Abstract
Objective
To assess spatial aggregates of amyotrophic lateral sclerosis (ALS) incident cases, using a solid geo-epidemiological statistical method, in France.
Methods
This population-based study (2003–2011) investigated 47.1 million person-years of follow-up (PYFU). Case ascertainment of incident ALS cases was based on multiple sources (ALS referral centers, hospital centres and health insurance data). Neurologists confirmed all ALS diagnoses. Exhaustiveness was estimated through capture-recapture. Aggregates were investigated in four steps: (a) geographical modelling (standardized incidence ratio (SIR) calculation), (b) analysis of the spatial distribution of incidence (Phothoff-Winttinghill’s test, Global Moran’s Index, Kulldorf’s spatial scan statistic, Local Moran’s Index), (c) classification of the level of certainty of spatial aggregates (i.e. definite cluster; probable over-incidence area; possible over-incidence area) and (d) evaluation of the robustness of the results.
Results
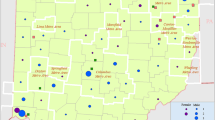
The standardized incidence of ALS was 2.46/100,000 PYFU (95% CI 2.31–2.63, European population as reference) based on 1199 incident cases. We identified 13 areas of spatial aggregates: one cluster (stable in robustness analysis), five probable over-incidence areas (2 stable in robustness analysis) and seven possible over-incidence areas (including 4 stable areas in robustness analysis). A cluster was identified in the Rhône-Alpes region: 100 observed vs 54.07 expected cases for 2,411,514 PYFU, SIR: 1.85 (95% CI 1.50–2.25).
Conclusion
We report here one of the largest investigations of incidence and spatial aggregation of ALS ever performed in a western country. Using a solid methodology framework for case ascertainment and cluster analysis, we identified 13 areas that warrant further investigation.



Similar content being viewed by others
References
Marin B, Boumédiene F, Logroscino G, et al. Variation in worldwide incidence of amyotrophic lateral sclerosis: a meta-analysis. Int J Epidemiol. 2017;46:57–74. https://doi.org/10.1093/ije/dyw061.
Marin B, Logroscino G, Boumédiene F, et al. Clinical and demographic factors and outcome of amyotrophic lateral sclerosis in relation to population ancestral origin. Eur J Epidemiol. 2016;31:229–45. https://doi.org/10.1007/s10654-015-0090-x.
Renton AE, Chiò A, Traynor BJ. State of play in amyotrophic lateral sclerosis genetics. Nat Neurosci. 2014;17:17–23. https://doi.org/10.1038/nn.3584.
Bozzoni V, Pansarasa O, Diamanti L, et al. Amyotrophic lateral sclerosis and environmental factors. Funct Neurol. 2016;31:7–19.
Filippini T, Tesauro M, Fiore M, et al. Environmental and occupational risk factors of amyotrophic lateral sclerosis: a population-based case-control study. Int J Environ Res Pub Health. 2020. https://doi.org/10.3390/ijerph17082882.
Jacquez GM, Waller LA, Grimson R, Wartenberg D. The analysis of disease clusters, Part I: state of the art. Infect Control Hosp Epidemiol. 1996;17:319–27. https://doi.org/10.1086/647301.
Longinetti E, Pupillo E, Belometti C, et al. Geographical clusters of amyotrophic lateral sclerosis and the bradford hill criteria. Amyotroph Later Scler Front Degener. 2021;23(5–6):329–43.
Olsen SF, Martuzzi M, Elliott P. Cluster analysis and disease mapping–why, when, and how? a step by step guide. BMJ. 1996;313:863–6.
Plato CC, Garruto RM, Galasko D, et al. Amyotrophic lateral sclerosis and parkinsonism-dementia complex of Guam: changing incidence rates during the past 60 years. Am J Epidemiol. 2003;157:149–57. https://doi.org/10.1093/aje/kwf175.
Murch SJ, Cox PA, Banack SA, et al. Occurrence of beta-methylamino-l-alanine (BMAA) in ALS/PDC patients from guam. Acta Neurol Scand. 2004;110:267–9. https://doi.org/10.1111/j.1600-0404.2004.00320.x.
Araki S, Iwahashi Y, Kuroiwa Y. Epidemiological study of amyotrophic lateral sclerosis and allied disorders in the kii peninsula (Japan). J Neurol Sci. 1967;4:279–87. https://doi.org/10.1016/0022-510x(67)90106-2.
