo, L. I., de Iorio, A. F., & Rendina, A. E. (2018). Phytoextraction of heavy metals from a multiply contaminated dredged sediment by chicory (Cichorium intybus L.) and castor bean (Ricinus communis L.) enhanced with EDTA, NTA, and citric acid application. International Journal of Phytoremediation, 20, 1354–1361. https://doi.org/10.1080/15226514.2018.1524826
Chellaiah, E. R. (2018). Cadmium (heavy metals) bioremediation by Pseudomonas aeruginosa: A minireview. Applied Water Science, 8, 154. https://doi.org/10.1007/s13201-018-0796-5
Chen, L., Wang, F., Zhang, Z., Chao, H., He, H., Hu, W., Zeng, Y., Duan, C., Liu, J., & Fang, L. (2023). Influences of arbuscular mycorrhizal fungi on crop growth and potentially toxic element accumulation in contaminated soils: A meta-analysis. Critical Reviews in Environmental Science and Technology, 53(20), 1795–1816. https://doi.org/10.1080/10643389.2023.2183700
Cherif, J., Derbel, N., Nakkach, M., von Bergmann, H., Jemal, F., & Lakhdar, Z. B. (2012). Spectroscopic studies of photosynthetic responses of tomato plants to the interaction of zinc and cadmium toxicity. Journal of Photochemistry and Photobiology B: Biology, 111, 9–16. https://doi.org/10.1016/j.jphotobiol.2012.03.002
Cui, S., Zhou, Q. X., Wei, S. H., Zhang, W., Cao, L., & Ren, L. P. (2007). Effects of exogenous chelators on phytoavailability and toxicity of Pb in Zinnia elegans Jacq. Journal of Hazardous Materials, 146, 341–346. https://doi.org/10.1016/j.jhazmat.2006.12.028
Diarra, I., Kotra, K. K., & Prasad, S. (2021). Assessment of biodegradable chelating agents in the phytoextraction of heavy metals from multi–metal contaminated soil. Chemosphere, 273, 128483. https://doi.org/10.1016/j.chemosphere.2020.128483
Gautam, M., & Agrawal, M. (2017). Phytoremediation of metals using vetiver (Chrysopogon zizanioides (L.) Roberty) grown under different levels of red mud in sludge amended soil. Journal of Geochemical Exploration, 182, 218–227. https://doi.org/10.1016/j.gexplo.2017.03.003
Gluhar, S., Kaurin, A., & Lestan, D. (2020). Soil washing with biodegradable chelating agents and EDTA: Technological feasibility, remediation efficiency and environmental sustainability. Chemosphere, 257, 127226. https://doi.org/10.1016/j.chemosphere.2020.127226
Goswami, S., & Das, S. (2015). A study on cadmium phytoremediation potential of Indian mustard, Brassica juncea. International Journal of Phytoremediation, 17, 583–588. https://doi.org/10.1080/15226514.2014.935289
Guo, D., Ali, A., Ren, C., Du, J., Li, R., Lahori, A. H., Xiao, R., Zhang, Z., & Zhang, Z. (2019). EDTA and organic acids assisted phytoextraction of Cd and Zn from a smelter contaminated soil by potherb mustard (Brassica juncea Coss) and evaluation of its bioindicators. Ecotoxicology and Environmental Safety, 167, 396–403. https://doi.org/10.1016/j.ecoenv.2018.10.038
Haider, F. U., Liqun, C., Coulter, J. A., Cheema, S. A., Wu, J., Zhang, R., Wenjun, M., & Farooq, M. (2021). Cadmium toxicity in plants: Impacts and remediation strategies. Ecotoxicology and Environmental Safety, 211, 111887. https://doi.org/10.1016/j.ecoenv.2020.111887
Hseu, Z. Y., Jien, S. H., Wang, S. H., & Deng, H. W. (2013). Using EDDS and NTA for enhanced phytoextraction of cd by water spinach. Journal of Environmental Management, 117, 58–64. https://doi.org/10.1016/j.jenvman.2012.12.028
Huang, F., Hu, J., Chen, L., Wang, Z., Sun, S., Zhang, W., Jiang, H., Luo, Y., Wang, L., Zeng, Y., & Fang, L. (2023). Microplastics may increase the environmental risks of Cd via promoting Cd uptake by plants: A meta-analysis. Journal of Hazardous Materials, 448, 130887. https://doi.org/10.1016/j.jhazmat.2023.130887
Jia, L., Liu, Z., Chen, W., Ye, Y., Yu, S., & He, X. (2015). Hormesis effects induced by cadmium on growth and photosynthetic performance in a hyperaccumulator, Lonicera japonica Thunb. Journal of Plant Growth Regulation, 34, 13–21. https://doi.org/10.1007/s00344-014-9433-1
Jiang, H., Zhao, X., Fang, J., & Xiao, Y. (2018). Physiological responses and metal uptake of Miscanthus under cadmium/arsenic stress. Environmental Science and Pollution Research, 25, 28275–28284. https://doi.org/10.1007/s11356-018-2835-z
Jordán, M. M., Montero, M. A., Pina, S., & García-Sánchez, E. (2009). Mineralogy and distribution of Cd, Ni, Cr, and Pb in biosolids-amended soils from Castellon Province (NE, Spain). Soil Science, 174, 14–20. https://doi.org/10.1097/SS.0b013e3181957492
Kabata-Pendias, A. (2000). Trace elements in soils and plants. CRC Press.
