Abstract
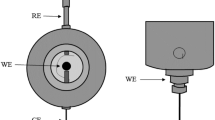
A comprehensive comparative validation for two different types of dissolved oxygen (DO) analyzers, amperometric and optical, is presented on two representative commercial DO analyzers. A number of performance characteristics were evaluated including drift, intermediate precision, accuracy of temperature compensation, accuracy of reading (under different measurement conditions), linearity, flow dependence of the reading, repeatability (reading stability), and matrix effects of dissolved salts. The matrix effects on readings in real samples were evaluated by analyzing the dependence of the reading on salt concentration (at saturation concentration of DO). The analyzers were also assessed in DO measurements of a number of natural waters. The uncertainty contributions of the main influencing parameters were estimated under different experimental conditions. It was found that the uncertainties of results for both analyzers are quite similar but the contributions of the uncertainty sources are different. Our results imply that the optical analyzer might not be as robust as is commonly assumed; however, it has better reading stability, lower stirring speed dependence, and typically requires less maintenance. On the other hand, the amperometric analyzer has a faster response and wider linear range. Both analyzers seem to have issues with the accuracy of temperature compensation. The approach described in this work will be useful to practitioners carrying out DO measurements for ensuring reliability of their measurements.






Similar content being viewed by others
References
Benson, B. B., & Krause, D. (1980). The concentration and isotopic fractionation of gases dissolved in freshwater in equilibrium with the atmosphere. 1. Oxygen: Oxygen in freshwater. Limnology and Oceanography, 25, 662–671. https://doi.org/10.4319/lo.1980.25.4.0662.
Benson, B. B., & Krause, D. (1984). The concentration and isotopic fractionation of oxygen dissolved in freshwater and seawater in equilibrium with the atmosphere. Limnology and Oceanography, 29, 620–632. https://doi.org/10.4319/lo.1984.29.3.0620.
Clesceri, L. S., & American Public Health Association, American Water Works Association, and Water Pollution Control Federation (Eds.). (1998). Standard methods: for the examination of water and wastewater (20th ed.). Washington: American Public Health Ass.
Diaz, R. J., & Rosenberg, R. (2008). Spreading dead zones and consequences for marine ecosystems. Science, 321, 926–929. https://doi.org/10.1126/science.1156401.
Garcia, H. E., Locarnini, R. A., Boyer, T. P., Antonov, J. I., Baranova, O. K., Zweng, M. M., et al. (2010). World ocean atlas 2009 Volume 3: Dissolved Oxygen, Apparent Oxygen Utilization, and Oxygen Saturation. Sydney Levitus, Ed. Washington, D.C.: NOAA Atlas NESDIS 70, U.S. Government Printing Office Available at: http://www.nodc.noaa.gov/OC5/indprod.html.
Gewehr, P. M., & Delpy, D. T. (1993). Optical oxygen sensor based on phosphorescence lifetime quenching and employing a polymer immobilised metalloporphyrin probe. Medical & Biological Engineering & Computing, 31, 2–10. https://doi.org/10.1007/BF02446879.
Gillanders, R. N., Tedford, M. C., Crilly, P. J., & Bailey, R. T. (2005). A composite thin film optical sensor for dissolved oxygen in contaminated aqueous environments. Analytica Chimica Acta, 545, 189–194. https://doi.org/10.1016/j.aca.2005.04.086.
HACH Company. (2006). HQ series portable meters, USER MANUAL, edition 5.
Häck, M. (2006). Optical measurement of oxygen concentration in water. Available at: https://cz.hach.com/cms/documents/parameter-4-downloads-1.pdf. Accessed December 7, 2017.
Hellat, K., Mashirin, A., Nei, L., & Tenno, T. (1986). Metrological garanteeing of devices for measuring oxygen in the water. Acta et Commentationes Universitatis Tartuensis de Mathematica, 757, 184–193.
Helm, I., Jalukse, L., & Leito, I. (2010). Measurement uncertainty estimation in Amperometric sensors: a tutorial review. Sensors, 10, 4430–4455. https://doi.org/10.3390/s100504430.
