Abstract
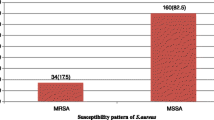
Development of multidrug-resistant pattern in the bacterial community is a major threat to the society. Staphylococcus aureus is perhaps the pathogen of the greatest concern because of its inherent virulence, its ability to cause a diverse array of life-threatening situations and capacity to adapt to different environmental conditions. The aims of this study is to investigate the multidrug-resistant pattern of the coagulase-positive S. aureus isolated from nasal carriage, food, paper currency and wastewater samples. We had also studied the multiple antibiotic resistance index and in vitro production of β-lactamase. The study had found out 130 coagulase-positive S. aureus strains isolated from total of 595 samples such as anterior nares of preschool children (195), hospital nurses (100), drivers (76), food (86), wastewater (3) and paper currency (135) (Indian rupee). The biotypes pattern were as follows; A > D > B > C> UT. Multiple antibiotic resistance (MAR) value clearly defines the multidrug-resistant pattern of the S. aureus among different sources. Statistical analysis (one-way ANOVA) of results obtained indicated that the difference in the antibiotic resistance observed in the 130 bacterial isolates against the 23 different antibiotics used in this study was statically significant (p < 0.01).
Similar content being viewed by others
References
Abd, A. A.-H. (2010). Prevalence of microflora in Lamb meat and Offal at Baghdad abattoir. Basrah Journal of Veterinary Research, 9, 28–34.
Abulreesh, H. H. (2011). Multidrug-resistant staphylococci in the environment. International Conference on Biotechnology and Environment Management, 18, 1–6.
Abulreesh, H. H., & Organji, S. R. (2011). The prevalence of multidrug-resistant staphylococci in food and environment of Makkah, Saudi Arabia. Research Journal of Microbiology, 6(6):510–523. doi:10.3923/jm.2011.510.523
Adegoke, A. A., & Okoh, A. I. (2011). The in vitro effect of vancomycin on multidrug resistant Staphylococcus aureus from hospital currency notes. African Journal of Microbiology Research, 5(14), 1881–1887.
Ahmed, U. S. M., Parveen, S., Nasreen, T., & Feroza, B. (2010). Evaluation of the microbial contamination of Bangladesh paper currency notes (Taka) in circulation. Advances in Biological Research, 4(5), 266–271.
Al-Sheddy, I. A., Fung, D. Y., & Kastner, C. L. (1995). Microbiology of fresh and restructured lab meat: a review. Critical Reviews in Microbiology, 21(1), 31–52.
Artan, O. M., Baykam, Z., & Artan, C. (2008). Nasal carriage of Staphylococcus aureus in healthy preschool children. Japanese Journal of Infectious Diseases, 61, 70–72.
Bauer, A. W., Kirby, W. M. M., Sherris, J. C., & Turck, M. (1966). Antibiotics susceptibility testing by a standardized single disk method. American Journal of Clinical Pathology, 45(4), 493–496.
Borjesson, S., Matussek, A., Melin, S., Lofgren, S., & Lindgren, P. E. (2010). Methicillin-resistant Staphylococcus aureus (MRSA) in municipal wastewater: an uncharted threat? Applied Microbiology, 108, 1244–1251.
Chatterjee, S. S., Ray, P., Aggarwal, A., Das, A., & Sharma, M. (2009). A community-based study on nasal carriage of Staphylococcus aureus. Indian Journal of Medical Research, 130(12), 742–748.
Chitnis, V., Chitnis, D., Patil, S., & Kant, R. (2000). Hospital effluent: a source of multiple drug-resistant bacteria. Current Science, 79(7), 989–991.
CLSI. (2011). Performance standards for antimicrobial susceptibility testing; 21st Information Supplement. Pa: Wayne.
Coia, J. E., Carter, F. T., Baird, D., & Platt, D. J. (1990). Characterisation of methicillin-resistant Staphylococcus aureus by biotyping, immunoblotting and restriction enzyme fragmentation patterns. Medical Microbiology, 31, 125–132.
CPCB (2011). Guide manual: water and wastewater analysis. Online at <http://www.cpcb.nic.in/upload/NewItems/NewItem_171_guidemanualw%26wwanalysis.pdf>. Accessed 19 December 2011.
Cunha, M. L. R. S., Sinzato, Y. K., & Silveira, L. V. A. (2004). Comparison of methods for the identification of coagulase-negative staphylococci. Memórias do Instituto Oswaldo Cruz, 99, 855–860.
