Abstract
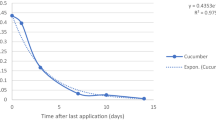
Persistence and risk assessment of spirotetramat and imidacloprid in chilli fruits were studied following three applications of a mixture formulation of spirotetramat (12 %) and imidacloprid (12 %) at 1000 and 2000 mL ha−1. Residues of spirotetramat and imidacloprid in chilli were estimated by high-performance liquid chromatograph (HPLC). Residues of spirotetramat and imidacloprid dissipated to more than 65 % after 3 days at both the dosages. Residues of spirotetramat on chilli fruits were found to be below its limit of quantification (LOQ) of 0.03 mg kg−1 after 5 and 7 days for recommended and double the recommended dosages, respectively. Similarly, imidacloprid residues were found to be below its LOQ of 0.01 mg kg−1 at 7 and 10 days, respectively. Half-life periods for spirotetramat were found to be 1.91 and 1.30 days, whereas, for imidacloprid, these values were observed to be 1.41 and 1.65 days at recommended and double the recommended dosages, respectively. Red chilli samples collected after 20 days of the last application did not show the presence of spirotetramat and imidacloprid at their respective determination limit. As the theoretical maximum residue contributions on chilli fruits are found to be less than the maximum permissible intake values on initial deposits, a waiting period of 1 day may follow to reduce risk before consumption at the recommended dose.

Similar content being viewed by others
References
Anonymous. (2002). Opinion of the scientific committee on food on capsaicin. European Commission Health & Consumer Protection Directorate-General, (http://ec.europa.eu/food/fs/sc/scf/out120_en.pdf)
Brittain, C., & Potts, S. G. (2011). The potential impacts of insecticides on the life-history traits of bees and the consequences for pollination. Basic and Applied Ecology, 12, 321–331.
Brück, E., Elbert, A., Fischer, R., Krueger, S., Kühnhold, J., Klueken, A. M., Nauen, R., Niebes, J. F., Reckmann, U., Schnorbach, H. J., Steffens, R., & Waetermeulen, X. (2009). Movento®, an innovative ambimobile insecticide for sucking insect pest control in agriculture: biological profile and field performance. Crop Protection, 28, 838–844.
Buchholz, A., & Nauen, R. (2002). Translocation and translaminar bioavailability of two neonicotinoid insecticides after foliar application to cabbage and cotton. Pest Management Science, 58, 10–16.
Chahil, G. S., Mandal, K., Sahoo, S. K., Battu, R. S., & Singh, B. (2014). Risk assessment of β-cyfluthrin and imidacloprid in chickpea pods and leaves. Ecotoxicology and Environmental Safety, 101, 177–183.
Chaudhary, B. (2000). Solanaceous fruits and cole crops vegetables (pp. 63–84). India: National Book Trust.
Dey, P. K., Sarkar, P. K., & Somchoudhury, A. K. (2001). Efficacy of different treatment schedules of profenofos against major pests of chilli. Pestology, 25, 26–29.
European Food Safety Authority. (2013). Reasoned opinion on the modification of the existing MRLs for spirotetramat in strawberries, bananas, table olives, pineapples and shallots. EFSA Journal, 11, 3361–3398.
Girolami, V., Greatti, M., Bernardo, A. D., Tapparo, A., & Giorio, C. (2009). Translocation of neonicotinoid insecticides from coated seeds to seedling guttation drops: a novel way of intoxication for bees. Journal of Economic Entomology, 102, 1808–1815.
Hoskins, W. M. (1966). Mathematical treatment of the rate of loss of pesticide residues. FAO Plant Protection Bulletin, 9, 163–168.
Jyot, G., Mandal, K., Battu, R. S., & Singh, B. (2013). Estimation of chlorpyriphos and cypermethrin residues in chilli (Capsicum annuum L.) by gas-liquid chromatography. Environmental Monitoring and Assessment, 185, 5703–5714.
Lozano, F., Kemper, K., & Tundisi, H. (2008). Field development of Movento Plus for sucking pest insect control in Brazil. Bayer Crop Science Journal, 61, 329–341.
Mandal, K., Chahil, G. S., Sahoo, S. K., Battu, R. S., & Singh, B. (2010). Dissipation kinetics of spirotetramat and imidacloprid in brinjal and soil under subtropical conditions of Punjab, India. Bulletin of Environmental Contamination and Toxicology, 84, 225–229.