Helm, I., Jalukse, L., & Leito, I. (2012). A highly accurate method for determination of dissolved oxygen: gravimetric Winkler method. Analytica Chimica Acta, 741, 21–31. https://doi.org/10.1016/j.aca.2012.06.049.
Hitchman, M. L. (1978). Measurement of dissolved oxygen. Canada: Wiley Limited Available at: https://books.google.ee/books?id=1qUPAQAAMAAJ.
ISO 17289:2014. (2014). Water quality—determination of dissolved oxygen—Optical sensor method.
ISO 5814:2012. (2012). Water quality. Determination of dissolved oxygen. Electrochemical probe method.
ISO/IEC 17043:2010. (2010). Conformity assessment—general requirements for proficiency testing (This standard replaces the ISO Guides 43–1 and 43–2).
Jalukse, L., & Leito, I. (2007). Model-based measurement uncertainty estimation in amperometric dissolved oxygen concentration measurement. Measurement Science and Technology, 18, 1877–1886.
JCGM 100:2008. (2008). Evaluation of measurement data—guide to the expression of uncertainty in measurement. Available at: https://www.bipm.org/utils/common/documents/jcgm/JCGM_100_2008_E.pdf.
Jessen, G. L., Lichtschlag, A., Ramette, A., Pantoja, S., Rossel, P. E., Schubert, C. J., Struck, U., & Boetius, A. (2017). Hypoxia causes preservation of labile organic matter and changes seafloor microbial community composition (Black Sea). Science Advances, 3, e1601897. https://doi.org/10.1126/sciadv.1601897.
Kragten, J. (1994). Tutorial review. Calculating standard deviations and confidence intervals with a universally applicable spreadsheet technique. The Analyst, 119, 2161. https://doi.org/10.1039/an9941902161.
Lakowicz, J. R. (Ed.). (2006). Principles of fluorescence spectroscopy (3rd ed.). Boston: Springer US. https://doi.org/10.1007/978-0-387-46312-4.
Langie, B., & Lewandow, M. (1999). Comparison between polarographic and galvanic dissolved oxygen sensor technologies. Available at: http://www.4oakton.com/TechTips/OAK_TT25.pdf. Accessed May 5, 2017.
Liu, H., Gu, Y., Kim, J. G., & Mason, R. P. (2004). Near-infrared spectroscopy and imaging of tumor vascular oxygenation. In Methods in Enzymology (pp. 349–378). Elsevier. https://doi.org/10.1016/S0076-6879(04)86017-8.
Magnusson, B., Näykky, T., Hovind, H., & Krysell, M. (2012). Handbook for calculation of measurement uncertainty in environmental laboratories (NT TR 537 - Edition 3.1). Available at: http://www.nordtest.info/index.php/technical-reports/item/handbook-for-calculation-of-measurement-uncertainty-in-environmental-laboratories-nt-tr-537-edition-3.html.
Mancy, K. H., & Jaffe, T. (1966). Analysis of dissolved oxygen in natural and waste waters. Cincinnati: U.S. Dept. of Health, Education, and Welfare, Robert A. Taft Sanitary Engineering Center.
McDonagh, C., Kolle, C., McEvoy, A. K., Dowling, D. L., Cafolla, A. A., Cullen, S. J., & MacCraith, B. D. (2001). Phase fluorometric dissolved oxygen sensor. Sensors and Actuators B: Chemical, 74, 124–130. https://doi.org/10.1016/S0925-4005(00)00721-8.
McDonagh, C., Burke, C. S., & MacCraith, B. D. (2008). Optical chemical sensors. Chemical Reviews, 108, 400–422. https://doi.org/10.1021/cr068102g.
McKeown, J. J., Brown, L. C., & Gove, G. W. (1967). Comparative studies of dissolved oxygen analysis methods. Journal - Water Pollution Control Federation, 39, 1323–1336.
Millero, F. J., Huang, F., & Graham, T. B. (2003). Solubility of oxygen in some 1-1, 2-1, 1-2, and 2-2 electrolytes as a function of concentration at 25°C. Journal of Solution Chemistry, 32, 473–487. https://doi.org/10.1023/A:1025301314462.
