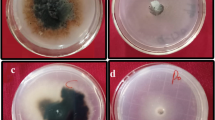
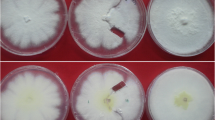
Abstract
Charcoal rot is an economically important fungal disease of economically important crops. Several species of the Macrophomina genus, such as M. pseudophaseolina and M. euphorbiicola, may act as the causal agents of charcoal rot. The current study evaluates: a) the diversity of vegetative compatibility reactions in Brazilian isolates of M. pseudophaseolina (totalling 27) and M. euphorbiicola (totalling 3), b) the genetic variability of Brazilian isolates, and c) the occurrence of parasexual recombination in M. pseudophaseolina isolates. In complementation tests, 2 and 18 Vegetative Compatibility Groups (VCGs) were identified for the M. euphorbiicola and M. pseudophaseolina isolates, respectively. Correlations between VCG and the isolates´ geographic origin were not found since most isolates derived from the same localities were grouped in distinct VCGs, demonstrating their genetic variability. Further, RAPD analysis of the isolates did not reveal significant correlations between genotype, geographic location and hosts, since isolates derived from distinct localities or hosts formed the same RAPD group. Heterozygous diploids were produced as fast-growing sectors by heterokaryons formed with M. pseudophaseolina isolates, demonstrating, for the first time, the occurrence of the parasexual cycle in the species, based on molecular and phenotypic evidence. Diploids spontaneously produced paternal segregants and parasexual recombinants, demonstrating that parasexuality is an important mechanism for transferring genetic material in filamentous fungi. Results suggest that parasexual recombination may play an important role in the genetic variability of M. pseudophaseolina and may be the cause for the origin of new pathotypes, which could compromise disease control strategies in crops.




Similar content being viewed by others
References
Babu, B. K., Reddy, S. S., Yadav, M. K., Sukumar, M., Mishra, V., Saxena, A. K., & Arora, D. K. (2010). Genetic diversity of Macrophomina phaseolina isolates from certain agro-climatic regions of India by using RAPD markers. Indian Journal of Microbiology, 50(2), 199–204. https://doi.org/10.1007/s12088-010-0033-x
Brooker, N. L., Leslie, J. F., & Dickman, M. B. (1991). Nitrate non-utilizing mutants of Colletotrichum and their use in studied of vegetative compatibility and genetic relatedness. Phytopathology, 81(6), 672–677. https://doi.org/10.1094/Phyto-81-672
Carlile, M. J. (1987). Genetic exchange and gene flow: Their promotion and prevention. In A. D. M. Rayner, C. M. Brasier, & D. Moore (Eds.), Evolutionary biology of the Fungi (pp. 203–214). Cambridge University Press.
Caten, C. E., & Newton, A. C. (2000). Variation in cultural characteristics, pathogenicity, vegetative compatibility and electrophoretic karyotype within field populations of Stagonospora nodorum. Plant Pathology, 49(2), 219–226. https://doi.org/10.1046/j.1365-3059.2000.00441.x
Correll, J. C., Klittich, C. J. R., & Leslie, J. F. (1989). Heterokaryon self-incompatibility in Gibberella fujikuroib (Fusarium moniliforme). Mycological Research, 93(1), 21–27. https://doi.org/10.1016/S0953-7562(89)80130-3
Crous, P. W., Slippers, B., Wingfield, M. J., Walter, J. R., Marasas, F. O., Philips, A. J. L., Alves, A., Burgess, T., Barber, P., & Groenewald, J. Z. (2006). Phylogenetic lineages in the Botryosphaeriaceae. Studies in Mycology, 55(1), 235–253. https://doi.org/10.3114/sim.55.1.235
Dhingra, O. D., & Sinclair, J. B. (1978). Biology and pathology of Macrophomina phaseolina. UFV, Imprensa Universitária.
