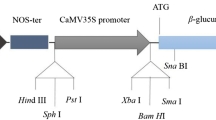
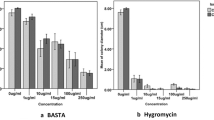
Abstract
The fungus Colletotrichum lindemuthianum has genes that encode different enzymes involved in plant cell wall degradation such as pectinases, which are important for the pathogenicity of various fungi. To analyze the function of pectinase-encoding genes in C. lindemuthianum, the expression of these genes during the interaction with the common bean was evaluated by qPCR. Using these results, the transformation technique mediated by Agrobacterium tumefaciens was performed for the isolation of mutants (ATMT). The expression of 12 genes that code for pectinases in C. lindemuthianum was analyzed. Nine genes displayed differential expression only in the necrotrophic phase, and two only in the biotrophic phase. From the results obtained, four genes were selected for the isolation of mutants. From the 180 transformants analyzed, two mutants for the pecCl6 gene were obtained. The ∆pecCl6 mutants showed no difference in morphology, growth, sporulation, or pathogenicity compared to the wild type. No mutants were isolated from the other analyzed genes. The expression of pectinase-encoding genes indicates the importance of these enzymes during the interaction of C. lindemuthianum with the common bean. The advantages of using ATMT in C. lindemuthianum were its ability to present a single copy of the cassette in the genome of the transformants. A disadvantage was the low number of mutants in relation to the number of transformants obtained. Thus, the ATMT system was not efficient for the deletion of genes encoding pectinases in C. lindemuthianum. The results revealed that the pecCl6 gene is not essential for the pathogenicity of C. lindemuthianum.




Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Auyong, A. S. M., Ford, R., & Taylor, P. W. J. (2012). Genetic transformation of Colletotrichum truncatum associated with anthracnose disease of chili by random insertional mutagenesis. Journal of Basic Microbiology, 52(4), 372–382. https://doi.org/10.1002/jobm.201100250
Catlett, N. L., Lee, B.-N., Yoder, O. C., & Turgeon, B. G. (2003). Split-Marker Recombination for Efficient Targeted Deletion of Fungal Genes. Fungal Genetics Reports, 50(1), 9–11. https://doi.org/10.4148/1941-4765.1150
Centis, S., Guillas, I., Séjalon, N., Esquerré-Tugayé, M. T., & Dumas, B. (1997). Endopolygalacturonase genes from Colletotrichum lindemuthianum: Cloning of CLPG2 and comparison of its expression to that of CLPG1 during saprophytic and parasitic growth of the fungus. Molecular Plant-Microbe Interactions, 10(6), 769–775. https://doi.org/10.1094/MPMI.1997.10.6.769
Chen, Z., Nunes, M. A., Silva, M. C., & Rodrigues, C. J. (2004). Appressorium turgor pressure of Colletotrichum kahawae might have a role in coffee cuticle penetration. Mycologia, 96(6), 1199–1208. https://doi.org/10.1080/15572536.2005.11832868
Cnossen-Fassoni, A., Bazzolli, D. M. S., Brommonschenkel, S. H., Fernandes de Araújo, E., & de Queiroz, M. V. (2013). The pectate lyase encoded by the pecCl1 gene is an important determinant for the aggressiveness of Colletotrichum lindemuthianum. Journal of Microbiology, 51(4), 461–470. https://doi.org/10.1007/s12275-013-3078-9
Cubero, B., & Scazzocchio, C. (1994). sites CREA-mediated. EMBO Journal, 13(2), 407–415.
Dickman, M. B., Patil, S. S., & Kolattukudy, P. E. (1982). Purification, characterization and rôle in infection of an extracellular cutinolytic enzyme from Colletotrichum gloeosporioides Penz. on Carica papaya L. Physiological Plant Pathology, 20(3), 333–347. https://doi.org/10.1016/0048-4059(82)90058-3
dos Santos, L. V., de Queiroz, M. V., Santana, M. F., Soares, M. A., de Barros, E. G., de Araújo, E. F., & Langin, T. (2012). Development of new molecular markers for the Colletotrichum genus using RetroCl1 sequences. World Journal of Microbiology and Biotechnology, 28(3), 1087–1095. https://doi.org/10.1007/s11274-011-0909-x
Dumas, B., Centis, S., Sarrazin, N., & Esquerré-Tugayé, M. T. (1999). Use of green fluorescent protein to detect expression of an endopolygalacturonase gene of Colletotrichum lindemuthianum during bean infection. Applied and Environmental Microbiology, 65(4), 1769–1771.
