Abstract
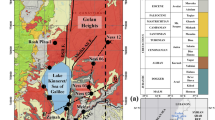
Groundwater flow and its geochemical evolution in mines are important not only in the study of contaminant migration but also in the effective planning of excavation. The effects of groundwater on the stability of rock slopes and other mine constructions especially in limestone quarries are crucial because calcite, the major mineral component of limestone, is moderately soluble in water. In this study, evolution of groundwater in a limestone quarry located in Chichibu city was monitored to understand the geochemical processes occurring within the rock strata of the quarry and changes in the chemistry of groundwater, which suggests zones of deformations that may affect the stability of rock slopes. There are three distinct geological formations in the quarry: limestone layer, interbedded layer of limestone and slaty greenstone, and slaty greenstone layer as basement rock. Although the hydrochemical facies of all groundwater samples were Ca-HCO3 type water, changes in the geochemical properties of groundwater from the three geological formations were observed. In particular, significant changes in the chemical properties of several groundwater samples along the interbedded layer were observed, which could be attributed to the mixing of groundwater from the limestone and slaty greenstone layers. On the rainy day, the concentrations of Ca2+ and HCO3− in the groundwater fluctuated notably, and the groundwater flowing along the interbedded layer was dominated by groundwater from the limestone layer. These suggest that groundwater along the interbedded layer may affect the stability of rock slopes.











Similar content being viewed by others
References
Acton, D. W., & Barker, J. F. (1992). In situ biodegradation potential of aromatic hydrocarbons in anaerobic groundwaters. Journal of Contaminant Hydrology, 9, 325–352.
Alberic, P., & Lepiller, M. (1998). Oxydation de la matière organique dans un système hydrologique karstique alimenté par des pertes fluviales (Loiret, France). Water Research, 32, 2051–2064.
American Society for Testing and Materials. (1997). Standards relating to environmental site characterization. Philadelphia: American Society for Testing and Materials.
Appelo, C. A. J., & Postma, D. (2005). Geochemistry, groundwater and pollution (2nd ed.). London: A.A. Balkema Publishers.
Batoit, C., Emblanch, C., Blavoux, B., Simler, R., & Daniel, M. (2000). Organic matter in karstic aquifers: a potential tracer in the carbon cycle. A small-scale laboratory model approach. IAHS Publication, 262, 459–463.
Benefield, L. D., & Morgan, J. S. (1990). Chemical precipitation. In F. W. Pontius (Ed.), Water quality and treatment. McGraw Hill, New York: The American Water Works Association Chapter 10.
Committee on the long-wall rock slopes. (2013). Research on the stability of the long-wall rock slopes of limestone quarries in Japan. Mining and Materials Processing Institute of Japan.
Deutsch, W. J., & Siegel, R. (1997). Groundwater geochemistry: fundamentals and applications to contamination. Boca Raton: CRC Press.
Dong, J. J., Tu, C. H., Lee, W. R., & Jheng, Y. J. (2012). Effects of hydraulic conductivity/strength anisotropy on the stability of stratified, poorly cemented rock slopes. Computers and Geotechnics, 40, 147–159.
Fitts, C. R. (2002). Groundwater science. San Diego: Academic Press (Elsevier Science).
Freeze, R. A., & Cherry, J. A. (1979). Groundwater. Englewood Cliffs, New Jersey: Prentice Hall.
Fujinuki, T. (1983). Chemical composition of limestones in Japan. In S. Kawada (Ed.), Limestones in Japan (pp. 43–73). Tokyo: Limestone Mining Association.
Hanson, P. J., Edwards, N. T., Garten, C. T., & Andrews, J. A. (2000). Separating root and soil microbial contributions to soil respiration: a review of methods and observations. Biogeochemistry, 48, 115–146.
Hayashi, S., Iijima, S., Ishii, I., Nakajima, T., Sawaguchi, H., Tanaka, H., & Yoshida, T. (1990). Late Paleozoic to Mesozoic formations in the southwestern Ashio Mountains. Bulletin of the Gunma Prefectural Museum of History, 11, 1–34.
Kodama, J., Kaneko, K., Tsuzuki, M., & Nishiyama, E. (2004). Measurement and analysis of long-term deformation of rock slope on Ikura limestone quarry. Journal of the Mining and Materials Processing Institute of Japan, 120, 182–189.
Kondo, M., Nakatani, K., Mikami, K. (2016). Stabilization works for the final slope and procedures for validation of their effects in the Une limestone quarry (pp. 1112). Morioka: Proceedings of the Mining and Materials Processing Institute of Japan.
Kuo, C. S. (2012). The mineral industry of Japan. U.S. Geological Survey Minerals Yearbook–2010, 12.1−12.13.
Laursen, S. (1991). On gaseous diffusion of CO2 in the unsaturated zone. Journal of Hydrology, 122, 61–69.
