Abstract
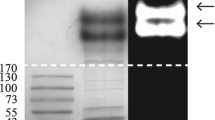
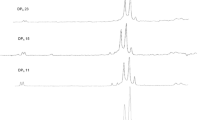
Detection of chitinase activity after electrophoresis in native polyacrylamide gels containing glycol chitin showed that Streptomyces macrosporeus VTCC 940003 produced several chitinases. Chito-oligosaccharides (COS) were produced from commercial chitosan using these chitinases. The degree of polymerization (DP) of the produced COS was confirmed by Matrix Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry (MALDI-TOF MS). Moreover, the biological activities of the COS against Botrytis cinerea in vitro and in vivo were assessed. The COS preparation contained a mixture of oligomers with DP ranging from 2 to 10. The COS also showed inhibition activities on spore germination, mycelial growth and infection of B. cinerea on tomato leaves.






Similar content being viewed by others
References
Badawy, M.E.I., & Rabea, E.I. (2017). Chitosan and its modifications as biologically active compounds in different applications. In M. Masuell, D. Renard (Eds.), Advances in physicochemical properties of biopolymers (pp. 1–108). Bentham e-Books. Bentham Science Publishers, Sharjah, ISBN: 978–1–68,108-545-6.
Cabrera, J. C., & Cutsem, P. V. (2005). Preparation of chitooligosaccharides with degree of polymerization higher than 6 by acid or enzymatic degradation of chitosan. Biochemical Engineering Journal, 25, 165–172.
Das, S. N., Madhuprakash, J., Sarma, P., Purushotham, P., Suma, K., Manjeet, K., & Podile, A. R. (2013). Biotechnological approaches for field applications of chitooligosaccharides (COS) to induce innate immunity in plants. Critical Reviews in Biotechnology, 35(1), 29–43.
Ghaouth, A. E., Asselin, J. A., & Benhamou, N. (1992). Antifungal activity of chitosan on post-harvest pathogens: induction of morphological and cytological alterations in Rhizopus stolonifer. Mycological Research, 96(9), 769–779.
Karpova, N., Shagdarova, B., Lunkov, A., Il’ina, A., & Varlamov, V. (2021). Antifungal action of chitosan in combination with fungicides in vitro and chitosan conjugate with gallic acid on tomatoes against Botrytis cinerea. Biotechnology Letters. https://doi.org/10.1007/s10529-021-03138-6.
Kawase, T., Yokokawa, S., Saito, A., Fujii, T., Nikaidou, N., Miyashita, K., & Watanabe, T. (2006). Comparison of enzymatic and antifungal properties between family 18 and 19 chitinase from S. coelicolor A3(2). Bioscience, Biotechnology, and Biochemistry, 70, 988–998.
Kim, S., & Rajapakse, N. (2005). Enzymatic production and biological activities of chitosan oligosaccharides (COS): a review. Carbohydrate Polymymers, 62, 357–368.
Kim, J. K., Yang, J. Y., & Kim, G. J. (2003). Purification and characterization of chitinase from Streptomyces sp. M-20. Journal of Biochemistry and Molecular Biology, 36(2), 185–189.
Koo, C. J., Lee, Y. S., Chun, J. H., Cheong, H. Y., Choi, S. J., Kawabata, S., Miyagi, M., Tsunasawa, S., Ha, S. K., Bae, W. D., Han, C., Lee, L. B., & Cho, J. M. (1998). Two hevein homologs isolated from the seed of Pharbitis nil L. exhibit potent antifungal activity. Biochimica et Biophysica Acta, 1382, 80–90.
Liaqat, F., & Eltem, R. (2018). Chitooligosaccharides and their biological activities: A comprehensive. Review. Carbohydrate Polymers, 184, 243–259.
Mallakuntla, K. M., Vaikuntapu, R. P., Bhuvanachandra, N. S. B., & Podile, R. A. (2017). Transglycosylation by a chitinase from Enterobacter cloacae subsp. cloacae generates longer chitin oligosaccharides. Scientific Reports, 7, 5113. https://doi.org/10.1038/s41598-017-05140-3.
Meng, X., Yang, L., Kennedy, J. F., & Tian, S. (2010). Effects of chitosan oligosaccharides on growth of two fungal pathogens and physiological properties in pear fruit. Carbohydrate Polymymers, 81, 70–75.
Miller, G. L. (1959). Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chemistry, 31(3), 426–428.
Montillaa, A., Ruiz-Matutea, A. I., Corzoa, N., Giacominib, C., & Irazoquib, G. (2013). Enzymatic generation of chitooligosaccharides from chitosan using soluble and immobilized glycosyltransferase (Branchzyme®). Journal of Agriculture and Food Chemistry, 61(43), 10,360–10,367.