Mortimer, C. H. (1981). The oxygen content of air-saturated fresh waters over ranges of temperature and atmospheric pressure of limnological interest. Stuttgart: Schweizerbart Science Publishers Available at: http://www.schweizerbart.de//publications/detail/isbn/9783510520220/Mitteilungen_IVL_Nr_22.
Narayanaswamy, R., & Wolfbeis, O. S. (2004). Optical sensors. Berlin: Springer. https://doi.org/10.1007/978-3-662-09111-1.
Näykki, T., Jalukse, L., Helm, I., & Leito, I. (2013). Dissolved oxygen concentration interlaboratory comparison: what can we learn? Water, 5, 420–442. https://doi.org/10.3390/w5020420.
Pagano, T., Carcamo, N., & Kenny, J. E. (2014). Investigation of the fluorescence quenching of 1-aminoanthracene by dissolved oxygen in cyclohexane. The Journal of Physical Chemistry. A, 118, 11512–11520. https://doi.org/10.1021/jp5094806.
Ramamoorthy, R., Dutta, P. K., & Akbar, S. A. (2003). Oxygen sensors: materials, methods, designs and applications. Journal of Materials Science, 38, 4271–4282. https://doi.org/10.1023/A:1026370729205.
Schmidtko, S., Stramma, L., & Visbeck, M. (2017). Decline in global oceanic oxygen content during the past five decades. Nature, 542, 335–339.
Shaw, A. D., Li, Z., Thomas, Z., & Stevens, C. W. (2002). Assessment of tissue oxygen tension: comparison of dynamic fluorescence quenching and polarographic electrode technique. Critical Care, 6, 76–80.
Stetter, J. R., & Li, J. (2008). Amperometric gas sensors—a review. Chemical Reviews, 108, 352–366. https://doi.org/10.1021/cr0681039.
Stramma, L., Prince, E. D., Schmidtko, S., Luo, J., Hoolihan, J. P., Visbeck, M., Wallace, D. W. R., Brandt, P., & Körtzinger, A. (2011). Expansion of oxygen minimum zones may reduce available habitat for tropical pelagic fishes. Nature Climate Change, 2, 33–37.
Tengberg, A., Hovdenes, J., Andersson, H. J., Brocandel, O., Diaz, R., Hebert, D., Arnerich, T., Huber, C., Körtzinger, A., Khripounoff, A., Rey, F., Rönning, C., Schimanski, J., Sommer, S., & Stangelmayer, A. (2006). Evaluation of a lifetime-based optode to measure oxygen in aquatic systems: lifetime-based optode to measure oxygen. Limnology and Oceanography: Methods, 4, 7–17. https://doi.org/10.4319/lom.2006.4.7.
Wang, X., & Wolfbeis, O. S. (2014). Optical methods for sensing and imaging oxygen: materials, spectroscopies and applications. Chemical Society Reviews, 43, 3666–3761. https://doi.org/10.1039/C4CS00039K.
Weilheim, WTW GmbH & Co. (2002). KG Operating Manual, CellOx 325 Dissolved oxygen sensor. Available at: http://www.globalw.com/downloads/WQ/cellox325.pdf.
Xiao, D., Mo, Y., & Choi, M. M. F. (2003). A hand-held optical sensor for dissolved oxygen measurement. Measurement Science and Technology, 14, 862–867.
YSI. (n.d.). The dissolved oxygen handbook: a practical guide to dissolved oxygen measurements. Available at: http://www.vanwalt.com/pdf/information-sheets/Dissolved-Oxygen-Handbook.pdf. Accessed May 6, 2017.
Funding
This research was supported by the EU through the European Regional Development Fund (TK141 “Advanced materials and high-technology devices for energy recuperation systems”) and by the Ministry of Education and Science of Estonia (institutional research grant no. IUT20-14).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Characteristics of the sensors and uncertainty calculations for the DO concentration data in different waters are available in the Electronic supplementary material.
ESM 1
(XLSX 183 KB)
Rights and permissions
About this article
Cite this article
Helm, I., Karina, G., Jalukse, L. et al. Comparative validation of amperometric and optical analyzers of dissolved oxygen: a case study. Environ Monit Assess 190, 313 (2018). https://doi.org/10.1007/s10661-018-6692-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-018-6692-5