Ebihara, Y., Nagao, H., Koike, M., Shiraisho, T., & Iijima, T. (1999). How do Japanese isolates of Verticillium dahliae correspond with standardized VCG testers? Mycoscience, 40(4), 333–343. https://doi.org/10.1007/BF02463877
Elias, K. S., & Schneider, R. W. (1991). Vegetative compatibility groups in fusarium oxysporum f. sp. lycopersici. Phytopathology, 81(2), 159–162. https://doi.org/10.1094/Phyto-81-159
Fuhlbohm, M. J., Ryley, M. J., & Aitken, E. A. B. (2012). New weed hosts of Macrophomina phaseolina in Australia. Australasian Plant Disease Notes, 7(1), 193–195. https://doi.org/10.1007/s13314-012-0082-6
Glass, N. L., Jacobson, D. J., & Shiu, P. K. T. (2000). The genetics of hyphal fusion and vegetative incompatibility in filamentous ascomycete fungi. Annual Review of Genetics, 34(1), 165–186. https://doi.org/10.1146/annurev.genet.34.1.165
Gupta, G. K., Sharma, S. K., & Ramteke, R. (2012). Biology, epidemiology and management of the pathogenic fungus Macrophomina phaseolina (Tassi) Goid with special reference to charcoal rot of soybean (Glycine max (L.) Merrill). Journal of Phytopathology, 160(4), 167–180. https://doi.org/10.1111/j.1439-0434.2012.01884.x
Hawthorne, B. T., & Rees-George, J. (1996). Use of nitrate non-utilizing mutants to study vegetative incompatibility in Fusarium solani (Nectria haematococca), especially members of mating populations I, V and VI. Mycological Research, 100(9), 1075–1081. https://doi.org/10.1016/S0953-7562(96)80216-4
Hernández-Delgado, S., Reyes-Valdés, M. H., Rosales-Serna, R., & Mayek-Pérez, N. (2009). Molecular markers associated with resistance to Macrophomina phaseolina (Tassi) Goldin common bean. Journal of Plant Pathology, 91, 163–170.
Iqbal, U., & Mukhtar, T. (2014). Morphological and pathogenic variability among Macrophomina phaseolina isolates associated with mungbean (Vigna radiata L.) Wilczek from Pakistan. The Scientific World Journal, 2014, article ID 950175. https://doi.org/10.1155/2014/950175
Jamshidi, S., & Jamshidi, S. (2011). NTSYSpc 2.02 e implementation in molecular biodata analysis (clustering, screening, and individual selection). In Proceedings of the 4th international conference on environmental and computer science (pp. 165–169).
Katan, T., Zamir, D., Sarfatti, M., & Katan, J. (1991). Vegetative compatibility groups and subgroups in fusarium oxysporum f.sp. radicis-lycopersici. Phytopathology, 81(3), 255–262. https://doi.org/10.1094/Phyto-81-255
Kaur, S., Dhillon, G. S., Brar, S., Vallad, G. E., Chand, R., & Chauhan, V. B. (2012). Emerging phytopathogen Macrophomina phaseolina: Biology, economic importance and current diagnostic trends. Critical Reviews in Microbiology, 38(2), 136–151. https://doi.org/10.3109/1040841X.2011.640977
Leslie, J. F. (1993). Fungal vegetative compatibility. Annual Review of Phytopathology, 31(1), 127–150. https://doi.org/10.1146/annurev.py.31.090193.001015
Lodha, S., & Mawar, R. (2020). Population dynamics of Macrophomina phaseolina in relation to disease management: A review. Journal of Phytopathology, 168(1), 1–17. https://doi.org/10.1111/jph.12854
Loudon, K. W., Burnie, J. P., Coke, A. P., & Matthews, R. C. (1993). Application of polymerase chain reaction to fingerprinting aspergillus fumigatus by random amplification of polymorphic DNA. Journal of Clinical Microbiology, 31(5), 1117–1121. https://doi.org/10.1128/jcm.31.5.1117-1121.1993
Machado, A. R., Pinho, D. B., Soares, D. J., Gomes, A. A. M., & Pereira, O. L. (2018). Bayesian analyses of five gene regions reveal a new phylogenetic species of Macrophomina associated with charcoal rot on oilseed crops in Brazil. European Journal Plant Pathology, 153(1), 89–100. https://doi.org/10.1007/s10658-018-1545-1
Mengistu, A., Wrather, A., & Rupe, J. C. (2015). Charcoal rot. In G. L. Hartman, J. C. Rupe, E. F. Sikora, L. L. Domier, J. A. Davis, & K. L. Steffey (Eds.), Compendium of soybean diseases and pests (pp. 67–69). American Phytopathological Society.
