Abstract
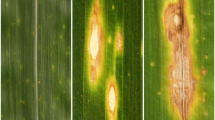
Fusarium head blight (FHB), predominantly caused by Fusarium graminearum and F. culmorum, is one of the most destructive diseases of wheat, reducing grain yield and quality of kernels. In diseased kernels trichothecne mycotoxins accumulate, which are harmful to human and animal health. Pathogen development and host responses to infection by F. graminearum were investigated in wheat spikes of the resistant cv. Sumai 3 and susceptible cv. Xiaoyan 22 to infection by means of electron microscopy and immunogold labelling techniques. The infection process of the pathogen in wheat spikes was similar in both the resistant and susceptible cultivars, but the pathogen developed more slowly in the resistant cv. Sumai 3 compared to the susceptible cv. Xiaoyan 22, indicating that fungal spread was restricted to the spike tissues of the resistant cultivar. The formation of thick-layered appositions and papillae was essentially more pronounced in the infected host tissues of the resistant cultivar than in the susceptible one. ß-1,3-glucan was detected in the appositions and papillae. Immunogold labelling studies demonstrated that labelling densities for lignin, thionins and hydroxyproline-rich glycoproteins (HRGP) over the cell walls of the infected tissues of the susceptible wheat cv. Xiaoyan 22 only slightly increased whereas these compounds intensely accumulated in the host cell walls of the infected wheat spikes of the resistant cv. Sumai 3. The labelling densities for the two plant hydrolases, ß -1,3-glucanase and chitinase, increased slightly in the infected wheat spike tissues of the susceptible cv. Xiaoyan 22, whereas higher labelling densities of both enzymes were found in the infected wheat spikes from the resistant cv. Sumai 3. Immunogold labelling of the Fusarium toxin DON in the infected wheat spike tissues showed that labelling densities in spike tissues for DON in the resistant cv. Sumai 3 were significantly lower than those in the susceptible cv. Xiaoyan 22. The significance of the induced morphological (e.g. thick-layered wall appositions and papillae) and chemical defence constituents (e.g. ß -1,3-glucanase, chitinase, lignin, thionin and HRGP) in resistance to FHB as well as the possible role of DON as an aggressiveness factor in translation of transcripts of defence response genes and spreading of F. graminearum and F. culmorum are discussed.





Similar content being viewed by others
Abbreviations
- FHB:
-
Fusarium head blight
- DON:
-
deoxynivalenol
- TBS:
-
Tris-buffered saline
- HRGP:
-
hydroxyproline-rich glycoproteins
- TEM:
-
transmission electron microscopy
References
Aist, J. R. (1976). Papillae and related wound plugs of plant cells. Annual Review of Phytopathology, 14, 145–163.
Arlorio, M., Ludwig, A., Boller, T., & Bonfante, P. (1992). Inhibition of fungal growth by plant chitinases and β-1,3-glucanases: A morphological study. Protoplasma, 171, 34–43.
Bai, G. H., Desjardins, A. E., & Plattner, R. D. (2002). Deoxynivalenol-nonproducing Fusarium graminearum causes initial infection, but does not cause disease spread in wheat spikes. Mycopathologia, 153, 91–98.
Bai, G. H., & Shaner, G. (1994). Scab of wheat: Prospects for control. Plant Disease, 78, 760–766.
Ban, T., & Suenaga, K. (2000). Genetic analysis of resistance to Fusarium head blight caused by Fusarium graminearum in Chinese wheat cultivar Sumai 3 and Japanese cultivar Saikai 165. Euphytica, 113, 87–99.
Benhamou, N., Joosten, M. H. A. J., & De Wit, P. J. G. M. (1990). Subcellular localization of chitinase and of its potential substrate in tomato root tissues infected by Fusarium oxysporum f.sp. radicis-lycopersici. Plant Physiology, 92, 1108–1120.
Benhamou, N., Mazau, D., Grenier, J., & Esquerré-Tugayé, M.-T. (1991). Time-course study of the accumulation of hydroxyproline-rich glycoproteins in root cells of susceptible and resistant tomato plants infected by Fusarium oxysporum f.sp. radicis-lycopersici. Planta, 184, 196–208.
Boller, T. (1987). Hydrolytic enzymes in plant disease resistance. In T. Kosuge, & E. W. Nester (Eds.) Plant-microbe interactions. Molecular and genetic perspectives, vol 2 (pp. 385–413). New York: Macmillan.
Boyacioglu, D., & Hettiarachchy, N. S. (1995). Changes in some biochemical components of wheat grain that was infected with Fusarium graminearum. Journal of Cereal Science, 21, 57–62.
