Abstract
Organophosphorus and carbamate are widely used in agricultural production. Caenorhabditis elegans is a model organism that is widely used in various toxicology studies. To understand the effects of two types of commonly used pesticides, phoxim (organophosphorus) and carbaryl (carbamate), we determined the activities of acetylcholinesterases (AChEs) and detected the expression of four ace genes by RT-qPCR in C. elegans following treatment with these pesticides. The results showed that phoxim and carbaryl could reduce acetylcholinesterase activities and up-regulate the ace-3 mRNA expression levels. We also detected the toxic effects of these pesticides on the ace-3 deletion mutant dc-2, and found that some characteristics, including LC50, development, movement, reproduction and lifespan, were reduced in the dc-2 mutant. However, the toxic effects of carbaryl were weaker than those of phoxim. Carbaryl treatment did not significantly affect the LC50, movement ability or lifespan. Interestingly, body and brood size increased with carbaryl treatment at low concentrations. These data showed that both phoxim and carbaryl could inhibit AChE but that the ace-3 was necessary for phoxim detoxification.
Graphical Abstract
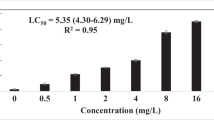
The LC50 of phoxim and carbaryl in wild type N2 and the ace-3 deletion mutant dc-2. **Higher significant differences (P < 0.01).







Similar content being viewed by others
References
Anbalagan C, Lafayette I, Antoniou-Kourounioti M, Gutierrez C, Martin JR, Chowdhuri DK, De Pomerai DI (2013) Use of transgenic GFP reporter strains of the nematode Caenorhabditis elegans to investigate the patterns of stress responses induced by pesticides and by organic extracts from agricultural soils. Ecotoxicology 22:72–85
Bao GZ, Dong DM, Li XM, Bai JR, Shen WB (2001) Progress of studies on ecological effects of ecoestrogen (in Chinese). Chin J Ecol 20:44–50
Bastiani C, Mendel J, (2006) Heterotrimeric G proteins in C. elegans. WormBook 1–25
Brinke M, Heininger P, Traunspurger W (2013) Effects of a bioassay-derived ivermectin lowest observed effect concentration on life-cycle traits of the nematode Caenorhabditis elegans. Ecotoxicology 22:148–155
Chang S, Opperman CH (1991) Characterization of acetylcholinesterase molecular forms of the root-knot nematode, Meloidogyne. Mol Biochem Parasitol 49:205–214
Chen JZ, Wang ZR, Qiu LP, Qu JH, Hu GD (2012) Research on endocrine disrupting effect of carbaryl on male tilapia (GIFT Oreochromis niloticus) (in Chinese). Asian J Ecotoxicol 6:627–632
Clark AE, Yoon S, Rebecca J, Sheesley RJ, Usenko S (2015) Pressurized liquid extraction technique for the analysis of pesticides, PCBs, PBDEs, OPEs, PAHs, alkanes, hopanes, and steranes in atmospheric particulate matter. Chemosphere 137:33–44
Cole RD, Anderson GL, Williams PL (2004) The nematode Caenorhabditis elegans as a model of organophosphate-induced mammalian neurotoxicity. Toxicol Appl Pharmacol 194:248–256
Combes D, Fedon Y, Toutant JP, Arpagaus M (2003) Multiple ace genes encoding acetylcholinesterases of Caenorhabditis elegans have distinct tissue expression. Eur J Neurosci 18:497–512
Culetto E, Combes D, Fedon Y, Roig A, Toutant JP, Arpagaus M (1999) Structure and promoter activity of the 50 flanking region of ace-1, the gene encoding acetylcholinesterase of class A in Caenorhabditis elegans. J Mol Biol 290:951–966
Culotti JG, Von Ehrenstein G, Culotti MR, Russell RL (1981) A second class of acetylcholinesterase-deficient mutants of the nematode Caenorhabditis elegans. Genetics 97:281–305
