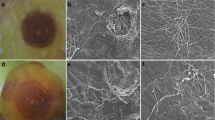
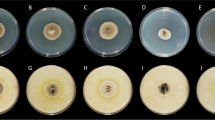
Abstract
Colletotrichum spp. infections are an increasing threat to the fruit production industry worldwide, as a postharvest disease. Gaining knowledge on the susceptibility of apple fruit cultivars to Colletotrichum species is essential for specific breeding programs aiming to decrease the disease susceptibility of commercial apple cultivars. We investigated fruit susceptibility of nine apple cultivars at harvest and after storage, and studied the relationship of the susceptibility with fruit ripening characteristics. An accurate artificial inoculation technique, by wounding and injecting the spore suspension, was used to consistently follow-up the relative susceptibility symptoms of fruits. The nine apple cultivars studied, cvs. Pinova, Nicoter, Nicogreen, Golden Delicious, Gala, Elstar, Jonathan, Idared and Topaz, showed clear differences in susceptibility to C. fioriniae, main species causing problems in Europe, based on lesion expansion growth rate data. Cultivar Nicoter and Nicogreen were evaluated as less susceptible cultivars, while cv. Golden Delicious and cv. Pinova were classified as high susceptible cultivars. After three months of storage all fruit cultivars showed a higher susceptibility than at harvest. We further explored the link between fruit ripening characteristics and postharvest pathogen developments. Results showed significant higher Brix, ethylene production and respiration and significantly lower firmness and titratable acids after storage, with the extent of these changes being cultivar dependent. The ripening parameters respiration and Brix had a positive and the firmness a negative correlation with the lesion expansion growth rate of C. fioriniae after artificial wound inoculations. These experiments help to give a better insight in the susceptibility of different cultivars. Moreover, our data support the hypothesis that the extent of the necrotrophic fungal development depends on the fruit ripening stage.








Similar content being viewed by others
References
Agrios, G. N. (2005). Plant pathology (5th ed. ed.). Amsterdam: Elsevier.
Alaniz, S., Hernandez, L., & Mondino, P. (2015). Colletotrichum fructicola is the dominant and one of the most aggressive species causing bitter rot of apple in Uruguay. Tropical Plant Pathology, 40, 265–274.
Alkan, N., Fortes, A.M. (2015). Insights into molecular and metabolic events associated with fruit response to post-harvest fungal pathogens. Frontiers in Plant Science, 6.
Baghat, R. G., Mehta, B. P., Patil, V. A., & Sharma, H. (2015). Screening of cultivars/varieties against mango anthracnose caused by Colletotrichum gloeosporioides. International Journal of Environmental and Agriculture research, 1, 21–23.
Barkai-Golan, R. (2001). Postharvest diseases of fruits and vegetables- development and control (p. 418). Amsterdam: Elsevier Science.
Baroncelli, R., Sreenivasaprasad, S., Thon, M. R., & Sukno, S. A. (2014). First report of apple bitter rot caused by Colletotrichum godetiae in the United Kingdom. Plant Disease, 98, 1000.
Baroncelli, R., Zapparata, A., Sarrocco, S., Sukno, S. A., Lane, C.R., Thon, M.R., Vannacci, G., Holub, E., Sreenivasaprasad, S. (2015). Molecular diversity of anthracnose pathogen populations associated with UK strawberry production suggests multiple introductions of three different Colletotrichum species. PLOS one. Available on: https://doi.org/10.1371/journal.pone.0129140, [13/07/2018].
Biggs, A. R., & Miller, S. S. (2001). Relative susceptibility of selected apple cultivars to Colletotrichum acutatum. Plant Disease, 85, 657–660.
Børve, J., & Stensvand, A. (2015). Colletotrichum acutatum on apple in Norway. IOBC-WPRS Bulletin, 110, 151–157.
Bulens, I., Van de Poel, B., Hertog, M., De Proft, M. P., Geeraerd, A. H., & Nicolaï, B. M. (2011). Protocol: An updated integrated methodology for analysis of metabolites and enzyme activities of ethylene biosynthesis. Plant Methods, 7, 17.
Cannon, P. F., Damm, U., Johnston, P. R., & Weir, B. S. (2012). Colletotrichum – Current status and future directions. Studies in Mycology, 73, 181–213.
Celetti, M. (2015). Bitter rot symptoms and management in apples during 2015. Geraadpleegd op 22 december 2018 via http://www.omafra.gov.on.ca/english/crops/hort/news/hortmatt/2015/14hrt15a1.html.
