Abstract
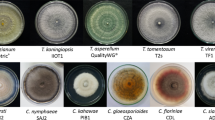
Rubber wood, one of the most preferred plantation timbers in the tropical regions, is easily prone to various moulds, decay and stain fungi. Lasiodiplodia theobromae, the dominant sapstain fungus of rubber wood, causes high economic loss to the industry through its bluish black sap wood discolouration. In our previous laboratory trials, Bacillus subtilis B1 isolated from market available compost was proved as a potent biocontrol agent against the sapstain fungus L. theobromae. The main aim of the present study was to demonstrate the biocontrol potential of B. subtilis B1 against rubber wood sapstain fungus on fresh rubber wood pieces during pre-monsoon, southwest monsoon and northeast monsoon seasons in the field. The visual ratings provided for the wood fungal growth and internal wood stain exhibited the bio-protectant efficiency of B. subtilis B1 even in the southwest monsoon season, which is considered as the most favourable period for the wood fungal growth. Biocontrol agent, B. subtilis B1 demonstrated significant inhibitions of sapstain fungus on open stacked rubber wood pieces in all the seasons. The frequent rainfalls during monsoon periods provide a conducive environment for fungal growth on felled rubber wood. Successful field evaluations of the biocontrol agent during pre-monsoon, monsoon and post-monsoon periods would guarantee year round protection against rubber wood sapstain. B. subtilis B1 displayed holistic biocontrol efficiency towards other stain (Ceratocystis sp., Fusarium sp.), mould (Aspergillus sp., Penicllium sp.) and decay fungi (Trametes versicolor) as well.


Similar content being viewed by others
References
Ashwini, N., & Srividya, S. (2014). Potentiality of Bacillus subtilis as biocontrol agent for management of anthracnose disease of chilli caused by Colletotrichum gloeosporioides. Biotechnology, 4, 127–136.
aWhere Inc. (2015). Weather Terrain Daily surfaced weather data aWhere Inc 2655. Blvd Suite: West Midway.
Barnett, H. L., & Hunter, B. B. (1972). Illusrated genera of imperfect fungi. 3rd edition, Burgess Publishing Co.
Behrendt, C. J., Blanchette, R. A., & Farrell, R. L. (1995). Biological control of blue-stain fungi in wood. Phytopathology, 85, 92–97.
Benko, R. (1988). Bacteria as possible organisms for biological control of blue stain. In The International Research Group on Wood Preservation, Document No: IRG/WP/1339. Stockholm: Sweden.
Bernier, R., Desrochers, M., & Jurasek, L. (1986). Antagonistic effect between Bacillus subtilis and wood staining fungi. Journal of the Institute of Wood Science, 10, 214–216.
Blanchette, R. A. & Farrell, R. L. (1997). Application of biological control agents in the forest products industry. In: Strategies for Improving Logs and Lumber. FRI Bulletin No. 204, Rotorua.
Bruce, A., Stewar, D., Verrall, S., & Wheatly, R. E. (2003). Effect of volatiles from bacteria and yeast on the growth and pigmentation of sapstain fungi. International Biodeterioration and Biodegradation, 5, 1101–1108.
Central Ground water information booklet of Thrissur district Kerala state (n.d.) http://cgwb.gov.in/District_Profile/Kerala/Thrissur.
Chow, S. (1983). Method of treating wood to prevent stain and decay, Patent No 4413023.
Croan, S. C. (1996). Biological control of sapstain fungi in wood. In The International Research Group on Wood Preservation Document, No: IRG/WP/96–10158. Stockholm: Sweden.
Damodharan, T. K., & Thulasidas, P. K. (2003).Transfer of some selected wood processing technologies to wood using industries in Kerala. KFRI Research Report, 301, 101.
Ejechi, B. O. (1997). Biological control of wood decay in an open tropical environment with Penicillium spp. and Trichoderma viride. International Biodeterioration & Biodegradation, 39, 295–299.
Ellis, M. B., & Ellis, J. P. (1985). Microfungi on land plants. In An identification hand book, Croom Helm. Ltd, Kent: U. K.
