Abstract
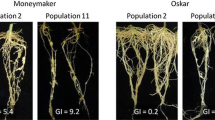
The response to Meloidogyne incognita and M. javanica of 29 cucurbitaceous genotypes belonging to the genera Cucurbita, Lagenaria and Luffa was evaluated to determine their relative host suitability and utility as rotational crops in sustainable agriculture. The pathogenic potential, parasitic success and host efficiency were estimated based on the ability of the nematode to form galls and generate egg masses on selected genotypes. Meloidogyne incognita showed higher pathogenic potential (gall formation) than M. javanica across all genotypes except for C. argyrosperma. However, M. javanica had higher parasitic success (egg mass production) than M. incognita on the Cucurbita genotypes. By contrast, on Lagenaria and Luffa, M. incognita had higher pathogenic potential and parasitic success than M. javanica. All genotypes except Luffa spp., showed higher host efficiency (rate of egg masses per gall) for M. javanica than for M. incognita. The genotypes of Cucurbita pepo ssp. pepo will be useful for M. incognita management because they reduce population build-up, but root damage would be more severe due to abundant root galling. The Cucurbita genotypes would be more tolerant to higher Pi when infected by M. javanica as they suffer less root damage, but the residual populations may affect the subsequent crop in the rotation. Lagenaria siceraria showed a good level of resistance to M. javanica and will be useful by itself or for grafting other cucurbitaceous crops. The species of Luffa were poor hosts of M. javanica and can be used also for grafting.

Similar content being viewed by others
References
Anwar, S. A., & McKenry, M. V. (2010). Incidence and reproduction of Meloidogyne incognita on vegetable crop genotypes. Pakistan Journal of Zoology, 42, 135–141.
Brito, J. A., Stanley, J. D., Mendes, M. L., Cetintas, R., & Dickson, D. W. (2007). Host status of selected cultivated plants to Meloidogyne mayaguensis in Florida. Nematropica, 37, 65–71.
Carneiro, R. M. D. G., Randig, O., Almeida, M., & Diniz Campos, A. (2000). Resistance of vegetable crops to Meloidogyne spp.: Suggestion for a crop rotation system. Nematologia Brasileira, 24, 49–54.
Carneiro, R. M. D. G., Ritta, M., Almeida, A., & Gomes, A. C. M. M. (2004). First record of Meloidogyne hispanica Hirschmann, 1986 on squash in state of Bahia, Brazil. Nematologia Brasileira, 28, 215–218.
Chandra, P., Sao, R., Gautam, S. K., & Poddar, A. N. (2010). Initial population density and its effect on the pathogenic potential and population growth of the root-knot nematode Meloidogyne incognita in four species of cucurbits. Asian Journal of Plant Pathology, 4, 1–15.
Davies, A. R., Perkins-Veazie, P., Sakata, Y., López-Galarza, S., Maroto, J. V., Lee, S. G., et al. (2008). Cucurbit grafting. Critical Reviews in Plant Sciences, 27, 50–74.
Djian-Caporalino, C., Molinari, S., Palloix, A., Ciancio, A., Fazari, A., Marteu, N., Ris, N., & Castagnone-Sereno, P. (2011). The reproductive potential of the root-knot nematode Meloidogyne incognita is affected by selection for virulence against major resistance genes from tomato and pepper. European Journal of Plant Pathology, 131, 431–440.
Edelstein, M., Oka, Y., Burguer, Y., Eizenberg, H., & Cohen, R. (2010). Variation in the response of cucurbits to Meloidogyne incognita and M. javanica. Israel Journal of Plant Sciences, 58, 77–84.
Fourie, H., McDonald, A. H., Mothata, T. S., Ntidi, K. N., & De Waele, D. (2012). Indications of variation in host suitability to root-knot nematode populations in commercial tomato varieties. African Journal of Agricultural Research, 7, 2344–2352.
Hallmann, J., & Kiewnick, S. (2018). Virulence of Meloidogyne incognita populations and Meloidogyne enterolobii on resistant cucurbitaceous and solanaceous plant genotypes. Journal of Plant Diseases and Protection, 125, 415–424. https://doi.org/10.1007/s41348-018-0165-5.
Holliday, P. (2001). A dictionary of plant pathology (2nd ed.). Cambridge, UK: Cambridge University Press.
Hussey, R. S., & Barker, K. R. (1973). A comparison of methods of collecting inocula of Meloidogyne spp. including a new technique. Plant Disease Reporter, 57, 1025–1028.
Johnson, A. W., & Leonard, R. A. (1995). Effects and carry-over benefits of nematicides in soil planted to a sweet corn-squash-vetch cropping system. Journal of Nematology, 27, 563–570.
Kokalis-Burelle, N., & Nyczepir, A. P. (2004). Host range studies for Meloidogyne floridensis. Journal of Nematology, 36, 328.
Levi, A., Thies, J., Ling, K., Simmons, A. M., Kousik, C., & Hasell, R. (2009). Genetic diversity among Lagenaria siceraria accessions containing resistance to root-knot nematodes, whiteflies, ZYMV or powdery mildew. Plant Genetic Resources, 7, 216–226.