Kumar, A., Prasad, M. N. V., & Sytar, O. (2012). Lead toxicity, defense strategies and associated indicative biomarkers in Talinum triangulare grown hydroponically. Chemosphere, 89(9), 1056–1065. https://doi.org/10.1016/j.chemosphere.2012.05.070
Lamhamdi, M., Galiou, O. E., Bakrim, A., Nóvoa-Muñoz, J. C., Arias-Estevez, M., Aarab, A., & Lafont, R. (2013). Effect of lead stress on mineral content and growth of wheat (Triticum aestivum) and spinach (Spinacia oleracea) seedlings. Saudi Journal of Biological Sciences, 20, 29–36. https://doi.org/10.1016/j.sjbs.2012.09.001
Lan, J., Zhang, S., Lin, H., Li, T., Xu, X., Li, Y., Jia, Y., & Gong, G. (2013). Efficiency of biodegradable EDDS, NTA and APAM on enhancing the phytoextraction of cadmium by Siegesbeckia orientalis L. grown in cd-contaminated soils. Chemosphere, 91, 1362–1367. https://doi.org/10.1016/j.chemosphere.2013.01.116
Liu, B., Wang, J., Xu, M., Zhao, L., & Wang, Z. (2019). Spatial distribution, source apportionment and ecological risk assessment of heavy metals in the sediments of Haizhou Bay national ocean park, China. Marine Pollution Bulletin, 149, 110651. https://doi.org/10.1016/j.marpolbul.2019.110651
Liu, C., Xiao, R., Dai, W., Huang, F., & Yang, X. (2021). Cadmium accumulation and physiological response of Amaranthus tricolor L. under soil and atmospheric stresses. Environmental Science and Pollution Research, 28, 14041–14053. https://doi.org/10.1007/s11356-020-11569-3
Liu, Z., Chen, W., & He, X. (2018). Evaluation of hyperaccumulation potentials to cadmium (Cd) in six ornamental species (Compositae). International Journal of Phytoremediation, 20, 1464–1469. https://doi.org/10.1080/15226514.2018.1501343
Lu, L. L., Tian, S. K., Yang, X. E., Peng, H. Y., & Li, T. Q. (2013). Improved cadmium uptake and accumulation in the hyperaccumulator Sedum alfredii: The impact of citric acid and tartaric acid. Journal of Zhejiang University Science B, 14, 106–114. https://doi.org/10.1631/jzus.B1200211
Mahmud, J. A., Hasanuzzaman, M., Nahar, K., Bhuyan, M. B., & Fujita, M. (2018). Insights into citric acid-induced cadmium tolerance and phytoremediation in Brassica juncea L.: Coordinated functions of metal chelation, antioxidant defense and glyoxalase systems. Ecotoxicology and Environmental Safety, 147, 990–1001. https://doi.org/10.1016/j.ecoenv.2017.09.045
Monteiro, M. S., Santos, C., Soares, A. M. V. M., & Mann, R. M. (2009). Assessment of biomarkers of cadmium stress in lettuce. Ecotoxicology and Environmental Safety, 72(3), 811–818. https://doi.org/10.1016/j.ecoenv.2008.08.002
Moral, R., Gilkes, R. J., & Jordán, M. M. (2005). Distribution of heavy metals in calcareous and non-calcareous soils in Spain. Water, Air, and Soil Pollution, 162, 127–142. https://doi.org/10.1007/s11270-005-5997-5
Nowack, B., Schulin, R., & Robinson, B. H. (2006). Critical assessment of chelant-enhanced metal phytoextraction. Environmental Science & Technology, 40, 5225–5232. https://doi.org/10.1021/es0604919
Paunov, M., Koleva, L., Vassilev, A., Vangronsveld, J., & Goltsev, V. (2018). Effects of different metals on photosynthesis: Cadmium and zinc affect chlorophyll fluorescence in durum wheat. International Journal of Molecular Sciences, 19, 787. https://doi.org/10.3390/ijms19030787
Quartacci, M. F., Irtelli, B., Baker, A. J., & Navari-Izzo, F. (2007). The use of NTA and EDDS for enhanced phytoextraction of metals from a multiply contaminated soil by Brassica carinata. Chemosphere, 68(10), 1920–1928. https://doi.org/10.1016/j.chemosphere.2007.02.058
Raina, R., Sharma, P., Batish, D. R., & Singh, H. P. (2023). Assessment of natural low molecular weight organic acids in facilitating cadmium phytoextraction by Lepidium didymus (Brassicaceae). Environmental Science and Pollution Research, Accepted, In Press. https://doi.org/10.1007/s11356-023-27719-2
Rani, D., & Kohli, R. K. (1991). Fresh matter is not an appropriate relation unit for chlorophyll content: Experience from experiments on effects of herbicide and allelopathic substance. Photosynthetica, 25, 655–658.