Datta, S., Akter, A., Shah, I. G., Fatema, K., Islam, T. H., Bandyopadhyay, B., Khan, Z. U. M., & Biswas, D. (2012). Microbiological quality assessment of raw meat and meat products, and antibiotic susceptibility of isolated Staphylococcus aureus. Agriculture, Food and Analytical Bacteriology, 2(3), 187–194.
Dubey, D., Rath, S., Sahu, M. C., Pattnaik, L., Debata, N. K., & Padhy, R. N. (2013). Surveillance of infection status of drug resistant Staphylococcus aureus in an Indian teaching hospital. Asian Pacific Journal of Tropical Disease, 3(2), 133–142.
Elmund, G. K., Morrison, S. M., Grant, D. W., & Nevins, M. P. (1971). Role of excreted chlortetracycline in modifying the decomposition process in feedlot waste. Bulletin of Environmental Contamination and Toxicology, 6, 129–135.
Elumalai, E. K., David, E., & Hemachandran, J. (2012). Bacterial contamination of Indian currency notes (Rupee). International Journal of Occupational and Environmental Medicine, 3(4), 204–205.
Eze, V. C., & Ivuoma, N. (2012). Evaluation of microbiological quality of fresh goat meat sold in Umuahi market, Abia State, Nigeria. Pakistan Journal of Nutrition, 11(9), 782–786.
Feinman, S. E., & Matheson, J. C. (1978). Draft environmental impact statement: Subtherapeutic antibacterial agents in animal feeds. Food and drug administration department of health, education and welfare report. Food and drug administration, p. 372, Washington.
Ghamdi, A. A., Abdelmalek, S. M. A., Azhar, E. I., Wakidi, M. H., & Alsaied, Z. (2011). Bacterial contamination of Saudi “one” riyal paper notes. Southeast Asian Journal of Tropical Medicine and Public Health, 42(3), 711–716.
Goktas, P., & Oktay, G. (1992). Bacteriological examination of the currency notes. Microbiology Bulletin, 26, 344–348.
Goud, R., Gupta, S., Neogi, U., Agarwal, D., Naidu, K., Chalannavar, R., & Subhaschandra, G. (2011). Community prevalence of methicillin and vancomycin resistant Staphylococcus aureus in and around Bangalore, southern India. Revista da Sociedade Brasileira de Medicina Tropical, 44(3), 309–312.
Greeson, K., Suliman, G. M., Sami, A., Alowaimer, A., & Koohmaraie, M. (2013). Frequency of antibiotic resistant Salmonella, Escherichia coli, Enterococcus, and Staphylococcus aureus in meat in Saudi Arabia. African Journal of Microbiology Research, 7(4), 309–316.
Gundogan, N., Citak, S., Yucel, N., & Devren, A. (2005). A note on the incidence and antibiotic resistance of Staphylococcus aureus isolated from meat and chicken samples. Meat Science, 69(4), 807–810.
Jay, J. (1986). Staphylococcal gastroenteritis. In Modern food microbiology (3rd ed., pp. 437–458). New York: Van Nostrand Reinhold.
Jevons, M. P. (1961). “Celbenin”-resistant staphylococci. British Medical Journal, 1, 124–125.
Joshi, D. R., Shrestha, S. N., Bomjan, R., & Poudel, K. (2008). Nasal carriage of Staphylococcus aureus among health school children in Kathmandu valley. Nepal Journal of Science and Technology, 9, 139–142.
Kakhandki, L. S., & Peerapur, B. V. (2012). Study of nasal carriage of MRSA among the clinical staff and health care workers of a teaching hospital of Karnataka, India. Al Ameen Journal of Medical Sciences, 5(4), 367–370.
Kirby, W. M. M. (1944). Extraction of high potent Penicillin inactivator from Penicillin resistant staphylococci. Clinical Investigation, 24(2), 170–174.
Kloos, W. E., & Schleifer, K. H. (1975). Simplified scheme for routine identification of human staphylococcus species. Clinical Microbiology, 1(1), 82–88.
Kluytmans, W. J. A. J. (2010). Methicillin-resistant Staphylococcus aureus in food products: cause for concern or case for complacency? Clinical Microbiology and Infection, 16, 11–15.
Krumperman, P. H. (1983). Multiple antibiotic resistance indexing of Escherichia coli to identify high-risk sources of fecal contamination of foods. Applied and Environmental Microbiology, 46(1), 165–170.
Kumar, R., & Prasad, A. (2010). Detection of E. coli and Staphylococcus in milk and milk products in and around Pantnagar. Veterinary World, 3(11), 495–496.
