Abstract
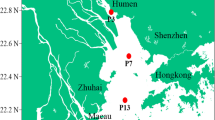
In natural habitats, the diversity of anaerobic ammonia-oxidizing (anammox) bacteria could be affected by multiple environmental variables. In this study, we investigated the distribution of the anammox bacterial community in surface sediment from the Dongjiang River (riverine sediment, DJ) to the Pearl River Estuary (estuarine sediment, PRE) and then to the South China Sea (coastal sediment, SCS). The results revealed evident differences in the structural diversity of anammox bacteria in three different habitats. Candidatus Brocadia accounted for approximately 90% of the total anammox bacteria in DJ, conversely, Ca. Scalindua dominated in the SCS. Nevertheless, Ca. Scalindua, Ca. Brocadia and Ca. Kuenenia coexisted in the PRE. The qPCR results indicated that anammox bacterial 16S rRNA gene abundance ranged from 2.23 × 105 to 1.19 × 107 copies g−1 of wet weight, but no significant correlation was found between the abundances and environmental variables (p > 0.05). The relative abundances of Ca. Brocadia gradually decreased with increasing salinity, and Ca. Scalindua showed the opposite trend, suggesting that salinity was a crucial factor in sculpturing the community composition of anammox bacteria in natural environments. Ca. Brocadia should be able to live in freshwater ecosystems, but it can also tolerate a certain level of salinity. Ca. Scalindua was halophilic anammox bacterium and exists only in saline environments. Ca. Kuenenia could adapt to a wide range of salinity and preferred to live in high DIN level conditions according to our search. The distribution pattern of anammox bacteria may be the result of microbial migration and long-term adaptation to salinity.






Similar content being viewed by others
References
Anderson DM, Glibert PM, Burkholder JM (2002) Harmful algal blooms and eutrophication: nutrient sources, composition, and consequences. Estuaries 25:704–726. https://doi.org/10.1007/BF02804901
Bernhard AE et al. (2005) Loss of diversity of ammonia-oxidizing bacteria correlates with increasing salinity in an estuary system. Environ Microbiol 7:1289–1297. https://doi.org/10.1111/j.1462-2920.2005.00808.x
Brandsma J et al. (2011) A multi-proxy study of anaerobic ammonium oxidation in marine sediments of the Gullmar Fjord, Sweden. Environ Microbiol Rep 3:360–366. https://doi.org/10.1111/j.1758-2229.2010.00233.x
Dale OR, Tobias CR, Song B (2009) Biogeographical distribution of diverse anaerobic ammonium oxidizing (anammox) bacteria in Cape Fear River Estuary. Environ Microbiol 11:1194–1207. https://doi.org/10.1111/j.1462-2920.2008.01850.x
Duce RA et al. (2008) Impacts of atmospheric anthropogenic nitrogen on the open ocean. Science 320:893–897. https://doi.org/10.1126/science.1150369
Egli K et al. (2003) Microbial composition and structure of a rotating biological contactor biofilm treating ammonium-rich wastewater without organic carbon. Microb Ecol 45:419–432. https://doi.org/10.1007/s00248-002-2037-5
Frank J et al. (2018) Resolving the complete genome of Kuenenia stuttgartiensis from a membrane bioreactor enrichment using Single-Molecule Real-Time sequencing. Sci Rep 8:4580. https://doi.org/10.1038/s41598-018-23053-7
Fu BB et al. (2015) Shift of anammox bacterial community structure along the Pearl Estuary and the impact of environmental factors. J Geophys Res Oceans 120(4):2869–2883. https://doi.org/10.1002/2014JC010554
Galloway JN et al. (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320:889–892. https://doi.org/10.1126/science.1136674
Gao DW et al. (2018) Anaerobic ammonia oxidizing bacteria: ecological distribution, metabolism, and microbial interactions. Front Environ Sci Eng 12:3. https://doi.org/10.1007/s11783-018-1035-x
Guan FJ et al. (2017) A fast sodium hypobromite oxidation method for the sequential determination of ammonia nitrogen in small volume. Ecol Sci 36:42–48. https://doi.org/10.14108/j.cnki.1008-8873.2017.02.006
Han P, Gu JD (2015) Further analysis of anammox bacterial community structures along an anthropogenic nitrogen-input gradient from the riparian sediments of the Pearl River delta to the deep-ocean sediments of the South China Sea. Geomicrobiol J 32(9):1–10. https://doi.org/10.1080/01490451.2014.1001502
Hou LJ et al. (2013) Anaerobic ammonium oxidation (anammox) bacterial diversity, abundance, and activity in marsh sediments of the Yangtze Estuary. J Geophys Res Biogeosci 118:1237–1246. https://doi.org/10.1002/jgrg.20108