Rascio, N., & Navari-Izzo, F. (2011). Heavy metal hyperaccumulating plants: How and why do they do it? And what makes them so interesting? Plant Science, 180, 169–181. https://doi.org/10.1016/j.plantsci.2010.08.016
Ren, M., Qin, Z., Li, X., Wang, L., Wang, Y., Zhang, J., Huang, Y., & Yang, S. (2020). Selenite antagonizes the phytotoxicity of Cd in the cattail Typha angustifolia. Ecotoxicology and Environmental Safety, 189, 109959. https://doi.org/10.1016/j.ecoenv.2019.109959
Sampanpanish, P., & Nanthavong, K. (2019). Effect of EDTA and NTA on arsenic bioaccumulation and translocation using phytoremediation by Mimosa pudica L. from contaminated soils. Bulletin of Environmental Contamination and Toxicology, 102, 140–145. https://doi.org/10.1007/s00128-018-2502-6
Shakoor, M. B., Ali, S., Hameed, A., Farid, M., Hussain, S., Yasmeen, T., Najeeb, U., Bharwana, S. A., & Abbasi, G. H. (2014). Citric acid improves lead (Pb) phytoextraction in Brassica napus L. by mitigating Pb-induced morphological and biochemical damages. Ecotoxicology and Environmental Safety, 109, 38–47. https://doi.org/10.1016/j.ecoenv.2014.07.033
Sharma, P., Ahmad, M., Rathee, S., Singh, H. P., Batish, D. R., & Kohli, R. K. (2021). Bridging the gap: Linking morpho-functional traits’ plasticity with hyperaccumulator. Environmental Monitoring and Assessment, 193, 762. https://doi.org/10.1007/s10661-021-09504-1
Sharma, P., Rathee, S., Ahmad, M., Raina, R., Batish, D. R., & Singh, H. P. (2023). Comparison of synthetic and organic biodegradable chelants in augmenting cadmium phytoextraction in Solanum nigrum. International Journal of Phytoremediation, 25, 1106–1115. https://doi.org/10.1080/15226514.2022.2133081
Sidhu, G. P. S., Singh, H. P., Batish, D. R., & Kohli, R. K. (2017a). Appraising the role of environment friendly chelants in alleviating lead by Coronopus didymus from Pb-contaminated soils. Chemosphere, 182, 129–136. https://doi.org/10.1016/j.chemosphere.2017.05.026
Sidhu, G. P. S., Singh, H. P., Batish, D. R., & Kohli, R. K. (2017b). Tolerance and hyperaccumulation of cadmium by a wild, unpalatable herb Coronopus didymus (L.) Sm. (Brassicaceae). Ecotoxicology and Environmental Safety, 135, 209–215. https://doi.org/10.1016/j.ecoenv.2016.10.001
Tananonchai, A., Sampanpanish, P., Chanpiwat, P., & Tancharakorn, S. (2019). Effect of EDTA and NTA on cadmium distribution and translocation in Pennisetum purpureum Schum cv. Mott. Environmental Science and Pollution Research, 26, 9851–9860. https://doi.org/10.1007/s11356-018-04103-z
Wang, K., Liu, Y., Song, Z., Wang, D., & Qiu, W. (2019). Chelator complexes enhanced Amaranthus hypochondriacus L. phytoremediation efficiency in Cd-contaminated soils. Chemosphere, 237, 124480. https://doi.org/10.1016/j.chemosphere.2019.124480
Wang, S., Shi, X., Sun, H., Chen, Y., Pan, H., Yang, X., & Rafiq, T. (2014). Variations in metal tolerance and accumulation in three hydroponically cultivated varieties of Salix integra treated with lead. PLoS One, 9, e108568. https://doi.org/10.1371/journal.pone.0108568
Wang, Y., Xu, Y., Qin, X., Zhao, L., Huang, Q., & Liang, X. (2021). Effects of S,S-ethylenediamine disuccinic acid on the phytoextraction efficiency of Solanum nigrum L. and soil quality in Cd-contaminated alkaline wheat soil. Environmental Science and Pollution and Research, 28, 2959–42974. https://doi.org/10.1007/s11356-021-13764-2