Desjardins, A. E., & Hohn, T. M. (1997). Mycotoxins in plant pathogenesis. Molecular Plant-Microbe Interactions, 10, 147–152.
Desjardins, A. E., Proctor, R. H., Bai, G., McCormick, S. P., Shaner, G., & Buechley, G., et al. (1996). Reduced virulence of trichothecene non-producing mutants of Gibberella zeae in wheat field tests. Molecular Plant-Microbe Interactions, 9, 775–781.
Enkerli, K., Hahn, M. G., & Mims, C. W. (1997). Ultrastructure of compatible and incompatible interactions of soybean roots infected with the plant pathogenic oomycete Phytophthora sojae. Canadian Journal of Botany, 75, 1493–1508.
Epple, P., Apel, K., & Bohlmann, H. (1997). Overexpression of an endogenous thionin enhances resistance of arabidopsis against Fusarium oxysporum. Plant Cell, 9(4), 509–520.
Kang, Z., & Buchenauer, H. (1999). Immunocytochemical localization of Fusarium toxins in infected wheat spikes by Fusarium culmorum. Physiological and Molecular Plant Pathology, 55, 275–288.
Kang, Z., & Buchenauer, H. (2000a). Cytology and ultrastructure of the infection of wheat spikes by Fusarium culmorum. Mycological Research, 104, 1083–1093.
Kang, Z., & Buchenauer, H. (2000b). Ultrastructural and cytochemical studies on cellulose, xylan and pectin degradation in wheat spikes infected by Fusarium culmorum. Journal of Phytopathology, 148, 263–275.
Kang, Z., & Buchenauer, H. (2000c). Ultrastructural and immunocytochemical investigation of pathogen development and host responses in resistant and susceptible wheat spikes infected by Fusarium culmorum. Physiological and Molecular Plant Pathology, 57, 255–268.
Kang, Z., & Buchenauer, H. (2002). Immunocytochemical localization of ß-1,3-glucanase and chitinase in Fusarium culmorum-infected wheat spikes. Physiological and Molecular Plant Pathology, 60, 141–153.
Kang, Z., & Buchenauer, H. (2003). Immunocytochemical localization of cell wall-bound thionins and hydroxyproline-rich glycoproteins in Fusarium culmorum-infected wheat spikes. Journal of Phytopathology, 151(3), 120–129.
Kang, Z., Huang, L., & Buchenauer, H. (2004). Ultrastructural and cytochemical studies on infection of wheat spikes by Microdochium nivale. Journal of Plant Diseases and Protection, 111(4), 351–361.
Kang, Z., Zingen-Sell, I., & Buchenauer, H. (2005). Infection of wheat spikes by Fusarium avenaceum and alteration of cell wall components in the infected tissue. European Journal of Plant Pathology, 111(1), 19–28.
Keen, N. T., & Yoshikawa, M. (1983). ß-1,3-Endoglucanases from soybean release elicitor-active carbohydrates from fungal cell walls. Plant Physiology, 71, 460–465.
Kombrink, E., Schröder, M., & Hahlbrock, K. (1988). Several pathogenesis-related proteins in potato are 1,3-ß-glucanases and chitinases. Proceedings of the National Academy of Sciences of the United States of America, 85, 782–786.
Langevin, F., Eudes, F., & Comeau, A. (2004). Effect of trichothecenes produced by Fusarium graminearum during Fusarium head blight development in six cereal species. European Journal of Plant Pathology, 110, 735–746.
McMullen, M., Jones, R., & Gallenberg, D. (1997). Scab of wheat and barley: A reemerging disease of devastating impact. Plant Disease, 81, 1340–1348.
Mesterházy, Á. (1995). Types and components of resistance against Fusarium head blight of wheat. Plant Breeding, 114, 377–386.
Mesterházy, Á., Bartók, T., Kászonyi, G., Varga, M., Tóth, B., & Varga, J. (2005). Common resistance to different Fusarium spp. causing Fusarium head blight in wheat. European Journal of Plant Pathology, 112, 267–281.
Miller, J. D., & Arnison, P. G. (1986). Degradation of deoxynivalenol by suspension cultures of the Fusarium head blight resistant wheat cultivar Frontana. Canadian Journal of Plant Pathology, 8, 147–150.
Miller, J. D., & Ewen, M. A. (1997). Toxic effects of deoxynivalenol on ribosomes and tissues of the spring wheat cultivars Frontana and Casavant. Natural Toxins, 5, 234–237.
Parry, D. W., Jenkinson, P., & McLeod, L. (1995). Fusarium ear blight (scab) in small grain cereals, a review. Plant Pathology, 44, 207–238.
