Abstract
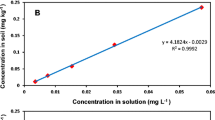
Tralkoxydim is a cyclohexanedione herbicide primarily used for gramineous weed control in China. In this paper, we present results of a tralkoxydim laboratory environmental fate study characterizing its degradation, adsorption, and mobility behavior in three different soils and two water-sediment systems (river and lake) in China. Degradation half-life of tralkoxydim in soil under aerobic conditions was 5.1, 7.7, and 7.9 days in Jiangxi red soil, Taihu paddy soil, and Northeast China black soil, respectively. Under anaerobic and flooding conditions, half-life values were 6.2, 15.1, and 19.8 days for the same three soils, respectively. Soil pH was the major factor effecting tralkoxydim degradation. In the aerobic water-sediment experiments, tralkoxydim degraded faster in the river system (total system half-life 43.3 days) than the lake system (total system half-life 99.0 days). Correspondingly, its anaerobic degradation half-life values were 46.2 and 53.3 days for the river and lake systems, respectively. Tralkoxydim adsorption in the three soils was found to follow the empirical Freundlich isotherm. The adsorption coefficient (K d ) was 8.60, 1.00, and 1.57 for Jiangxi red soil, Taihu paddy soil, and Northeast China black soil, respectively. Soil pH was the major factor effecting tralkoxydim adsorption. Adsorption free energy change was less than 40 kJ mol−1 in all three soils, indicating a physical mechanism in the process. Thin-layer chromatography (TLC) tests showed that relative to the solvent transport to 11.5 cm, the travel distance of tralkoxydim was 8–10 cm in the three soils, corresponding Rf values at 0.05, 0.35, and 0.75 for Jiangxi red soil, Taihu paddy soil, and Northeast China black soil, respectively. Results of this work suggest that under alkaline conditions, tralkoxydim adsorption becomes smaller; thus, assessments on its mobility and potential groundwater impact should focus on these soil types.




Similar content being viewed by others
References
Arias-Estévez, M., López-Periago, E., Martínez-Carballo, E., & Simal-Gándara, J. (2006). Carbofuran sorption kinetics by corn crop soils. Bulletin of Environmental Contamination and Toxicology, 77, 267–273.
Chen, G., Lin, C., Chen, L., & Yang, H. (2010). Effect of size-fractionation dissolved organic matter on the mobility of prometryne in soil. Chemosphere, 79, 1046–1055.
Ding, Q., Wu, H. L., Xu, Y., Guo, L. J., Liu, K., Gao, M. H., et al. (2011). Impact of low molecular weight organic acids and dissolved organic matter on sorption and mobility of isoproturon in two soils. Journal of Hazardous Materials, 190, 823–832.
Fang, H., Yu, Y. L., Chu, X. Q., Wang, X. G., Yang, X. E., & Yu, J. Q. (2009). Degradation of chorpyrifos in laboratory soil and its impact on soil microbial functional diversity. Journal of Environmental Sciences, 21, 380–386.
General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China (GAQSIQ). (2014). Test guidelines on environmental safety assessment for chemical pesticides-PART 5: Leaching in soil (p. 2014). Beijing: China Standard Press.
General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China GAQSIQ. (2014a). Test guidelines on environmental safety assessment for chemical pesticides-PART 1: Transformation in soils. Beijing: China Standard Press.
General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China GAQSIQ. (2014b). Test guidelines on environmental safety assessment for chemical pesticides-PART 4: Adsorption/desorption in soils. Beijing: China Standard Press.
General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China GAQSIQ. (2014c). Test guidelines on environmental safety assessment for chemical pesticides-PART 8: Degradation in water-sediment systems. Beijing: China Standard Press.
Gonzalez, P. E., Fernandez, P. M., & Villafran, C. S. M. (1999). Use of bentonite and humic acid as modifying agents in alginate based controlled-release formulations of imidacloprid. Pestic Science, 55(5), 546–552.
Hamaker J W. (1972).Degradation: quantitative aspects //Goring C A I, Hamaker J W. Organic chemicals in the soil Environment. New York: Dekker:253–340.