Wen, H., Zhang, Y., Cloquet, C., Zhu, C., Fan, H., & Luo, C. (2015). Tracing sources of pollution in soils from the Jinding Pb–Zn mining district in China using cadmium and lead isotopes. Applied Geochemistry, 52, 147–154. https://doi.org/10.1016/j.apgeochem.2014.11.025
Yan, A., Wang, Y., Tan, S. N., Mohd Yusof, M. L., Ghosh, S., & Chen, Z. (2020). Phytoremediation: A promising approach for revegetation of heavy metal-polluted land. Frontiers in Plant Science, 11, 359. https://doi.org/10.3389/fpls.2020.00359
Yang, W., Dai, H. P., Skuza, L., & Wei, S. H. (2020). The front-heavy and back-light nitrogen application mode to increase stem and leaf biomass significantly improved cadmium accumulation in Solanum nigrum L. Journal of Hazardous Materials, 393, 122482–122489. https://doi.org/10.1016/j.jhazmat.2020.122482
Yang, X., Feng, Y., He, Z., & Stoffella, P. J. (2005). Molecular mechanisms of heavy metal hyperaccumulation and phytoremediation. Journal of Trace Elements in Medicine and Biology, 18, 339–353. https://doi.org/10.1111/j.1744-7909.2005.00144.x
Zacchini, M., Pietrini, F., Mugnozza, G. S., Iori, V., Pietrosanti, L., & Massacci, A. (2009). Metal tolerance, accumulation and translocation in poplar and willow clones treated with cadmium in hydroponics. Water, Air, and Soil Pollution, 197, 23–34. https://doi.org/10.1007/s11270-008-9788-7
Zhang, S., Chen, M., Li, T., Xu, X., & Deng, L. (2010). A newly found cadmium accumulator—Malva sinensis Cavan. Journal of Hazardous Materials, 173, 705–709. https://doi.org/10.1016/j.jhazmat.2009.08.142
Zhang, Y., Wang, X., & Ji, H. (2020). Co-remediation of Pb contaminated soils by heat modified sawdust and Festuca arundinacea. Scientific Reports, 10, 4663. https://doi.org/10.1038/s41598-020-61668-x
Zhao, T., Tan, L., Huang, W., & Wang, J. (2019). The interactions between micro polyvinyl chloride (mPVC) and marine dinoflagellate Karenia mikimotoi: The inhibition of growth, chlorophyll and photosynthetic efficiency. Environmental Pollution, 247, 883–889. https://doi.org/10.1016/j.envpol.2019.01.114
Funding
No funds were received for this study.
Author information
Authors and Affiliations
Contributions
Harminder Pal Singh: Project leader, experimental layout, manuscript editing; Daizy Rani Batish: Co-Project leader, manuscript editing; Ravinder Kumar Kohli: experimental design, manuscript editing; Riya Raina: Establishment and execution of experiment, data collection and analysis, manuscript preparation; Padma Sharma: Data analysis and manuscript preparation.
Corresponding author
Ethics declarations
Ethical responsibilities of authors
All authors have read, understood, and have complied as applicable with the statement on “Ethical responsibilities of Authors” as found in the Instructions for Authors and are aware that with minor exceptions, no changes can be made to authorship once the paper is submitted.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 6693 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Raina, R., Sharma, P., Batish, D.R. et al. Comparative assessment of two biodegradable chelants, S,S-ethylenediamine disuccinic acid and nitrilotriacetic acid, in facilitating Cd remediation by lesser swine cress (Coronopus didymus, Brassicaceae). Environ Monit Assess 195, 1526 (2023). https://doi.org/10.1007/s10661-023-12073-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-023-12073-0