Moon, C., Seo, J. D., Song, S. Y., Hong, H. S., Choi, H. S., & Jung, J. V. (2017). Antifungal activity and patterns of N-acetyl-chitooligosaccharide degradation via chitinase produced from Serratia marcescens PRNK-1. Microbiol Pathogenesis, 113, 218–224.
Olicón-Hernández, R. D., Vázquez-Landaverde, A. P., Cruz-Camarillo, R., Rojas-Avelizapa, I. L., Tomas, S., & Hidalgo, M. D. (2016). Comparison of chitooligosaccharide production from three different colloidal chitosans using the endochitonsanolytic system of Bacillus thuringiensis. Preparative Biochemistry & Biotechnology, 47, 116–122. https://doi.org/10.1080/10826068.2016.1181086.
Rahman, M. H., Shovan, L. R., Hjeljord, L. G., Aam, B. B., Eijsink, V. G. H., & S⌀rlie, M., Tronsmo, A. (2014). Inhibition of fungal plant pathogens by synergistic action of chito-oligosaccharides and commercially available fungicides. PloS ONE, 9(4), e93192. https://doi.org/10.1371/journal.pone.0093192.
Rahman, M. H., Hjeljord, L. G., Aam, B. B., & S⌀rlie, M., Tronsmo, A. (2015). Antifungal effect of chito-oligosaccharides with different degrees of polymerization. European Journal of Plant Patholology, 141, 147–158.
Singh, V., Haque, S., Singh, H., Verma, Vibha, J. K., Singh, R., Jawed, A., & Tripath, C. K. M. (2016). Isolation, screening, and identification of novel isolates of actinomycetes from India for antimicrobial applications. Frontiers in Microbiology, 7, 1921. https://doi.org/10.3389/fmicb.2016.01921.
SØrbotten, A., Horn, S. J., Eijsink, V. G. H., & Vårum, K. M. (2005). Degradation of chitosans with chitinase B from Serratia marcescens. FEBS Journal, 272, 538–549.
Sun, G., Yang, Q., Zhang, A., Guo, J., Liu, X., Wang, Y., & Ma, Q. (2018). Synergistic effect of the combined bio-fungicides ε-poly-L-lysine and chitooligosaccharide in controlling grey mould (Botrytis cinerea) in tomatoes. International Journal of Food Microbiology, 276, 46–53.
Trudel, J., & Asselin, A. (1989). Detection of chitinase activity after polyacrylamide gel electrophoresis. Analytical Biochemistry, 178(2), 362–636.
Van Aubel, G., Cambier, P., Dieu, M., & Van Cutsem, P. (2016). Plant immunity induced by COS-OGA elicitor is a cumulative process that involves salicylic acid. Plant Science, 247, 60–70.
Williamson, B., Tudzynski, B., Tudzynski, L. P., & Vankan, L. A. J. (2007). Botrytis cinerea: the cause of grey mould disease. Molecular Plant Pathology, 8, 561–580.
Zhang, H., Du, Y., Yu, X., Mitsutomi, M., & Aiba, S. (1999). Preparation of chito-oligosaccharides from chitosan by a complex enzyme. Carbohydrate Research, 320, 257–260.
Acknowledgements
The authors wish to acknowledge the financial support of the grant “Scale-up production of chitosan oligomer from shrimp shells waste to be used in plant protection”, code QG.16.87 of Vietnam National University, Hanoi. The support from Walloniae-Brussels International through the grant “Industrialisation de la production d’un biopesticide pour la protection de cultures de légumes au Vietnam” (Project 13, period 2016–2018) was also greatly appreciated.
Data availability
Not applicable.
Code availability
Not applicable.
Funding
the financial support: i) of the grant “Scale-up production of chitosan oligomer from shrimp shells waste to be used in plant protection”, code QG.16.87 of Vietnam National University, Hanoi; ii) Walloniae-Brussels International through the grant „ Industrialisation de la production d’un biopesticide pour la protection de cultures de légumes au Vietnam“(Project 13, period 2016–2018).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no conflict of interest, financial or otherwise.
Rights and permissions
About this article
Cite this article
Anh, T.T.V., Uyen, N.Q., Hop, D.V. et al. Chito-oligosaccharide production by chitinase of Streptococcus macrosporeus VTCC 940003 and their inhibition activities on Botrytis cinerea. Eur J Plant Pathol 161, 185–193 (2021). https://doi.org/10.1007/s10658-021-02313-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-021-02313-9