Kummerer, K. (2004). Resistance in the environment. Antimicrobial Chemotherapy, 54, 311–320.
Lamichhane, J., Adhikary, S., Gautam, P., Maharjan, R., & Dhakal, B. (2009). Risk of handling paper currency in circulation chances of potential bacterial transmittance. Nepal Journal of Science and Technology, 10, 161–166.
Lamprell, H., Villard, L., Chamba, J. F., Beuvier, E., Borges, E., Maurin, F., Mazerolles, G., Noel, Y., & Kodjo, A. (2004). Identification and biotyping of coagulase positive staphylococci (CPS) in ripened French raw milk cheeses and their in vitro ability to produce enterotoxin. Revue de Médecine Vétérinaire, 155(2), 92–96.
Lowy, F. D. (1998). Staphylococcus aureus infections. New England Journal of Medicine, 339, 520–532.
Mims, C., Dockrell, H. M., Goering, H. V., Roitt, I., Wakelin, D., & Zuckerman, M. (2006). A text book of medical microbiology (3rd ed., p. 585). U. K: Mosby press.
Morandi, S., Brasca, M., Lodi, R., Cremonesi, P., & Castiglioni, B. (2007). Detection of classical enterotoxin genes in Staphylococcus aureus from milk and dairy products. Veterinary Microbiology, 124, 66–72.
Nazir, K. H. M. N. H., & Islam, T. (2007). Association of bacteria in stored bakery foods of retailers shops in Mymensingh, Bangladesh. Bangladesh Society of Agriculture Science and Technology, 4(1&2), 21–24.
Neel, R. (2013). Multidrug resistance of isolates of methicillin resistant Staphylococcus aureus (MRSA) in paper currency notes from restaurants and hotels in Lusaka in Zambia. International Journal of Pharmacy and Pharmaceutical Sciences, 5(1), 363–366.
Normanno, G., La Salandra, G., Dambrosio, A., Quaglia, N. C., Corrente, M., Parisi, A., Santagad, G., Firinu, A., Crisetti, E., & Celano, G. V. (2007). Occurrence, characterization and antimicrobial resistance of enterotoxigenic Staphylococcus aureus isolated from meat and dairy products. International Journal of Food Microbiology, 115, 290–296.
Oyero, O. G., & Emikpe, B. O. (2007). Preliminary investigation on the microbial contamination of Nigerian currency. International Journal of Tropical Medicine, 2(2), 29–32.
Pathak, A., Marothi, Y., Iyer, R. V., Singh, B., Sharma, M., Eriksson, B., Macaden, R., & Lundborg, C. S. (2010). Nasal carriage and antimicrobial susceptibility of Staphylococcus aureus in healthy preschool children in Ujjain, India. BMC Pediatrics, 10, 1–8.
Peles, F., Wagner, M., Varga, L., Hein, I., Rieck, P., Gutser, K., Keresturi, P., Kardos, G., Turcsanyi, I., Beri, B., & Szabo, A. (2007). Characterization of Staphylococcus aureus strains isolated from bovine milk in Hungry. International Journal of Food Microbiology, 118, 186–193.
Rachel, E., Golstein, R., Micallef, S. A., Gibbs, S. G., Davis, J. A., He, X., George, A., Kleinfelter, L. M., Schreiber, N. A., Mukherjee, S., Sapkota, A., & Joseph, S. W. (2012). Methicillin-resistant Staphylococcus aureus (MRSA) detected at four U.S. wastewater treatment plants. Environmental Health Perspectives, 120(11), 1551–1558.
Rashid, Z., Farzana, K., Sattar, A., & Murtaza, G. (2012). Prevalence of nasal Staphylococcus aureus and methicillin-resistant Staphylococcus aureus in hospital personal and associated risk factors. Acta Poloniae Pharmacutica -Drug Research, 69(5), 985–991.
Robert, G. W., & Edmund, F. W. (1970). Activity of penicillinase in Staphylococcus aureus as studied by the iodometric method. Infectious Diseases, 121(4), 433–437.
Rongpharpi, S. R., Hazarika, N. K., & Kalita, H. (2013). The prevalence of nasal carriage of Staphylococcus aureus among healthcare workers at tertiary care hospital in Assam with special reference to MRSA. Clinical and Diagnostic Research, 7(2), 257–260.
Sawalha, H., & Mowais, M. A. (2012). Bacterial contamination of paper banknotes in circulation; a case study in the Jenin district, Palestine. Science, 1(2), 36–39.