Kihira T, Yoshida S, Kondo T, et al. An increase in ALS incidence on the Kii Peninsula, 1960–2009: a possible link to change in drinking water source. Amyotroph Later Scler. 2012;13:347–50. https://doi.org/10.3109/17482968.2012.674140.
Migliaretti G, Berchialla P, Dalmasso P, et al. Amyotrophic lateral sclerosis in piedmont (Italy): a Bayesian spatial analysis of the incident cases. Amyotroph Later Scler Front Degener. 2013;14:58–65. https://doi.org/10.3109/21678421.2012.733401.
Lannuzel A, Mecharles S, Tressières B, et al. Clinical varieties and epidemiological aspects of amyotrophic lateral sclerosis in the caribbean island of guadeloupe: a new focus of ALS associated with parkinsonism. Amyotroph Later Scler Front Degener. 2015;16:216–23. https://doi.org/10.3109/21678421.2014.992026.
De Benedetti S, Lucchini G, Del Bò C, et al. Blood trace metals in a sporadic amyotrophic lateral sclerosis geographical cluster. Biometals Int J Role Met Ions Biol Biochem Med. 2017;30:355–65. https://doi.org/10.1007/s10534-017-0011-4.
Kilness AW, Hichberg FH. Amyotrophic lateral sclerosis in a high selenium environment. JAMA. 1977;237:2843–4.
Masseret E, Banack S, Boumédiène F, et al. Dietary BMAA exposure in an amyotrophic lateral sclerosis cluster from southern France. PLoS ONE. 2013;8: e83406. https://doi.org/10.1371/journal.pone.0083406.
Banack SA, Metcalf JS, Bradley WG, Cox PA. Detection of cyanobacterial neurotoxin β-N-methylamino-l-alanine within shellfish in the diet of an ALS patient in Florida. Toxicon Off J Int Soc Toxinol. 2014;90:167–73. https://doi.org/10.1016/j.toxicon.2014.07.018.
Delzor A, Couratier P, Boumédiène F, et al. Searching for a link between the L-BMAA neurotoxin and amyotrophic lateral sclerosis: a study protocol of the French BMAALS programme. BMJ Open. 2014;4: e005528. https://doi.org/10.1136/bmjopen-2014-005528.
Delzor A, Marin B, Boumediene F, et al. BMAALS: a French national project searching for a link between amyotrophic lateral sclerosis and the neurotoxic amino acid L-BMAA. Amyotroph Later Scler Front Degener. 2014;15:155–6. https://doi.org/10.3109/21678421.2013.837933.
Marin B, Hamidou B, Couratier P, et al. Population-based epidemiology of amyotrophic lateral sclerosis (ALS) in an ageing Europe–the French register of ALS in Limousin (FRALim register). Eur J Neurol. 2014;21(1292–1300):e78-79. https://doi.org/10.1111/ene.12474.
Brooks BR, Miller RG, Swash M, et al. El escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph Later Scler Mot Neuron Disord. 2000;1:293–9. https://doi.org/10.1080/146608200300079536.
Hook EB, Regal RR. Capture-recapture methods in epidemiology: methods and limitations. Epidemiol Rev. 1995;17:243–64. https://doi.org/10.1093/oxfordjournals.epirev.a036192.
Orton H, Rickard R, Gabella B. Capture-recapture estimation using statistical software. Epidemiol Camb Mass. 1999;10:563–4. https://doi.org/10.1097/00001648-199909000-00021.
Sakamoto Y, Ishiguro M, Kitagawa G. Akaike Information Criterion Statistics. Netherlands: Springer; 1986.
Schwarz G. Estimating the dimension of a model. Ann Stat. 1978;6:461–4. https://doi.org/10.1214/aos/1176344136.
Germonneau P, Thillaut H, Santo GDE, E, Borraz O,. Aspects méthodologiques de l’investigation des signalements d’agrégats spatio-temporels de maladies non infectieuses. Environ Risques Santé. 2005;4:329–34.
Breslow NE, Day NE (1987) Statistical methods in cancer research. volume II--the design and analysis of cohort studies. IARC Sci Publ: 1–406
Potthoff RF (2014) Homogeneity, Potthoff-Whittinghill Tests of. In: Wiley StatsRef: Stat Refer Online American Cancer Soc
Anselin L. Local Indicators of spatial association—ISA. Geogr Anal. 2010;27:93–115. https://doi.org/10.1111/j.1538-4632.1995.tb00338.x.