López-Gómez, M., Flor-Peregrín, E., Talavera, M., & Verdejo-Lucas, S. (2015a). Suitability of zucchini and cucumber genotypes to populations of Meloidogyne arenaria, M. incognita and M. javanica. Journal of Nematology, 47, 79–85.
López-Gómez, M., Flor-Peregrín, E., Talavera, M., Sorribas, F. J., & Verdejo-Lucas, S. (2015b). Population dynamics of Meloidogyne javanica and relationship with leaf chlorophyll content on zucchini. Crop Protection, 70, 8–14.
López-Gómez, M., Talavera, M., & Verdejo-Lucas, S. (2016). Differential reproduction of Meloidogyne incognita and M. javanica in watermelon cultivars and cucurbit rootstocks. Plant Pathology, 65, 145–153.
Maleita, C., Curtis, R., Powers, S., & Abrantes, I. (2012). Host status of cultivated plants to Meloidogyne hispanica. European Journal of Plant Pathology, 133, 449–460.
Mukhtar, T., Kayani, M. Z., & Hussain, M. A. (2013). Response of selected cucumber cultivars to Meloidogyne incognita. Crop Protection, 44, 13–17.
Ojo, D. (2016). Cucurbits: Importance, botany, uses, cultivation, nutrition, genetic resources, diseases, and pests. In M. Pessarakli (Ed.), Handbook of Cucurbits: Growth, Cultural Practices and Physiology (pp. 23–68). Boca Raton Fl, USA: CRC Press.
Omwega, C., Thomason, I. J., & Roberts, P. A. (1988). A non-destructive technique for screening bean germplasm for resistance to Meloidogyne incognita. Plant Disease, 72, 970–972.
Sigüenza, C., Schochow, M., Turini, T., & Ploeg, A. (2005). Use of Cucumis metuliferus as a rootstock for melon to manage Meloidogyne incognita. Journal of Nematology, 37, 276–280.
Sikora, R. A., & Fernandez, E. (2005). Nematode parasites of vegetables. In M. Luc, R. A. Sikora, & J. Bridge (Eds.), Plant parasitic nematodes in subtropical and tropical agriculture (pp. 319–392). Wallingford UK: CAB International.
de Souza Galatti, F., Junqueira Franco, A., Akemi Ito, L., de Oliveira Charlo, H., Aparecido Gaion, L., & Trevisan Braz, L. (2013). Rootstocks resistant to Meloidogyne incognita and compatibility of grafting in net melon. Revista Ceres, 60, 432–436.
Talavera, M., Sayadi, S., Chirosa-Ríos, M., Salmerón, T., Flor-Peregrín, E., & Verdejo-Lucas, S. (2012). Perception of the impact of root-knot nematode induced diseases in horticultural protected crops of South-Eastern Spain. Nematology, 14, 517–527.
Talavera-Rubia, M., Pérez de Luque, A., López-Gómez, M., & Verdejo-Lucas, S. (2018a). Differential feeding site development and reproductive fitness of Meloidogyne incognita and M. javanica on zucchini, a source of resistance to M. incognita. Nematology, 20, 187–199.
Talavera-Rubia, M., Fernández-Plaza, M., Verdejo-Lucas, S., & Vela, M. D. (2018b). Susceptibilidad y tolerancia del calabacín (Cucurbita pepo) a Meloidogyne incognita y M. javanica. Phytoma España, 295, 42–46.
Tamilselvi, N. A., Pugalendhi, L., & Sivakumar, M. (2017). Response of cucurbitaceous rootstocks and bitter gourd scions to root knot nematode Meloidogyne incognita Kofoid and white. International Journal of Horticulture and Floriculture, 5, 247–251.
Thies, J. A., Ariss, J. J., Hassell, R. L., Olson, S., Kousik, C. S., & Levi, A. (2010). Grafting for management of southern root-knot nematode, Meloidogyne incognita in watermelon. Plant Disease, 94, 1195–1199.
Vela, M. D., Giné, A., López-Gómez, M., Sorribas, F. J., Ornat, C., Verdejo-Lucas, S., & Talavera, M. (2014). Thermal time requirements of root-knot nematodes on zucchini-squash and population dynamics with associated yield losses on spring and autumn cropping cycles. European Journal of Plant Pathology, 140, 481–490.
Zeck, W. M. (1971). A rating scheme for field evaluation of root-knot nematode infestations. Pflanzenschtz-Nachrichten, 24, 141–144.
Funding
This study was funded by INIA (grant number RTA2014–00078), IFAPA (grant number PP.TRA.TRA201600.9) and FEDER from the European Union.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The Banco de Germoplasma de Valencia is acknowledged for providing the accessions used in this study. Thanks are given to, Technobioplant, Tozer Ibérica, Rijk Zwaan Ibérica and Claus Ibérica for kindly providing seeds for the experiments. The authors declare that they have no conflict of interest and give their consent for publishing the results of the present study.
Rights and permissions
About this article
Cite this article
Verdejo-Lucas, S., Talavera, M. Pathogenic potential, parasitic success and host efficiency of Meloidogyne incognita and M. javanica on cucurbitaceous plant genotypes. Eur J Plant Pathol 153, 1287–1297 (2019). https://doi.org/10.1007/s10658-018-01642-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-018-01642-6