Macpherson, G. L., Roberts, J. A., Blair, J. M., Townsend, M. A., Fowle, D. A., & Beisner, K. R. (2008). Increasing shallow groundwater CO2 and limestone weathering, Konza Prairie, USA. Geochimica et Cosmochimica Acta, 72, 5581–5599.
Matsuda, H., Adachi, H., Nishimura, Y., & Shimizu, N. (2002). Applicability of the trend model for smoothing measured displacements by using global positioning system and methods for predicting displacement behavior. Journal of Japan Society of Civil Engineers, 715, 333–343.
Merkel, B. J., & Planer-Friedrich, B. (2008). In D. K. Nordstrom (Ed.), Groundwater geochemistry: a practical guide to modeling of natural and contaminated aquatic systems (2nd ed.). Boulder: U.S. Geological Survey 80303.
Naja, G. M., Rivero, R., Davis III, S. E., & Lent, T. V. (2011). Hydrochemical impacts of limestone rock mining. Water, Air, & Soil Pollution, 217, 95–104.
Ozawa, K., Aoyama, H., Kondo, M., Nakatani, K. (2016). Analysis on the final slope behaviors by rainfall and consideration on the effects of stabilizing measures (pp. 1113). Morioka: Proceedings of the Mining and Materials Processing Institute of Japan.
Paces, T. (1975). Kinetics of natural water systems. Vienna: I.A.E.A.
Parkhurst, D. L., Appelo, C. A. J. (2013). Description of input and examples for PHREEQC (Version 3) – a computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations. U.S. Geological Survey. Techniques and Methods, Book 6, Chap. A43, 497.
Peyraube, N., Lastennet, R., Denis, A., Malaurent, P., & Villanueva, J. D. (2014). Interpreting CO2-SI c relationship to estimate CO2 baseline in limestone aquifers. Environmental Earth Sciences, 72, 4207–4215.
Plummer, L. N., Wigley, M. L., Parkhurst, D. L. (1978). The kinetics of calcite dissolution in CO2-water systems at 5°C to 60°C and 0.0 to 1.0 ATM CO2. American Journal of Science, 278, 179−216.
Reilly, T. E., Plummer, L. N., Phillips, P. J., & Busenberg, E. (1994). The use of simulation and multiple environmental tracers to quantify groundwater flow in a shallow aquifer. Water Resources Research, 30(2), 421–433.
Sjöberg, J. (1999). Analysis of large scale rock slopes (Doctoral Thesis of Lulea University of Technology). Retrieved from http://geotechpedia.com/Publication/Show/3769/ANALYSIS-OF-LARGE-SCALE-ROCK-SLOPES. Accessed 20 Aug 2016.
Szabo, Z., Rice, D. E., Plummer, L. N., Busenberg, E., Drenkard, S., & Schlosser, P. (1996). Age-dating of shallow groundwater with chlorofluorocarbons, tritium/helium-3, and flow path analysis, southern New Jersey coastal plain. Water Resources Research, 32, 1023–1038.
Tabelin, C. B., Igarashi, T., & Takahashi, R. (2012). The roles of pyrite and calcite in the mobilization of arsenic and lead from hydrothermally altered rocks excavated in Hokkaido, Japan. Journal of Geochemical Exploration, 119, 17–31.
Tabelin, C. B., Hashimoto, A., Igarashi, T., & Yoneda, T. (2014). Leaching of boron, arsenic and selenium from sedimentary rocks: I. Effects of contact time, mixing speed and liquid-to-solid ratio. Science of the Total Environment, 472, 620–629.
Todd, D. K., & Mays, L. W. (2005). Groundwater hydrology (3rd ed.). New York: John Wiley and Sons, Inc..
Ulusay, R., Ekmekci, M., Tuncay, E., & Hasancebi, N. (2014). Improvement of slope stability based on integrated geotechnical evaluations and hydrogeological conceptualisation at a lignite open pit. Engineering Geology, 181, 261–280.
Yamaguchi, U., & Shimotani, T. (1986). A case study of slope failure in a limestone quarry. International Journal of Rock Mechanics and Mining Science & Geomechanics Abstracts, 23(1), 95–104.
Yoshida, H., & Nozaki, I. (2008). Study on the stability of retaining wall during rainfall. Structural Engineering, Japan Society of Civil Engineers, 54A, 102–113.
Yoshida, H., Kinoshita, N., & Ohno, M. (2008). Investigation of collapse of retaining wall and inspection of its stability. Journal of Japan Society of Civil Engineers, 64(1), 57–66.
Záruba, Q., & Mencl, V. (1976). Engineering geology: developments in geotechnical engineering (Vol. 10). New York: American Elsevier Publishing Company, Inc..
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 263 kb)
Rights and permissions
About this article
Cite this article
Eang, K.E., Igarashi, T., Fujinaga, R. et al. Groundwater monitoring of an open-pit limestone quarry: groundwater characteristics, evolution and their connections to rock slopes. Environ Monit Assess 190, 193 (2018). https://doi.org/10.1007/s10661-018-6561-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-018-6561-2