Helke, R., Andreas, H. R., & Robert, K. (1999). Sorption behavior of prochloraz in different soils. Journal of Agricultural and Food Chemistry, 47(3), 1242–1246.
Jiang, L., Ma, L., Sui, Y., Han, S. Q., & Yang, H. (2011). Mobilization and plant accumulation of prometryne in soil by two different sources of organic matter. Journal of Environmental Monitoring, 13, 1935–1943.
Kah, M., & Brown, C. D. (2006). Adsorption of ionisable pesticides in soils. Reviews of Environmental Contamination and Toxicology, 188, 149–217.
Liu, W. P. (2006). Pesticide environmental chemistry. Beijing: Chemical Industry Press.
Mallawatantri, A. P., & Mulla, D. J. (1992). Herbicide adsorption and organic carbon contents on adjacent low-input versus conventional farms. Journal of Environmental Quality, 21, 546–551.
Margoum, C., Malessard, C., & Gouy, V. (2006). Investigation of various physicochemical and environmental parameter influence on pesticide sorption to ditch bed substratum by means of experimental design. Chemosphere, 63, 1835–1841.
McCALL P J. (1980). Test protocol for environmental fate and migration of toxicants. Proceedings of AOAC. Washington DC:Association of Official Analytical Chemists,:89-109.
Pateiro-Moure, M., Nóvoa-Muñoz, J. C., Arias-Estévez, M., López-Periago, E., Martínez-Carballo, E., & Simal-Gándara, J. (2009a). Quaternary herbicides retention by the amendment of acid soils with a bentonite-based waste from wineries. Journal of Hazardous Materials, 164, 769–775.
Pateiro-Moure, M., Pérez-Novo, C., Arias-Estévez, M., Rial-Otero, R., & Simal-Gándara, J. (2009b). Effect of organic matter and iron oxides on quaternary herbicide sorption–desorption in vineyard-devoted soils. Journal of Colloid and Interface Science, 333, 431–438.
Pateiro-Moure, M., Arias-Estévez, M., & Simal-Gándara, J. (2010). Competitive and non-competitive adsorption/desorption of paraquat, diquat and difenzoquat in vineyard-devoted soils. Journal of Hazardous Materials, 178, 194–201.
Rodríguez-Cruz, M. S., Sánchez-Martín, M. J., Andrades, M. S., & Sánchez-Camazano, M. (2006). Comparison of pesticide sorption by physicochemically modified soils with natural soils as a function of soil properties and pesticide hydrophobicity. Soil and Sediment Contamination, 15, 401–415.
SEPA (1992) Toxic chemicals management office, study on the technology and management of toxic chemicals. Shanghai: Shanghai Popular Science Press,:124.
UNEP (2001)Chemicals. Stockholm convention on persistent organic pollutions (POPs). Geneva, Switzerland, 46.
US EPA. (2008). Fate, transport and transformation test guidelines: OPPTS 835.1240, Leaching studies, EPA712–C–08–010. Washington DC: United States of America.
USEPA. (1998). Sediment/water microcosm biodegradation test, OPPTS 835.3180. Washington DC: United States of America.
USEPA (2008a). Fate transport and transformation test guidelines: OPPTS 835.1230. Adsorption/desorption (batch equilibrium) EPA 712-C-08-009, Washington DC, United States of America.
USEPA (2008b) Fate transport and transformation test guidelines: OPPTS 835.4100. Aerobic soil metabolism, EPA 712-C-08-016, EPA 712-C-08-017. Washington DC, United States of America.
Wu, W. Z., Guo, M., & He, J. (2016). Analysis of tralkoxydim in environmental samples by HPLC. Agrochemicals, 55(11), 811–813.
Acknowledgments
This study was funded by the major national research and development projects (2015ZX07203-007-001-02).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, W.Z., Shan, Z.J., Kong, D.Y. et al. Degradation and adsorption of tralkoxydim in Chinese soils and water-sediment environments. Environ Monit Assess 189, 273 (2017). https://doi.org/10.1007/s10661-017-5984-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-017-5984-5