Crusius, L. U., Forcelini, C. A., Sanhueza, R. M. V., & Fernandes, J. M. C. (2002). Epidemiology of apple leaf spot. Fitopatologia Brasileira, 27, 65–70.
Damm, U., Cannon, P. F., Woudenberg, J. H., & Crous, P. W. (2012). The Colletotrichum acutatum species complex. Studies in Mycology, 73, 37–113.
Dean, R., Van Kan, J. A. L., Pretorius, Z. A., Hammond-Kosack, K. E., & Di Pietro, A. (2012). The top 10 fungal pathogens in molecular plant pathology. Molecular Plant Pathology, 13, 414–430.
DeLong, J. M., Prange, R. K., & Harrison, P. A. (1999). Using the Streif index as a final harvest window for controlled-atmosphere storage of apples. Horticultural Science, 13, 1251–1255.
Demeyer R., De Baets T., de Schaetzen C., D’hooghe J., Keulemans W., Marchand F., Wustenberghs H. (2013). Duurzame fruitbedrijven: bepalende factoren – indicatoren – hot issues, Vlaamse overheid, Departement Landbouw- en Visserij, afdeling Monitoring en Studie, Brussel.
Dinh, S. Q., Chongwungse, J., Pongam, P., & Sangchote, S. (2003). Fruit infection by Colletotrichum gloeosporioides and anthracnose resistance of some mango cultivars in Thailand. Australian Plant Pathology, 32, 533–538.
Freeman, S., Katan, T., & Shabi, E. (1998). Characterization of Colletotrichum species responsible for anthracnose diseases of various fruits. Plant Disease, 82, 596–605.
Gonzales, E., Sutton, T. B., & Correll, J. C. (2006). Clarification of the etiology of Glomerella leaf spot and bitter rot of apple caused by Colletotrichum spp. based on morphology and genetic, molecular, and pathogenicity tests. Phytopathology, 96, 982–992.
Gooday. (1995). Cell walls. In N. A. R. Gow & G. M. Gadd (Eds.), The growing fungus. London: Chapman and Hall.
Grammen, A., Wenneker, M., Van Campenhout, J., Pham, K. T. K., Van Hemelrijck, W., Bylemans, D., Geeraerd, A., & Keulemans, W. (2019). Identification and pathogenicity assessment of Colletotrichum isolates causing bitter rot of apple fruit in Belgium. European Journal of Plant Pathology, 153, 47–63.
Gwanpua, S. G., Van Buggenhout, S., Verlinden, B. E., Christiaens, S., Shpigelman, A., Vicent, V., Kermani, Z. J., Nicolai, B. M., Hendrickx, M., & Geeraerd, A. (2014). Pectin modifications and the role of pectin-degrading enymes during postharvest softening of Jonagold apples. Food Chemestry, 158, 283–291.
Ivic, D., Voncina, D., Sever, Z., Simon, S., & Pejic, I. (2013). Identification of Colletotrichum species causing bitter rot of apple and pear in Croatia. Journal of Phytopathology, 161, 284–286.
Jackson, J. E. (2003). Eating quality and its retention. In J. E. Jackson (Ed.), Biology of apples and pears (pp. 341–383). Cambridge: Cambridge University Press.
Jeffries, P., Dodd, J. C., Jeger, M. J., & Plumbey, R. A. (1990). The biology and control of Colletotrichum species on tropical fruit crops. Plant Pathology, 39, 343–366.
Johnston, J. W., Hewett, E. W., & Hertog, M. (2002). Postharvest softening of apple (Malus x domestica) fruit: A review. New Zealand Journal of Crop and Horticultural Science, 30, 145–160.
Köhl, J., Wenneker, M., Groenenboom-de-haas, B. H., Anbergen, R., Goossen-van de Geijn, H. M., Lombaers-van der Plas, C. H., Pinto, F. A. M. F., & Kastelein, P. (2018). Dynamics of post-harvest pathogens Neofabraea spp. and Cadophora spp. in plant residues in Dutch apple and pear orchards. Plant Pathology, 67, 1264–1277.
Kou, L., Gaskins, V. L., Luo, Y., & Jurick, W. M., II. (2013). First report of Colletotrichum fioriniae causing postharvest decay on ‘Nittany’ apple fruit in the United States. Plant Disease, 98, 993.
Moral, J., Xaviér, C. J., Viruega, J. R., Roca, L. F., Caballero, J., & Trapero, A. (2017). Frontiers in Plant Science, 8, 1892.
Munda, A. (2014). First report of Colletotrichum fioriniae and C. godetiae causing apple rot in Slovenia. Plant Disease, 98, 1892.