Hou LJ et al. (2015) Anaerobic ammonium oxidation and its contribution to nitrogen removal in China’s coastal wetlands. Sci Rep 5:15621. https://doi.org/10.1038/srep15621
Hu BL et al. (2012) Distribution and diversity of anaerobic ammonium-oxidizing bacteria in the sediments of the Qiantang River. Environ Microbiol Rep 4:540–547. https://doi.org/10.1111/j.1758-2229.2012.00360.x
Humbert S, Zopfi J, Tarnawski SE (2012) Abundance of anammox bacteria in different wetland soils. Environ Microbiol Rep 4:0–0. https://doi.org/10.1111/j.1758-2229.2012.00347.x
Jaeschke A et al. (2009) 16S rRNA gene and lipid biomarker evidence for anaerobic ammonium-oxidizing bacteria (anammox) in California and Nevada hot springs. Fems Microbiol Ecol 3:3. https://doi.org/10.1111/j.1574-6941.2008.00640.x
Jiang XF et al. (2017) Salinity-driven shifts in the activity, diversity, and abundance of anammox bacteria of estuarine and coastal wetlands. Phys Chem Earth, Parts A/B/C 97:46–53. https://doi.org/10.1016/j.pce.2017.01.012
Jiao LJ et al. (2018) Significant microbial nitrogen loss from denitrification and anammox in the land-sea interface of low permeable sediments. Int Biodeterior Biodegrad 135:80–89. https://doi.org/10.1016/j.ibiod.2018.10.002
Kartal B et al. (2006) Adaptation of a freshwater anammox population to high salinity wastewater. J Biotechnol 126:546–553. https://doi.org/10.1016/j.jbiotec.2006.05.012
Kartal B et al. (2007a) Anammox bacteria disguised as denitrifiers: nitrate reduction to dinitrogen gas via nitrite and ammonium. Environ Microbiol 9:635–642. https://doi.org/10.1111/j.1462-2920.2006.01183.x
Kartal B et al. (2007b) Candidatus “Anammoxoglobus propionicus” a new propionate oxidizing species of anaerobic ammonium oxidizing bacteria. Syst Appl Microbiol 30:39–49. https://doi.org/10.1016/j.syapm.2006.03.004
Kim H et al. (2016) Distributions, abundances and activities of microbes associated with the nitrogen cycle in riparian and stream sediments of a river tributary. Water Res 106:51–61. https://doi.org/10.1016/j.watres.2016.09.048
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Kuypers MMM et al. (2003) Anaerobic ammonium oxidation by anammox bacteria in the Black Sea. Nature 422:608. https://doi.org/10.1038/nature01472
Li M et al. (2013) Using the variation of anammox bacteria community structures as a bio-indicator for anthropogenic/terrestrial nitrogen inputs in the Pearl River Delta (PRD). Appl Microbiol Biotechnol 97:9875–9883. https://doi.org/10.1007/s00253-013-4990-y
Li P et al. (2018) Seasonal variation of anaerobic ammonium oxidizing bacterial community and abundance in tropical mangrove wetland sediments with depth. Appl Soil Ecol 130:149–158. https://doi.org/10.1016/j.apsoil.2018.06.009
Mulder A et al. (1995) Anaerobic ammonium oxidation discovered in a denitrifying fluidized bed reactor. Fems Microbiol Ecol 16:177–184. https://doi.org/10.1016/0168-6496(94)00081-7
Oshiki M, Satoh H, Okabe S (2016) Ecology and physiology of anaerobic ammonium oxidizing bacteria. Environ Microbiol 18:2784–2796. https://doi.org/10.1111/1462-2920.13134
Pynaert K et al. (2003) Characterization of an autotrophic nitrogen-removing biofilm from a highly loaded lab-scale rotating biological contactor. Appl Environ Microbiol 69:3626–3635. https://doi.org/10.1128/AEM.69.6.3626-3635.2003
Quan ZX et al. (2008) Diversity of ammonium-oxidizing bacteria in a granular sludge anaerobic ammonium-oxidizing (anammox) reactor. Environ Microbiol 10:3130–3139. https://doi.org/10.1111/j.1462-2920.2008.01642.x
Sato Y et al. (2012) Detection of anammox activity and 16S rRNA genes in ravine paddy field soil. Bull Jpn Soc Microb Ecol 27:316. https://doi.org/10.1264/jsme2.ME11330
Schloss PD (2020) Reintroducing mothur: 10 years later. Appl Environ Microbiol 86:e02343–19. https://doi.org/10.1128/AEM.02343-19
Schmid M et al. (2000) Molecular evidence for genus level diversity of bacteria capable ofcatalyzing anaerobic ammonium oxidation. Syst Appl Microbiol 23:93–106. https://doi.org/10.1016/S0723-2020(00)80050-8
Schmid M et al. (2003) Candidatus “Scalindua brodae”, sp. nov., Candidatus “Scalindua wagneri”, sp. nov., two new species of anaerobic ammonium oxidizing bacteria. Syst Appl Microbiol 26:529–538. https://doi.org/10.1078/072320203770865837