Fontenelle, M. R., Santana, M. F., Cnossen, A., Bazzolli, D. M. S., Bromonschenkel, S. H., de Araújo, E. F., & de Queiroz, M. V. (2017). Differential expression of genes during the interaction between Colletotrichum lindemuthianum and Phaseolus vulgaris. European Journal of Plant Pathology, 147(3), 653–670. https://doi.org/10.1007/s10658-016-1033-4
Gan, P., Ikeda, K., Irieda, H., Narusaka, M., Connell, R. J. O., Narusaka, Y., et al. (2012). Comparative genomic and transcriptomic analyses reveal the hemibiotrophic stage shift of Colletotrichum fungi. New Phytologist, 197, 1236–1249.
Glass, N. L., Schmoll, M., Cate, J. H. D., & Coradetti, S. (2013). Plant Cell Wall Deconstruction by Ascomycete Fungi. Annual Review of Microbiology, 67(1), 477–498. https://doi.org/10.1146/annurev-micro-092611-150044
Haghighi, M. Y., Soltani, J., & Nazeri, S. (2013). A survey on optimization of Agrobacterium-mediated genetic transformation of the fungus Colletotrichum gloeosporioides. Journal of Cell and Molecular Research, 5(1), 35–41. http://jm.um.ac.ir/index.php/biology/article/view/17830
Herbert, C., O’Connell, R., Gaulin, E., Salesses, V., Esquerré-Tugayé, M. T., & Dumas, B. (2004). Production of a cell wall-associated endopolygalacturonase by Colletotrichum lindemuthianum and pectin degradation during bean infection. Fungal Genetics and Biology, 41(2), 140–147. https://doi.org/10.1016/j.fgb.2003.09.008
Kleemann, J., Takahara, H., Stüber, K., & O’Connell, R. (2008). Identification of soluble secreted proteins from appressoria of Colletotrichum higginsianum by analysis of expressed sequence tags. Microbiology, 154(4), 1204–1217. https://doi.org/10.1099/mic.0.2007/014944-0
Korn, M., Schmidpeter, J., Dahl, M., Müller, S., Voll, L. M., & Koch, C. (2015). A genetic screen for pathogenicity genes in the hemibiotrophic fungus Colletotrichum higginsianum identifies the plasma membrane proton pump Pma2 required for host penetration. PLoS ONE, 10(5), 1–35. https://doi.org/10.1371/journal.pone.0125960
Krappmann, S., Sasse, C., & Braus, G. H. (2006). Gene targeting in Aspergillus fumigatus by homologous recombination is facilitated in a nonhomologous end-joining-deficient genetic background. Eukaryotic Cell, 5, 212–215. https://doi.org/10.1128/EC.5.1.212-215.2006
Krappmann, S. (2007). Gene targeting in filamentous fungi: The benefits of impaired repair. Fungal Biology Reviews, 21, 25–29. https://doi.org/10.1016/j.fbr.2007.02.004
Kumar, A., Sharma, P. N., Sharma, O. P., & Tyagi, P. D. (1999). Epidemiology of bean anthracnose under sub-humid mid hills zone of Himachal Pradesh. Indian Phytopathology, 52(4), 393–397.
Lara-Márquez, A., Zavala-Páramo, M. G., Lápez-Romero, E., Calderán-Cortés, N., Lápez-Gámez, R., Conejo-Saucedo, U., & Cano-Camacho, H. (2011). Cloning and characterization of a pectin lyase gene from Colletotrichum lindemuthianum and comparative phylogenetic/structural analyses with genes from phytopathogenic and saprophytic/opportunistic microorganisms. BMC Microbiology, 11(1), 260. https://doi.org/10.1186/1471-2180-11-260
Lu, J., Cao, H., Zhang, L., Huang, P., & Lin, F. (2014). Systematic Analysis of Zn2Cys6 Transcription Factors Required for Development and Pathogenicity by High-Throughput Gene Knockout in the Rice Blast Fungus. PLoS Pathogens, 10, 20–22. https://doi.org/10.1371/journal.ppat.1004432
Maruthachalam, K., Nair, V., Rho, H. S., Choi, J., Kim, S., & Lee, Y. H. (2008). Agrobacterium tumefaciens-mediated transformation in Colletotrichum falcatum and C. acutatum. Journal of Microbiology and Biotechnology, 18(2), 234–241.