Florence, E. J. M. (1996). Sapstain microorganisms associated with selected commercially important timbers of Kerala and their possible control, Ph.D. Thesis, Cochin University of Science and Technology, Kerala.
Florence, E. J. M., & Balasundaran, M. (2009). Use of bioprotectant against fungal deterioration of rubberwood, KFRI Research Report No, 327, pp 49.
Florence, E. J. M., & Sharma, J. K. (1990). Botryodiplodia theobromae associated with blue staining in commercially important timbers. Material und Organismen, 25, 193–199.
Fravel, D. R. (2005). Commercialization and implementation of biocontrol. Annual Review of Phytopathology, 43, 337–359.
George, J. G., & Chandrashekar, H. M. (2014). Growth and trends in production and marketing of natural rubber in Kerala, India. International Journal of Current Research of Academic Review, 2, 53–61.
Helisto, P., Aktuganov, G., Galimzianova, N., Melentjev, A., & Korpela, T. (2001). Lytic enzyme complex of an antagonistic Bacillus sp X-b: Isolation and purification of components. Journal of Chromatography B, 758, 197–205.
Hiratsuka, Y., Chakravarty, P., Miao, S., & Ayer, W. A. (1994). Potential for biological protection against blue stain in Populus tremuloides with a hypomycetous fungus Stachybotrys cylindrospora. Canadian Journal of Forest Research, 24, 157–179.
Hong, L. T., Mohammed Ali, S., Tan, A. G., & Aljeet, S. K. (1982). Preservation and protection of rubberwood against biodeteriorating organism for more efficient utilization. The Malayan Forester, 45, 299–315.
Huh, N., Jang, Y., Lee, J., Kim, G. H., & Kim, J. J. (2010). Phylogenetic analysis of major molds inhabiting wood and their discolouration characteristics: Part 1 Genus Trichoderma. Holzforschung, 65, 257–263.
Kidd, G. J. D. (2001). CCA-treated lumber poses danger from arsenic and chromium. Pesticides and You, 21, 13–15.
Killmann, W., & Hong, L. T. (2000). Rubberwood — the success of an agricultural by-product. Unasylva, 51, 66–72.
Kreber, B. (1995). Advances in the understanding of hemlock brownstain. Material und Organismen, 28, 17–37.
Kreber, B., & Morrell, J. J. (1993). Ability of selected bacterial and fungal protectants to limit fungal stain in ponderosa pine sapwood. Wood and Fiber Science, 25, 23–24.
Kundu, A., Chakraborty, M. R., & Chatterjee, N. C. (2008). Biocontrol of wood decay by Trichoderma spp – retrospect and prospect. Asian Journal of Experimental Sciences, 22, 373–384.
Melentev, A. I., Helisto, P., Kuzmina, L. Y., Aktuganov, G. E., & Korpela, T. (2006). Use of antagonistic Bacilli for biocontrol of fungi degrading fresh wood. Applied Biochemistry and Microbiology, 42, 62–66.
Moita, C., Feio, S. S., Nunes, L., Curto, M. J. M., & Roseiro, J. C. (2005). Optimisation of physical factors on the production of active metabolites by Bacillus subtilis 355 against wood surface contaminant fungi. International Biodeterioration and Biodegradation, 55, 261–269.
Nazma, P., Ganapathy, P. M., Sasidharan, N., Bhat, K. M., & Gnanaharan, R. (1981). A handbook of kerala timbers. KFRI Research Report, 9, 260.
Nobuyuki, S., & Yoshio, O. (2006). Structure of fungal cell wall polysaccharides. Japanese Journal of Medical Mycology, 47, 179–184.
Payne, C., Bruce, A., & Staines, H. (2000). Yeast and bacteria as biological control agents against fungal discolouration of Pinus sylvestris pieces in laboratory – based tests and the role of antifungal volatiles. Holzforschung, 54, 563–569.
Ricard, J. L., & Bollen, W. B. (1967). Inhibition of Poria carbonica by Scytalidium sp an imperfect fungus isolated from Douglas fir poles. Canadian Journal of Botany, 46, 643–647.