Kulldorff M, Nagarwalla N. Spatial disease clusters: detection and inference. Stat Med. 1995;14:799–810. https://doi.org/10.1002/sim.4780140809.
Blum A, Biraben J-N. Cliff A.D. et Ord J.K. — spatial processes. Mode appl Popul. 1982;37:963–963.
Lagrange E, Bonneterre V, Talbot K, et al (2017) A high-incidence cluster of ALS in the French Alps: common environment and multiple exposures. In: RISE 2nd International Meeting on Environmental Health 2017, Strasbourg, France.
Lagrange E, Vernoux JP, Reis J, et al. An amyotrophic lateral sclerosis hot spot in the French alps associated with genotoxic fungi. J Neurol Sci. 2021;427: 117558. https://doi.org/10.1016/j.jns.2021.117558.
Lagrange E, Juntas-Morales R, Pageot N, et al. The cyanotoxin BMAA and increased incidence in ALS clusters: a public health concern in France. Amyotroph Later Scler Front Degener. 2018;19:180.
Nicoletti A, Vasta R, Venti V, et al. The epidemiology of amyotrophic lateral sclerosis in the Mount Etna region: a possible pathogenic role of volcanogenic metals. Eur J Neurol. 2016;23:964–72. https://doi.org/10.1111/ene.12973.
Boumediene F, Vasta R, Rascunà C, et al. Amyotrophic lateral sclerosis spatial epidemiology in the Mount Etna region, Italy. Eur J Neurol. 2019;26:e90–1. https://doi.org/10.1111/ene.14011.
Luna J, Leleu J-P, Preux P-M, et al. Residential exposure to ultra high frequency electromagnetic fields emitted by Global System for Mobile (GSM) antennas and amyotrophic lateral sclerosis incidence: A geo-epidemiological population-based study. Environ Res. 2019;176: 108525. https://doi.org/10.1016/j.envres.2019.108525.
Logroscino G, Traynor BJ, Hardiman O, et al. Incidence of amyotrophic lateral sclerosis in Europe. J Neurol Neurosurg Psychiatry. 2010;81:385–90. https://doi.org/10.1136/jnnp.2009.183525.
Logroscino G, Piccininni M. Amyotrophic lateral sclerosis descriptive epidemiology: the origin of geographic difference. Neuroepidemiology. 2019;52:93–103. https://doi.org/10.1159/000493386.
Erazo D, Luna J, Preux P-M, et al. Amyotrophic lateral sclerosis mortality rates in latin America and the caribbean: a meta-analysis. Amyotroph Later Scler Front Degener. 2022. https://doi.org/10.1080/21678421.2022.2048310.
Logroscino G, Piccininni M, Marin B, et al. Global, regional, and national burden of motor neuron diseases 1990–2016: a systematic analysis for the global burden of disease study 2016. Lancet Neurol. 2018;17:1083–97. https://doi.org/10.1016/S1474-4422(18)30404-6.
Rooney J, Vajda A, Heverin M, et al. Spatial cluster analysis of population amyotrophic lateral sclerosis risk in Ireland. Neurology. 2015;84:1537–44. https://doi.org/10.1212/WNL.0000000000001477.
Uenal H, Rosenbohm A, Kufeldt J, et al. Incidence and geographical variation of amyotrophic lateral sclerosis (als) in southern germany – completeness of the ALS registry Swabia. PLoS ONE. 2014;9: e93932. https://doi.org/10.1371/journal.pone.0093932.
Caller TA, Chipman JW, Field NC, Stommel EW. Spatial analysis of amyotrophic lateral sclerosis in northern new England, USA, 1997–2009. Muscle Nerve. 2013;48:235–41. https://doi.org/10.1002/mus.23761.
Torbick N, Hession S, Stommel E, Caller T. Mapping amyotrophic lateral sclerosis lake risk factors across northern New England. Int J Health Geogr. 2014;13:1. https://doi.org/10.1186/1476-072X-13-1.
Henry KA, Fagliano J, Jordan HM, et al. Geographic variation of amyotrophic lateral sclerosis incidence in new Jersey, 2009–2011. Am J Epidemiol. 2015;182:512–9. https://doi.org/10.1093/aje/kwv095.