Michielse, C. B., Hooykaas, P. J. J., van den Hondel, C. A. M. J. J., & Ram, A. F. J. (2005). Agrobacterium-mediated transformation as a tool for functional genomics in fungi. Current Genetics, 48(1), 1–17. https://doi.org/10.1007/s00294-005-0578-0
Myers, J. R., & Kmiecik, K. (2017). Common Bean: Economic Importance and Relevance to Biological Science Research, 1–20. https://doi.org/10.1007/978-3-319-63526-2_1
Ninomiya, Y., Suzuki, K., Ishii, C., & Inoue, H. (2004). From The Cover: Highly efficient gene replacements in Neurospora strains deficient for nonhomologous end-joining. Proceedings of the National Academy of Sciences of the United States of America, 101, 12248–12253. https://doi.org/10.1073/pnas.0402780101
O’Connell, R. J., Bailey, J. A., & Richmond, D. V. (1985). Cytology and physiology of infection of Phaseolus vulgaris by Colletotrichum lindemuthianum. Physiological Plant Pathology, 27(1), 75–98. https://doi.org/10.1016/0048-4059(85)90058-X
Paccanaro, M. C., Sella, L., Castiglioni, C., Giacomello, F., Martínez-Rocha, A. L., D’Ovidio, R., et al. (2017). Synergistic effect of different plant cell wall-degrading enzymes is important for virulence of Fusarium graminearum. Molecular Plant-Microbe Interactions, 30(11), 886–895. https://doi.org/10.1094/MPMI-07-17-0179-R
Paz, Z., García-Pedrajas, M. D., Andrews, D. L., Klosterman, S. J., Baeza-Montañez, L., & Gold, S. E. (2011). One Step Construction of Agrobacterium-Recombination-ready-plasmids (OSCAR), an efficient and robust tool for ATMT based gene deletion construction in fungi. Fungal Genetics and Biology, 48(7), 677–684. https://doi.org/10.1016/j.fgb.2011.02.003
Pereira, M. F., & de Araújo dos Santos, C. M., de Araújo, E. F., de Queiroz, M. V., & Bazzolli, D. M. S. (2013). Beginning to understand the role of sugar carriers in Colletotrichum lindemuthianum: The function of the gene mfs1. Journal of Microbiology, 51(1), 70–81. https://doi.org/10.1007/s12275-013-2393-5
Perfect, S. E., Hughes, H. B., O’Connell, R. J., & Green, J. R. (1999). Colletotrichum: A Model Genus for Studies on Pathology and Fungal-Plant Interactions. Fungal Genetics and Biology, 27(2–3), 186–198. https://doi.org/10.1006/fgbi.1999.1143
Rogers, L. M., Kim, Y. K., Guo, W., González-Candelas, L., Li, D., & Kolattukudy, P. E. (2000). Requirement for either a host- or pectin-induced pectate lyase for infection of Pisum sativum by Nectria hematococca. Proceedings of the National Academy of Sciences of the United States of America, 97(17), 9813–9818. https://doi.org/10.1073/pnas.160271497
Ruiz, G. B., Di Pietro, A., & Roncero, M. I. G. (2015). Combined action of the major secreted exo- and endopolygalacturonases is required for full virulence of Fusarium oxysporum. Molecular Plant Pathology, 17(3), 339–353. https://doi.org/10.1111/mpp.12283
Sambrook, J., Fritsch, E. F., & Maniatis, T. (1989). Molecular cloning. A laboratory manual (2nd ed.). Cold Spring Harbor Laboratory.