Munir, M., Amsden, B., Dixon, E., Vaillancourt, L., & Ward Gauthier, N. A. (2016). Characterization of Colletotrichum species causing bitter rot of apple in Kentucky orchards. Plant Disease, (11), 2194–2203.
Nath, P., Trivedi, P. K., Sane, V. A., & Sane, A. P. (2006). Role of ethylene in fruit ripening. In N. A. Khan (Ed.), Ethylene action in plants (pp. 151–184). Heidelberg: Springer.
Neuwald, D. A., & Kittemann, D. (2016). The incidence of spp. in 'Pinova' apples can be reduced at elevated storage temperatures. Acta Hortic. 1144, 231-236. https://doi.org/10.17660/ActaHortic.2016.1144.34
Nodet, P., Baroncelli, R., Faugère, D., & Le Floch, G. (2016). First report of apple bitter rot caused by Colletotrichum fioriniae in Brittany, France. Plant Disease, 100, 1497.
Nodet, P., Chalopin, M., Crété, X., Baroncelli, R., Le Floch, G. 2019. First report of Colletotrichum fructicola causing apple bitter rot in Europe. American Phytopathological Society https://doi.org/10.1094/PDIS-11-18-1915-PDN.
Ozkaya, O., & Dundar, O. (2009). Influence of 1-methylcyclopropene (1-MCP) on 'Fuiji' apple quality during long-term storage. Journal of Food Agriculture and Environment, 7, 146–148.
Paniagua, C., Posé, S., Morris, V.J., Kirby, A.R., Quesada, M.A., Mercado, J.A. 2014. Fruit softening and pectin disassembly: an overview of nanostructural pectin modifications assessed by atomuc force microscopy. 114, 1375–1383.
Phoulivong, S., McKenzi, E. H. C., & Hyde, K. D. (2012). Cross infection of Colletotrichum species; a case study with tropical fruits. Current research in environmental and applied mycology, 2, 99–111.
Polashock, J. J., Ehlenfeldt, M. K., Stretch, A. W., & Kramer, M. (2005). Anthracnose fruit rot resistance in blueberry cultivars. Plant Disease, 89, 33–38.
Prusky, D. (1996). Pathogen quiescence in postharvest diseases. Annual Review of Phytopathology, 34, 413–434.
Prusky, D., & Lichter, A. (2007). Activation of quiescent infectionsby postharvest pathogens during transition from biotrophic to the necrotrophic stage. FEMS Microbiology Letters, 268, 1–8.
Prusky, D., Alkan, N., Mengiste, T., & Fluhr, R. (2013). Quiescent and necrotrophic lifestyle choice during postharvest disease development. Annual Review of Phytopathology, 51, 55–76.
Saquet, A. (2017). Storability of "Jonagold" apple fruit under extreme controlled atmosphere conditions. Journal of Agricultural Science and Technology B, 6, 262–268.
SAS Institute Inc. (2013). Using JMP 11. Cary: SAS Institute Inc.
Shangguan, I., Song, C., Leng, X., Kayesh, E., Sun, X., & Fang, J. (2014). Mining and comparison of the genes encoding the key enzymes involved in sugar biosynthesis in apple, grape and sweet orange. Scientia Horticulturae, 165, 311–318.
Storch, T. T., Finatto, T., Bruneau, M., Orsel-Baldwin, M., Renou, J. P., Rombaldi, C. V., & Girardi, C. L. (2017). Contrasting transcriptional programs control postharvest development of apples (Malus x domestica Borkh.) submitted to cold storage and ethylene blockage. Journal of Agricultural and Food Chemistry, 65, 7813–7826.
Temiyakul, P., Taylor, P. W. J., & Mongkolporn, O. (2010). Development of a double inoculation method to assess resistance to anthracnose in trispecies Capsicum hybrid. Journal of Phytopathology, 158, 561–565.
Turechek, W. W. (2004). Apple diseases and their management. In S. A. M. H. Naqvi (Ed.), Diseases of fruits and vegetables (pp. 1–108). Dordrecht: Kluwer Academic Publishers.
Velho, A. C., Alaniz, S., Casanova, L., Mondino, P., & Stadnik, M. J. (2015). New insights into the characterization of Colletotrichum species associated with apple diseases in southern Brazil and Uruguay. Fungal Biology, 119, 229–244.
Vendrell, M., Dominguez-Puigjaner, E., & Llop-Tous, I. (2001). Climacteric versus non-climacteric physiology. Acta Horticulturae, (553), 345–349.
Wakasa, Y., Kudo, H., Ishikawa, R., Akada, S., Senda, M., Niizeki, M., & Harada, T. (2006). Low expression of an endopolygalacturonase gene in apple fruit with longterm storage potential. Postharvest Biology and Technology, 39, 193–198.