Boumédiène F, Druet-Cabanac M, Marin B, et al. Contribution of geolocalisation to neuroepidemiological studies: incidence of ALS and environmental factors in limousin, France. J Neurol Sci. 2011;309:115–22. https://doi.org/10.1016/j.jns.2011.07.002.
Alonso V, Villaverde-Hueso A, Hens MJ, et al. Increase in motor neuron disease mortality in Spain: temporal and geographical analysis (1990–2005). Amyotroph Later Scler Off Publ World Fed Neurol Res Group Mot Neuron Dis. 2011;12:192–8. https://doi.org/10.3109/17482968.2010.543688.
Bradley WG, Stommel EW, Shi X, et al. Spatial cluster analysis of population amyotrophic lateral sclerosis risk in Ireland. Neurology. 2015;85:1822–3. https://doi.org/10.1212/01.wnl.0000473800.89178.a9.
Knox E (1989) Detection of clusters. In: Methodology of enquiries into disease clustering. London: small area health statistics unit Elliot P: 17–20
Elliott P, Wartenberg D. Spatial epidemiology: current approaches and future challenges. Environ Health Perspect. 2004;112:998–1006. https://doi.org/10.1289/ehp.6735.
Huisman MHB, de Jong SW, van Doormaal PTC, et al. Population based epidemiology of amyotrophic lateral sclerosis using capture-recapture methodology. J Neurol Neurosurg Psychiatry. 2011;82:1165–70. https://doi.org/10.1136/jnnp.2011.244939.
Combes A, El Abdellaoui S, Sarazin C, et al. Validation of the analytical procedure for the determination of the neurotoxin β-N-methylamino-L-alanine in complex environmental samples. Anal Chim Acta. 2013;771:42–9. https://doi.org/10.1016/j.aca.2013.02.016.
Combes A, El Abdellaoui S, Vial J, et al. Development of an analytical procedure for quantifying the underivatized neurotoxin β-N-methylamino-L-alanine in brain tissues. Anal Bioanal Chem. 2014;406:4627–36. https://doi.org/10.1007/s00216-014-7872-y.
Svoboda P, Combes A, Petit J, et al. Synthesis of a molecularly imprinted sorbent for selective solid-phase extraction of β-N-methylamino-L-alanine. Talanta. 2015;144:1021–9. https://doi.org/10.1016/j.talanta.2015.07.052.
Acknowledgements
We thank all the French bodies coordinating the ALS referral centers, public and private hospitals, health insurance sources and neurologists who collaborated with the BMAALS consortium. BMAALS consortium (in alphabetical order by last name): Eric Abadie, Béatrice Bec, Gérard Besson, Dominique Bicout, Vincent Bonneterre, Farid Boumédiene, Luc Brient, William Camu, Audrey Combes, Philippe Couratier, Aurelie Delzor, Saïda El Abdellaoui, Edwige Garnier, Emmeline Lagrange, Jean Philippe Leleu, Marion Lengronne, Jaime Luna, Benoit Marin, Estelle Masseret, Annick Mejean, Raoul Juntas-Morales, Marie Nicol, Nicolas Pageot, François Paraf, Valérie Pichon, Olivier Ploux, Pierre-Marie Preux, Véronique Rigaud.
Funding
This work was supported by the French National Research Agency (ANR) grant number programme ANR-11-CESA-0014 (Project “BMAALS”) and by the French Association against ALS (BMAA-LIM project).Agence Nationale de la Recherche,ANR-11-CESA-0014
Author information
Authors and Affiliations
Author notes
Farid Boumédiene and Benoît Marin have contributed equally.
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors reported no potential conflict of interest.
Ethical approval
The collection of data was authorized by the CCTIRS (Comité consultatif sur le traitement de l’information en matière de recherche dans le domaine de la santé, authorization no. 10487bis) on 20th January 2011 and the CNIL (Commission Nationale de l’Informatique et des Libertés, authorization no. 911038) on 27 May 2011. The protocol was reviewed by an ethics review board (Comité de Protection des Personnes Sud-Ouest Outre Mer IV) on 10th February 2011.
Consent to participate
All patients agreed to have their data recorded and analyzed in accordance with French law.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Boumédiene, F., Marin, B., Luna, J. et al. Spatio-temporal clustering of amyotrophic lateral sclerosis in France: A population-based study. Eur J Epidemiol 37, 1181–1193 (2022). https://doi.org/10.1007/s10654-022-00904-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10654-022-00904-2