Schmitz, K., Protzko, R., Zhang, L., & Benz, J. P. (2019). Spotlight on fungal pectin utilization—from phytopathogenicity to molecular recognition and industrial applications. Applied Microbiology and Biotechnology, 103(6), 2507–2524. https://doi.org/10.1007/s00253-019-09622-4
Shih, J., Wei, Y., & Goodwin, P. H. (2000). A comparison of the pectate lyase genes, pel-1 and pel-2, of Colletotrichum gloeosporioides f.sp. malvae and the relationship between their expression in culture and during necrotrophic infection. Gene, 243(1–2), 139–150. https://doi.org/10.1016/S0378-1119(99)00546-6
Specht, C. A., DiRusso, C. C., Novotny, C. P., & Ullrich, R. C. (1982). A method for extracting high-molecular-weight deoxyribonucleic acid from fungi. Analytical Biochemistry, 119(1), 158–163. https://doi.org/10.1016/0003-2697(82)90680-7
Talhinhas, P., Muthumeenakshi, S., Neves-Martins, J., Oliveira, H., & Sreenivasaprasad, S. (2008). Agrobacterium-mediated transformation and insertional mutagenesis in Colletotrichum acutatum for investigating varied pathogenicity lifestyles. Molecular Biotechnology, 39(1), 57–67. https://doi.org/10.1007/s12033-007-9028-1
Teixeira, J. A., Gonçalves, D. B., de Queiroz, M. V., & De Araújo, E. F. (2011). Improved pectinase production in Penicillium griseoroseum recombinant strains. Journal of Applied Microbiology, 111(4), 818–825. https://doi.org/10.1111/j.1365-2672.2011.05099.x
Teixeira, J. A., Nogueira, G. B., de Queiroz, M. V., & de Araújo, E. F. (2014). Genome organization and assessment of high copy number and increased expression of pectinolytic genes from Penicillium griseoroseum: A potential heterologous system for protein production. Journal of Industrial Microbiology and Biotechnology, 41(10), 1571–1580. https://doi.org/10.1007/s10295-014-1486-4
Untergasser, A., Nijveen, H., Rao, X., Bisseling, T., Geurts, R., & Leunissen, J. A. M. (2007). Primer3Plus, an enhanced web interface to Primer3. Nucleic Acids Research, 35(SUPPL.2), 71–74. https://doi.org/10.1093/nar/gkm306
Villa-Rivera, M. G., Conejo-Saucedo, U., Lara-Marquez, A., Cano-Camacho, H., Lopez-Romero, E., & Zavala-Paramo, M. G. (2016). The Role of Virulence Factors in the Pathogenicity of Colletotrichum sp. Current Protein & Peptide Science, 18(10), 1005–1018. https://doi.org/10.2174/1389203717666160813160727
Walton, J. D. (1994). Deconstructing the cell wall. Plant Physiology, 104(4), 1113–1118.
Wise, A. A., Liu, Z., & Binns, A. N. (2006). Three methods for the introduction of foreign DNA into Agrobacterium. Methods in molecular biology (Clifton, N.J.), 343, 43–53. https://doi.org/10.1385/1-59745-130-4:43
Xu, C., Zhang, X., Qian, Y., Chen, X., Liu, R., Zeng, G., et al., (2014). A high-throughput gene disruption methodology for the entomopathogenic fungus Metarhizium robertsii. PLoS ONE, 9(9). https://doi.org/10.1371/journal.pone.0107657
Yarullina, L. G., Akhatova, A. R., & Kasimova, R. I. (2016). Hydrolytic enzymes and their proteinaceous inhibitors in regulation of plant–pathogen interactions. Russian Journal of Plant Physiology, 63(2), 193–203. https://doi.org/10.1134/S1021443716020151
Yu, X., Pan, J., Wei, D., & Zhu, X. (2015). High frequency of homologous gene disruption by single-stranded DNA in the taxol-producing fungus Pestalotiopsis microspora. Annals of Microbiology, 65(4), 2151–2160.
Acknowledgements
We are grateful to support by the following Brazilian agencies: the Minas Gerais Science Foundation (FAPEMIG– Fundação de Amparo à Pesquisa do Estado de Minas Gerais), the Brazilian Federal Agency of Support and Evaluation of Postgraduate Education – Finance code 001 (CAPES–Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) and the National Council of Scientific and Technological Development (CNPq– Conselho Nacional de Desenvolvimento Científico e Tecnológico).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest/competing interests
The authors declare that they have no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
All authors contributed to the study conception and design. All authors read and approved the final manuscript.
Consent for publication
All authors gave consent to submit for publication.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
da Silva, L.L., Rosa, R.O., Garcia, E.A. et al. Use of Agrobacterium tumefaciens-mediated transformation for the inactivation of pectinase genes in Colletotrichum lindemuthianum. Eur J Plant Pathol 162, 595–608 (2022). https://doi.org/10.1007/s10658-021-02423-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-021-02423-4