Wenneker, M., Pham, K., Lemmers, M., de Boer, A., van der Lans, A., van Leeuwen, P., & Hollinger, T. (2016). First reports of Colletotrichum godetiae causing bitter rot on ‘Golden delicious’ apples in the Netherlands. Plant Disease, 100, 218.
Wharton, P. S., & Diéguez-Uribeondo, J. (2004). The biology of Colletotrichum acutatum. Anales Del Jardin Bontanico de Madrid, 61, 3–22.
Yahia, E. M. (2011). Postharvest biology and technology of tropical and subtropical fruits. 1: Fundamental issues. Woodhead: Cambridge.
Zhu, Z., Liu, R., Li, B., & Tian, S. (2013). Characterisation of genes encoding key enzymes involved in sugar metabolism of apple fruit in controlled atmosphere storage. Food Chemistry, 141, 3323–3328.
Zucolata, M., Ku, K.M., Kim, M.J., Kushad, M. (2017). Influence of 1-methylcyclopropene treatment on postharvest quality of four scab (Venturia inequalis) resistant apple cultivars. Journal of Food Quality, 1–12.
Acknowledgments
The authors thank the Fund for Scientific Research (FWO) Flanders for providing funding for this research (grant number 1S44116N).
Funding
This study was funded by FWO (research foundation Flanders) Grant number: 1S44116N.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Supplementary figure 1
Average number of lesions per fruit counted in time (days post inoculation, abbrev. DPI) after nebulizing intact fruits of nine different apple cultivars (Elstar, Gala, Golden Delicious, Idared, Jonathan, Nicogreen, Nicoter, Pinova and Topaz) with Colletotrichum fioriniae at harvest and after three months of storage at 2 °C ambient air in year 2. (PNG 1316 kb)
Supplementary figure 2
Average results (and standard deviation) of Brix (in %), firmness (in kg/cm2), titrable acids (in mg/mL) and LEGR of C. fioriniae (in mm/day) of nine apple cultivars (Elstar, Gala, Golden Delicious, Idared, Jonathan, Nicogreen, Nicoter, Pinova and Topaz) measured at harvest and after three months of storage in year 1. (JPG 1855 kb)
Supplementary figure 3
Average results of ethylene production (in nmol/kg.s) and LEGR of C. fioriniae (in mm/day) of nine apple cultivars (Elstar, Gala, Golden Delicious, Idared, Jonathan, Nicogreen, Nicoter, Pinova and Topaz) measured at harvest and after three months of storage in year 1. (JPG 1850 kb)
Supplementary figure 4
Average results of CO2 production (in nmol/kg.s), O2 consumption (in nmol/kg.s) and LEGR of C. fioriniae (in mm/day) of nine apple cultivars (Elstar, Gala, Golden Delicious, Idared, Jonathan, Nicogreen, Nicoter, Pinova and Topaz) measured at harvest and after three months of storage in year 1. (JPG 1806 kb)
Supplementary figure 5
Average results of Brix (in %), firmness (in kg/cm2), titrable acids (in mg/mL) and LEGR of C. fioriniae (in mm/day) of nine apple cultivars (Elstar, Gala, Golden Delicious, Idared, Jonathan, Nicogreen, Nicoter, Pinova and Topaz) measured at harvest and after three months of storage in year 2. (JPG 1887 kb)
Supplementary figure 6
Average results of ethylene production (in nmol/kg.s) and LEGR of C. fioriniae (in mm/day) of nine apple cultivars (Elstar, Gala, Golden Delicious, Idared, Jonathan, Nicogreen, Nicoter, Pinova and Topaz) measured at harvest and after three months of storage in year 2. (JPG 1670 kb)
Supplementary figure 7
Average results of CO2 production (in nmol/kg.s), O2 consumption (in nmol/kg.s) and LEGR of C. fioriniae (in mm/day) of nine apple cultivars (Elstar, Gala, Golden Delicious, Idared, Jonathan, Nicogreen, Nicoter, Pinova and Topaz) measured at harvest and after three months of storage in year 2. (JPG 1652 kb)
Rights and permissions
About this article
Cite this article
Grammen, A., Van Campenhout, J., Geeraerd, A. et al. Susceptibility of apple fruits (Malus x domestica Borkh.) to the postharvest pathogen Colletotrichum fioriniae: cultivar differences and correlation with fruit ripening characteristics. Eur J Plant Pathol 155, 801–816 (2019). https://doi.org/10.1007/s10658-019-01812-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-019-01812-0