Mohapatra, S., Deepa, M., Leka, S., Nethravathi, B., Radhika, B., & Gourishanker, S. (2012). Residue dynamics of spirotetramat and imidacloprid in/on mango and soil. Bulletin of Environmental Contamination and Toxicology, 89, 862–867.
Mukherjee, I., & Gopal, M. (2000). Environmental behavior and translocation of imidacloprid in eggplant, cabbage and mustard. Pest Management Science, 56, 932–936.
Narayanan, S. S., Hedge, S., Sadananda, A. R., & Chelliah, S. (1999). Commerce and utility considerations of chillies. Kisan World, 26, 73–75.
Pandiselvi, S., Sathyanarayanan, S., & Ramesh, A. (2010). Determination of spirotetramat and imidacloprid residues in cotton seed, lint, oil and soil by HPLC UV method and their dissipation in cotton plant. Pesticide Research Journal, 22, 168–173.
Raju, K. V., & Luckose, C. K. (1991). Trends in area, production and exports of chillies from India. Agricultural Situation in India, 45, 767–772.
Ramanaidu, K., & Cutler, G. C. (2012). Different toxic and hormetic responses of Bombus impatiens to Beauveria bassiana, Bacillus subtilis and Spirotetramat. Pest Management Science, 69, 949–954.
Reddy, K. N., Satyanarayana, S., & Reddy, K. D. (2007a). Persistence of some insecticides in chillies. Pesticide Research Journal, 19, 234–236.
Reddy, K. D., Reddy, K. N., & Mahalingappa, P. B. (2007b). Dissipation of fipronil and profenofos residues in chillies (Capsicum annum L.). Pesticide Research Journal, 19, 106–107.
Robson, J. D., Wright, M. G., & Almeida, R. P. P. (2007). Effect of imidacloprid foliar treatment and banana leaf age on Pentalonia nigronervosa (Hemiptera, Aphididae) survival. New Zealand Journal of Crop and Horticultural Science, 35, 415–422.
Sahoo, S. K., Chahil, G. S., Mandal, K., Battu, R. S., & Singh, B. (2012). Estimation of spirotetramat and imidacloprid in okra fruits and soil by chromatography technique. Journal of Environmental Science and Health. Part. B, 47, 42–50.
Salles, L. A. (2002). Os insetos como vetores de patógenos de plantas. Cultivar, 13, 3–6.
Sharma, K.K. (2013). Pesticide residue analysis manual. Directorate of Information and Publications of Agriculture, Indian Council of Agricultural Research, New Delhi, p. 251.
Singh, B., Mandal, K., Sahoo, S. K., Bhardwaj, U., & Battu, R. S. (2013). Development and validation of HPLC method for determination of spirotetramat and spirotetramat cis enol in various vegetables and soil. Journal of Association of Official Analytical Chemists - J. AOAC Int., 96, 670–675.
Smiley, R. W., Marshall, J. M., & Yan, G. P. (2011). Effect of foliarly applied spirotetramat on reproduction of Heterodera avenae on wheat roots. Plant Disease, 95, 983–989.
Tomizawa, M., Latli, B., & Casida, J. E. (1999). Structure and function of insect nicotinic acetylcholine receptors studied with nicotinic insecticide affinity probes. In I. Yamamoto & J. E. Casida (Eds.), Nicotinoid insecticides and the nicotinic acetylcholine receptor (pp. 271–292). Tokyo: Springer.
Van Waetermeulen, X., Bruck, E., Elbert, A., Fischer, R., Krueger, S., Kuhnhold, J., Nauen, R., Niebes, J. F., Reckmann, U., Schnorbach, H. J., & Steffens, R. (2007). Spirotetramat, an innovative fully systemic insecticide for sucking insect pest control in agriculture: biological profile and field performance. XVI International Plant Protection Congress Proceeding, 1, 60–67.
Acknowledgments
The authors are thankful to M/s Bayer CropScience, India, and the Indian Council of Agricultural Research, India, for sponsoring the project. The authors are also thankful to the Head of the Department of Entomology, PAU, Ludhiana, for providing research facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chahil, G.S., Mandal, K., Sahoo, S.K. et al. Risk assessment of mixture formulation of spirotetramat and imidacloprid in chilli fruits. Environ Monit Assess 187, 4105 (2015). https://doi.org/10.1007/s10661-014-4105-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-014-4105-y