Pritsch, C., Muehlbauer, G. J., Bushnell, W. R., Somers, D. A., & Vance, C. P. (2000). Fungal development and induction of defense response genes during early infection of wheat spikes by Fusarium graminearum. Molecular Plant-Microbe Interactions, 13, 159–169.
Proctor, R. H., Desjardins, A. E., McCormick, S. P., Plattner, R. D., Alexander, N. J., & Brown, D. W. (2002). Genetic analysis of the role of trichothecene and fumonisin mycotoxins in the virulence of Fusarium. European Journal of Plant Pathology, 108, 691–698.
Schroeder, H. W., & Christensen, J. J. (1963). Factors affecting resistance of wheat to scab caused by Gibberella zeae. Phytopathology, 53, 831–818.
Snijders, C. H. (2004). Resistance of wheat to Fusarium infection and trichothecene formation. Toxicology Letter, 153, 37–46.
Snijders, C. H. A., & Krechting, C. F. (1992). Inhibition of deoxynivalenol translocation and fungal colonization in Fusarium head blight resistant wheat. Canadian Journal Botany, 70, 1570–1576.
Snijders, C. H. A., & Perkowski, J. (1990). Effects of head blight caused by Fusarium culmorum on toxin content and weight of wheat kernels. Phytopathology, 80, 566–570.
Usleber, E., Märtlbauer, E., Dietrich, R., & Terplan, G. (1991). Direct enzyme-linked immunosorbent assays for the detection of the 8-ketotrichothecene mycotoxins deoxynivalenol, 3-acetyldeoxynivalenol and 15-acetyldeoxynivalenol, in buffer solutions. Journal of Agricultural and Food Chemistry, 39, 2091–2095.
Van Ginkel, M., Van der Schaar, W., Zhuping, Y., & Rajaram, S. (1996). Inheritance of resistance to scab in two wheat cultivars from Brazil and China. Plant Disease, 80, 863–867.
Wang, Y. Z., & Miller, J. D. (1988). Effects of Fusarium graminearum metabolites on wheat tissue in relation to fusarium head blight resistance. Journal of Phytopathology, 122, 118–125.
Wanyoike, M. W., Kang, Z., & Buchenauer, H. (2002). Importance of cell wall degrading enzymes produced by Fusarium graminearum during infection of wheat heads. European Journal of Plant Pathology, 108(1), 803–810.
Wessels, J. G. H., & Sietsma, J. H. (1981). Fungal cell walls: A survey. In W. Tanner, & F. A. Loewus (Eds.) Plant carbohydrates II. Extracellular carbohydrates. Encyclopedia of plant physiology, new series vol. 13B (pp. 352–395). Berlin, Germany: Springer Verlag.
Wubben, J., Joosten, M. H. A. J., Van Kan, J. A. L., & De Wit, P. J. G. M. (1992). Subcellular localization of plant chitinases and 1,3-β-gluanases in Cladosporium fulvum (syn. Fulvia fulva)-infected tomato leaves. Physiological and Molecular Plant Pathology, 41, 23–32.
Zadoks, J. C., Chang, T., & Konzak, C. F. (1974). A decimal code for the growth stages of cereals. Weed Research, 14, 415–421.
Zhou, W. C., Kolb, F. L., Bai, G. H., Domier, L. L., & Yao, J. B. (2002). Effect of individual Sumai 3 chromosomes on resistance to scab spread within spikes and deoxynivalenol accumulation with kernels in wheat. Hereditas, 137(2), 81–89.
Acknowledgements
The authors wish to thank Dr. E. Usleber (Institute for Hygiene and Technology, University Munich, Germany), Dr. Ruel (Centre de Recherches sur les Macromolécules Végétales, Université Joseph Fourier, France), Dr. K. Apel (Institut für Pflanzenwissenschaften, Swiss Institute of Technology (ETH), Zürich, Switzerland), and Dr. E. Hood (Department of Bichemistry and Biophysics, Texas A&M University, USA) for kindly supplying antibodies. This study has been financially supported by the National Basic Research Programme of China (No.2006CB101901), Key Research Project, Ministry of Education of China (03158), Nature Science Foundation of China (No.30125031), the Programme for Changjiang Scholars and Innovative Research Teams in Universities, Ministry of Education of China (No.200558) and the 111 Project from Ministry of Education of China (B07049).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, Z., Buchenauer, H., Huang, L. et al. Cytological and immunocytochemical studies on responses of wheat spikes of the resistant Chinese cv. Sumai 3 and the susceptible cv. Xiaoyan 22 to infection by Fusarium graminearum . Eur J Plant Pathol 120, 383–396 (2008). https://doi.org/10.1007/s10658-007-9230-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-007-9230-9