Scherrer, D., Corti, S., Muehlherr, J. E., Zweifel, C., & Stephan, R. (2004). Phenotypic and genotypic characteristics of Staphylococcus aureus isolates from raw bulk-tank milk samples of goats and sheep. Veterinary Microbiology, 101, 101–107.
Schwartz, T., Kohnen, W., Jansen, B., & Obst, U. (2003). Detection of antibiotic-resistant bacteria and their resistance genes in wastewater, surface water and drinking water biofilms. FEMS Microbiology Ecology, 43, 325–335.
Selvan, P., Narendra Babu, R., Sureshkumar, S., & Venkataramanujam, V. (2007). Microbial quality of retail meat products available in Chennai city. American Journal of Food Technology, 2(1), 55–59.
Shanth, J., Saravanan, T., & Balagurunathan, R. (2012). Isolates of tannery effluent and their antibiogram from effluent plant in south India. Chemical and Pharmaceutical Research, 4(4), 1974–1977.
Sharma, A., & Dhanashree, B. (2011). Screening of currency in circulation for bacterial contamination. Current Science, 100(6), 822–825.
Singh, P., & Prakash, A. (2008). Isolation of Escherichia coli, Staphylococcus aureus and Listeria monocytogenes from milk products sold under market conditions at Agra region. Acta Agriculturae Slovenica, 92(11), 83–88.
Singh, D. V., Thakur, K., Kalpna, & Goel, A. (2002). Microbiological surveillance of currency. Indian Journal of Medical Microbiology, 20(1), 53.
The Tribune (2013a). 45 Hospitals get notice for violating norms. Online at <http://www.tribuneindia.com/2013/20130309/punjab.htm#3>. Accessed 8 March 2013.
The Tribune (2013b). Punjab Pollution Control Board team raids PIMS, SGL hospitals finds irregularities in biomedical waste disposal system in both institutes. Online at <http://www.tribuneindia.com/2013/20130104/jaltrib.htm>. Accessed 3 January 2013.
The Tribune (2010). Punjab Pollution Control Board directed against erring hospitals. Online at http://www.tribuneindia.com/2010/20101119/punjab.htm#1. Accessed 18 November 2010.
The Tribune (2011). Punjab Pollution Control Board orders closures of 3 erring hospitals were found violating biomedical waste disposal norms; several others left off with warning. Online at <http://www.tribuneindia.com/2011/20110717/punjab.htm#1>. Accessed 16 July 2011.
Vaishnavi, C., Singh, S., Grover, R., & Singh, K. (2001). Bacteriological study of Indian cheese (paneer) sold in Chandigarh. Indian Journal of Medical Microbiology, 19(4), 224–226.
Vinodhkumaradithyaa, A., Uma, A., Srinivasan, M., Ananthalakshmi, I., Nallasivam, P., & Thirumalaikolundusubramanian, P. (2009). Nasal carriage of methicillin-resistant Staphylococcus aureus among surgical unit staff. Japanese Journal of Infectious Diseases, 62, 228–229.
Waldvogel, F. A. (2000). Staphylococcus aureus (Including staphylococcal toxic shock). In G. L. Mandell, J. E. Bennet, & R. Dolin (Eds.), Principles and practice of infectious diseases (pp. 2069–2092). Philadelphia: Churchill Livingstone.
Wertheirn, H. F. L., Melles, D. C., Vos, M. C., Leeuwen, W. V., Belkum, A. V., Verbrugh, H. A., & Nouwen, J. L. (2005). The role of nasal carriage in Staphylococcus aureus infections. The Lancet Infectious Diseases, 5, 751–762.
Williams, R. E. O. (1963). Healthy carriage of Staphylococcus aureus: its prevalence and importance. Bacteriological Reviews, 27, 56–71.
Yu-Cheng, C. H., Wan-Wen, L., Chin-Ming, F., Wan-Yu, P., Chien-Shun, C. H., & Hau-Yang, T. (2008). PCR detection of Staphylococcal enterotoxins (SEs) N, O, P, Q, R, U, and survey of SE types in Staphylococcus aureus isolates from food-poisoning cases in Taiwan. International Journal of Food Microbiology, 121, 66–73.
Acknowledgments
We are thankful to the Super Religare Laboratories, Beas, for providing reference strain of S. aureus ATCC-25923 for this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, H., Palaha, R., Kaur, N. et al. Prevalence of multidrug-resistant, coagulase-positive Staphylococcus aureus in nasal carriage, food, wastewater and paper currency in Jalandhar city (north-western), an Indian state of Punjab. Environ Monit Assess 187, 4134 (2015). https://doi.org/10.1007/s10661-014-4134-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-014-4134-6