Sajitha K. L. (2016). Identification and mass production of bacterial biocontrol against sapstain on rubberwood. Ph. D. Thesis. Forest Research Institute, Deemed University, Dehra Dun.
Sajitha, K. L., & Florence, E. J. M. (2013). Effects of Streptomyces sp on growth of rubberwood sapstain fungus Lasiodiplodia theobromae. Journal of Tropical Forest Science, 25, 393–399.
Sajitha, K. L., Florence, E. J. M., & Dev, S. A. (2014). Screening of bacterial biocontrol against sapstain fungus (Lasiodiplodia theobromae Pat ) of rubberwood (Hevea brasiliensis Muell Arg ). Research in Microbiology, 165, 541–548.
Sajitha, K. L., Dev, S. A., & Maria Florence, E. J. (2016). Identification and characterization of lipopeptides from Bacillus subtilis B1against sapstain fungus of rubberwood through MALDI-TOF-MS and RT-PCR. Current Microbiology, 73, 46–53.
Seifert, K. A., Breuil, C., Rossignol, L., Best, M., & Saddler, J. N. (1988). Screening for microorganisms with the potential for biological control of sapstain on unseasoned lumber. Material und Organismen, 23, 81–95.
Smith, R. S. (1991). Lumber protection today Proceedings of lumber protection in the 90’s Forintek Canada Corp Vancouver B C.
SPSS Inc Released. (2008). SPSS Statistics for Windows Version 17 0. Chicago: SPSS Inc.
Stilwell, M. A. (1966). A growth inhibitor produced by Cryptosporiopsis sp., an imperfect fungus isolated from yellow birch, Betula alleghaniensis Britt. Canadian Journal of Botany, 44, 259–267.
Subramanian, C. V. (1983). Hyphomycetes in the marine habitat. In Hyphomycetes: Taxonomy and Biology (pp. 283–294). London: Academic Press.
Susi, P., Aktuganov, G., Himanen, J., & Korpela, T. (2011). Biological control of wood decay against fungal infection. Journal of Environmental Management, 92, 1681–1689.
Uzunovic, A., Yang, P., Gagne, C., Breuil, L., Bernier, A., Gignac, B. M., Kim, M., & Can, S. H. (1999). Fungi that cause sapstain in Canadian softwoods. Canadian Journal of Microbiology, 45, 914–922.
Veenin, T., Veenin, A., & Denrungruang, P. (1999). Biological control of stain fungus in rubberwood (Hevea brasiliensis Muell Arg) by Trichoderma sp. Thailand Journal of Forest, 18, 73–86.
Viitanan, H. (1996). Factors affecting the development of mould and brown rot decay in wooden material and wooden structures- Effect of humidity temperature and exposure time, Ph.D. Thesis, The Swedish University of Agricultural Sciences Department of Forest Products, Uppsala.
Warrior, P., Konduru, K., & Vasudevan, P. (2002). Formulation of biological control agents for pest and disease management. In S. S. Gnanamanickam (Ed.), Biological Control of Crop Diseases (pp. 421–441). New York: Marcel Dekker.
Zhang, X. H., Zhao, G. H, Li, D. W., Li, S. P., & Hong, Q. (2014). Identification and evaluation of strain B37 of Bacillus subtilis antagonistic to sapstain fungi on poplar wood. The Scientific World Journal, doi:10.1155/2014/149342.
Acknowledgements
This study was conducted at Kerala Forest Research Institute, Peechi, Thrissur, India, supported by grants (BT/PR14542/AGR/05/527/2010) from the Department of Biotechnology, Ministry of Science and Technology, Government of India. We also thank Dr. P. Rugmini, Retired Scientist, KFRI for her valuable help in statistical data analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Sajitha, K.L., Dev, S.A. & Maria Florence, E.J. Biocontrol potential of Bacillus subtilis B1 against sapstain fungus in rubber wood. Eur J Plant Pathol 150, 237–244 (2018). https://doi.org/10.1007/s10658-017-1272-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-017-1272-z