Schmid MC et al. (2007) Anaerobic ammonium-oxidizing bacteria in marine environments: widespread occurrence but low diversity. Environ Microbiol 9:1476–1484. https://doi.org/10.1111/j.1462-2920.2007.01266.x
Schubert CJ et al. (2010) Anaerobic ammonium oxidation in a tropical freshwater system (Lake Tanganyika). Environ Microbiol 8:1857–1863. https://doi.org/10.1111/j.1462-2920.2006.01074.x
Shen LD et al. (2016) Distribution and activity of anaerobic ammonium-oxidising bacteria in natural freshwater wetland soils. Appl Microbiol Biotechnol 100:3291–3300. https://doi.org/10.1007/s00253-015-7191-z
Strous M et al. (1999a) Missing lithotroph identified as new planctomycete. Nature 400:446–449. https://doi.org/10.1038/22749
Strous M, Kuenen JG, Jetten MS (1999b) Key physiology of anaerobic ammonium oxidation. Appl Environ Microbiol 65(7):3248–3250. https://doi.org/10.3171/jns.2004.100.2.0332
Sun W et al. (2014) Diversity and distribution of planktonic anaerobic ammonium-oxidizing bacteria in the Dongjiang River, China. Microbiological Res 169:897–906. https://doi.org/10.1016/j.micres.2014.05.003
ter Braak CJF, Šmilauer P (2012) Canoco reference manual and user’s guide: software for ordination (version 5.10). Microcomputer Power, Ithaca, USA
Van der Star WRL et al. (2008) The membrane bioreactor: a novel tool to grow anammox bacteria as free cells. Biotechnol Bioeng 101:286–294. https://doi.org/10.1002/bit.21891
Wang Y, Gu JD (2014) Effects of allylthiourea, salinity, and pH on ammonia/ammonium-oxidizing prokaryotes in mangrove sediment incubated in laboratory microcosms. Appl Microbiol Biotechnol 98:3257. https://doi.org/10.1007/s00253-013-5399-3
Wang Y et al. (2012) Co-occurrence and distribution of nitrite-dependent anaerobic ammonium and methane-oxidizing bacteria in a paddy soil. Fems Microbiol Lett 336:79–88. https://doi.org/10.1111/j.1574-6968.2012.02654.x
Woebken D et al. (2008) A microdiversity study of anammox bacteria reveals a novel Candidatus Scalindua phylotype in marine oxygen minimum zones. Environ Microbiol 10:3106–3119. https://doi.org/10.1111/j.1462-2920.2008.01640.x
Wu JP et al. (2016) A rapid and high-throughput microplate spectrophotometric method for field measurement of nitrate in seawater and freshwater. Sci Rep. 6:20165. https://doi.org/10.1038/srep20165
Wu JP et al. (2019) Unexpectedly high diversity of anammox bacteria detected in deep-sea surface sediments of the South China Sea. Fems Microbiology Ecology 95. https://doi.org/10.1093/femsec/fiz013
Wu JP et al. (2018) Anaerobic ammonium oxidation in acidic red soils. Front Microbiol 9:2142. https://doi.org/10.3389/fmicb.2018.02142
Yang XR et al. (2017) An overlooked nitrogen loss linked to anaerobic ammonium oxidation in estuarine sediments in China. J Soils Sediment 17(5):1–10. https://doi.org/10.1007/s11368-017-1728-y
Yin KD, Harrison PJ (2007) Influence of the Pearl River estuary and vertical mixing in Victoria Harbor on water quality in relation to eutrophication impacts in Hong Kong waters. Mar Pollut Bull 54:646–656. https://doi.org/10.1016/j.marpolbul.2007.03.001
Zheng YL et al. (2016a) Community composition and activity of anaerobic ammonium oxidation bacteria in the rhizosphere of salt-marsh grass Spartina alterniflora. Appl Microbiol Biotechnol 100:8203–8212. https://doi.org/10.1007/s00253-016-7625-2
Zheng YL et al. (2016b) Shifts in the community structure and activity of anaerobic ammonium oxidation bacteria along an estuarine salinity gradient. J Geophys Res Biogeosciences 121:6. https://doi.org/10.1002/2015JG003300
Zhu GB et al. (2013) Hotspots of anaerobic ammonium oxidation at land-freshwater interfaces. Nat Geosci 6:103–107. https://doi.org/10.1038/ngeo1683
Acknowledgements
Our research was supported by the National Natural Science Foundation of China [No. 31870100 and No. 91851111], Natural Science Foundation of Guangdong Province [No. 2019B1515120066], and Research Foundation for Talented Scholars of Guangzhou University [No. GU2017001]. We would like to thank Shuai Chen, Guangshi Chen and Tianzheng Huang for assistance in measuring environmental parameters.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Li, Y., Hong, Y., Wu, J. et al. Spatial variability pattern of the anaerobic ammonia-oxidizing bacterial community across a salinity gradient from river to ocean. Ecotoxicology 30, 1743–1753 (2021). https://doi.org/10.1007/s10646-020-02282-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-020-02282-5