Noguchi, M. T., Yasuda, N., & Fujita, Y. (2006). Evidence of genetic exchange by parasexual recombination and genetic analysis of pathogenicity and mating type of parasexual recombinants in rice blast fungus, Magnaporthe oryza. Phytopathology, 96(7), 746–750. https://doi.org/10.1094/PHYTO-96-0746
Paccola-Meirelles, L. D., & Azevedo, J. L. (1991). Parasexuality in Beauveria bassiana. Journal of Invertebrate Pathology, 57(2), 172–176. https://doi.org/10.1016/0022-2011(91)90113-5
Pereira, T. S., Machado, G. N. E., Franco, C. C. S., Mathias, P. C. F., Soares, D. J., Bock, C. H., Nunes, W. M. C., Zanutto, C. A., & Castro-Prado, M. A. A. (2018). Heterokaryosis and diploid formation among Brazilian isolates of Macrophomina phaseolina. Plant Pathology, 67(9), 1857–1864. https://doi.org/10.1111/ppa.12919
Pontecorvo, G. (1956). The parasexual cycle in fungi. Annual Review of Microbiology, 10(1), 393–400. https://doi.org/10.1146/annurev.mi.10.100156.002141
Poudel, B., Shivas, R. G., Adorada, D. L., Barbetti, M. J., Bithell, S. L., Kelly, L. A., Moore, N., Sparks, A. H., Tan, Y. P., Thomas, G., Van Leur, J., & Vaghefi, N. (2021). Hidden diversity of Macrophomina associated with broadacre and horticultural crops in Australia. European Journal of Plant Pathology, 161(1), 1–23. https://doi.org/10.1007/s10658-021-02300-0
Reis, E. M., Boaretto, C., & Danelli, A. L. D. (2014). Macrophomina phaseolina: Density and longevity of microsclerotia in soybean root tissues and free on the soil, and competitive saprophytic ability. Summa Phytopathologica, 40(2), 128–133. https://doi.org/10.1590/0100-5405/1921
Reznikov, S., Chiesa, M. A., Pardo, E. M., De Lisi, V., Bogado, N., González, V., Ledesma, F., Morandi, E. N., Ploper, D., & Castagnaro, A. P. (2019). Soybean-Macrophomina phaseolina-specific interactions and identification of a novel source of resistance. Phytopathology, 109(1), 63–73. https://doi.org/10.1094/PHYTO-08-17-0287-R
Rodríguez-Guerra, R., Ramírez-Rueda, M.-T., Martínez de la Veja, O., & Simpson, J. (2003). Variation in genotype, pathotype and anastomosis groups of Colletotrichum lindemuthianum isolates from Mexico. Plant Pathology, 52(2), 228–235. https://doi.org/10.1046/j.1365-3059.2003.00808.x
Romero-Luna, M. P., Mueller, D., Mengistu, A., Singh, A. K., Hartman, G. L., & Wise, K. A. (2017). Advancing our understanding of charcoal rot in soybeans. Journal of Integrated Pest Management, 8(1), 1–8. https://doi.org/10.1093/jipm/pmw020
Rosada, L. J., Sant'Anna, J. R., Franco, C. C. S., Esquissato, G. N. M., Santos, P. A. S. R., Yajima, J. P. R. S., Ferreira, F. D., Machinski, M., Corrêa, B., & Castro-Prado, M. A. A. (2013). Identification of aspergillus flavus isolates as potential biocontrol agents of aflatoxin contamination in crops. Journal of Food Protection, 76(6), 1051–1055. https://doi.org/10.4315/0362-028X.JFP-12-436
Sarr, M. P., Ndiaye, M., Groenewald, J. Z., & Crous, P. (2014). Genetic diversity in Macrophomina phaseolina, the causal agent of charcoal rot. Phytopathologia Mediterranea, 53(3), 250–268. https://doi.org/10.14601/Phytopathol_Mediterr-13736
Saupe, S. J. (2000). Molecular genetics of heterokaryon incompatibility in filamentous ascomycetes. Microbiology and Molecular Biology Reviews, 64(3), 489–502. https://doi.org/10.1128/MMBR.64.3.489-502.2000
Sexton, Z. F., Hughes, T. J., & Wise, K. A. (2016). Analyzing isolate variability of Macrophomina phaseolina from a regional perspective. Crop Protection, 81, 9–13. https://doi.org/10.1016/j.cropro.2015.11.012
Souza-Júnior, S. A., Becker, T. C. A., & Castro-Prado, M. A. A. (2007). Asexual recombination in a uvsH mutant of Aspergillus nidulans. Biological Research, 40(1), 65–71. https://doi.org/10.4067/S0716-97602007000100007
Souza-Paccola, E. A., Fávaro, L. C. L., Casela, C. R., & Paccola-Meirelles, L. D. (2003). Genetic recombination in Colletotrichum sublineolum. Journal of Phytopathology, 151(6), 329–334. https://doi.org/10.1046/j.1439-0434.2003.00727.x
Strom, N. B., & Bushley, K. E. (2016). Two genomes are better than one: History, genetics, and biotechnological applications of fungal heterokaryons. Fungal Biology and Biotechnology, 3(1), 1–4. https://doi.org/10.1186/s40694-016-0022-x
Sybuia, P. A., Pereira, T. S., Esquissato, G. N. M., Castro-Prado, G., Oliveira, H. C. G., Azevedo, J. L., Pamphile, J. A., Pereira, O. C. N., Nunes, W. M. C., Zanutto, C. A., & Castro-Prado, M. A. A. (2020). Limitation of nitrogen source facilitated the production of nonmeiotic recombinants in aspergillus nidulans. Journal of Basic Microbiology, 60(4), 380–385. https://doi.org/10.1002/jobm.201900588
Tindall, H. D. (1983). Vegetables in the tropics. The Macmillan Press LTD.
Türkkan, M., Benli, H. I., Yılmaz, O., Özer, G., Yaman, M., Şahin, N., & Erper, I. (2020). First report of charcoal rot caused by Macrophomina phaseolina on kiwifruit in Turkey. Journal of Plant Patholology, 102(2), 535–535. https://doi.org/10.1007/s42161-019-00428-x
Zhao, L., Cai, J., He, W., & Zhang, Y. (2019). Macrophomina vaccinii sp. nov. causing blueberry stem blight in China. MycoKeys, 55, 1–14. https://doi.org/10.3897/mycokeys.55.35015
Zhao, D., Fan, S., Zhang, D., Pan, Y., Gu, Q., Wang, J., Yang, Z., & Zhu, J. (2021). Parasexual reproduction in Alternaria solani: Simple sequence repeat molecular evidence for haploidization. Mycologia, 113(5), 949–955. https://doi.org/10.1080/00275514.2021.1922243
Acknowledgements
The authors would like to thank Dr. Rui Sales Júnior for kindly sending the isolates 9–27.
Availability of data and material
Fungal isolates are available from Brazilian culture collections.
Code availability
Not applicable.
Funding
The current research was funded by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). P.A.S. is a holder of CAPES fellowship.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Priscila A. Sybuia, Gabriela de Castro-Prado Carlos A. Zanutto, Edilson N. Kaneshima, and Marialba A. A. de Castro-Prado. The first draft of the manuscript was written by Marialba A. A. Castro-Prado and all authors commented on previous versions of the manuscript. All authors read and approved the manuscript and consented for publication in the European Journal of Plant Pathology.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
ESM 1
(DOCX 17 kb)
Fig. S1
RAPD products of M. pseudophaseolina (1–27) and M. euphorbiicola (28–30) isolates using the primer OPW-02. Column M = molecular weight DNA ladder in bp (100–1500), bp = base pairs. Arrows indicate polymorphic bands of 490 bp (a), 600 bp (b), 800 bp (c), 900 bp (d), 950 bp (e), 1100 bp (f), and 1300 bp (g) (PNG 785 kb)
Rights and permissions
About this article
Cite this article
Sybuia, P.A., de Castro-Prado, G., de Carvalho Nunes, W.M. et al. Parasexual recombination in Macrophomina pseudophaseolina and vegetative compatibility reactions in M. euphorbiicola. Eur J Plant Pathol 163, 937–950 (2022). https://doi.org/10.1007/s10658-022-02530-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-022-02530-w