Daniela P, Jasmine S, Chen YC, Patrick A (2015) Reproductive toxicity and meiotic dysfunction following exposure to the pesticides Maneb, Diazinon and Fenarimol. Toxicol Res 4:645–654
Ellman GL, Courtney DK, Andres VJ, Feather-stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Eltzov E, Marks R (2011) Whole-cell aquatic biosensors. Anal Bioanal Chem 400:895–913
Garrity PA, Goodman MB, Samuel AD, Sengupta P (2011) Running hot and cold: behavioral strategies, neural circuits, and the molecular machinery for thermotaxis in C. elegans and Drosophila. Genes Dev 24:2365–2382
Guo YL, Yang YC, Wang DY (2009) Induction of reproductive deficits in nematode Caenorhabditis elegans exposed to metals at different developmental stages. Reprod Toxicol 28:90–95
Hoogewijs D, Houthoofd K, Matthijssens F, Vandesompele J, Vanfleteren J (2008) Selection and validation of a set of reliable reference genes for quantitative sod gene expression analysis in C. elegans. BMC Mol Biol 9:1–8
Johnson CD, Duckett JG, Culotti JG, Herman RK, Meneely PM, Russell RL (1981) An acetylcholinesterase-deficient mutant of the nematode Caenorhabditis elegans. Genetics 97:261–279
Johnson CD, Rand JR, Herman RK, Stern BD, Russell RL (1988) The acetylcholinesterase genes of C. elegans: identification of a third gene (ace-3) and mosaic mapping of a synthetic lethal phenotype. Neuron 1:165–173
Kang JS, Moon YS, Lee SH (2012) Inhibition properties of three acetylcholinesterases of the pinewood nematode Bursaphelenchus xylophilus by organophosphates and carbamates. Pestic Biochem Physiol 104:157–162
Kolson DL, Russel RL (1985) A novel class of acetylcholinesterase, revealed by mutations, in the nematode Caenorhabditis elegans. J Neurogenet 2:93–110
Kolson DL, Russell RL (1985) New acetylcholinesterase-deficient mutants of the nematode Caenorhabditis elegans. J Neurogenet 2:69–91
Lee J, Kim KY, Paik YK (2014) Alteration in cellular acetylcholine influences dauer formation in Caenorhabditis elegans. BMB Rep 47:80–85
Leomanni A, Schettino T, Calisi A, Gorbi S, Mezzelani M, Regoli F, Lionetto MG (2015) Antioxidant and oxidative stress related responses in the Mediterranean land snail Cantareus apertus exposed to the carbamate pesticide Carbaryl. Comp Biochem Physiol Part C 168:20–27
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real time quantitative PCR and the \( 2^{{ - \varDelta \varDelta C_{t} }} \) method. Methods 25:402–408
Matsumura F (1985) Toxicology of Insecticides, 2nd edn. Plenum Press, New York, p 368
Opperman CH, Chang S (1992) Nematode acetylcholinesterases: molecular forms and their potential role in nematode behavior. Parasitol Today 8:406–411
Qiu Y, Chen JF, Song L, He J, Liu R, Zhang CW, Wang XR (2005) Effect of carbaryl on serum steroid hormone and the function of antioxidant system in female rats (in Chinese). Chin J Ind Hyg Occup Dis 23:290–293
Reinke V, Smith HE, Nance J, Wang J, Van Doren C, Begley R, Jones SJM, Davis EB, Scherer S, Ward S, Kim SK (2000) A global profile of germline gene expression in C. elegans. Mol Cell 6:605–616
Ritter L, Solomon K, Sibley P, Hall K, Keen P, Mattu G, Linton B (2002) Sources, pathways, and relative risks of contaminants in surface water and groundwater: a perspective prepared for the Walkerton inquiry. J Toxicol Environ Health Part A 65:1–142
Robatzek M, Thomas JH (2000) Calcium/calmodulin-dependent protein kinase II regulates Caenorhabditis elegans locomotion in concert with a G(o)/G(q) signaling network. Genetics 156:1069–1082
Roh JY, Choi J (2011) Cyp35a2 gene expression is involved in toxicity of fenitrothion in the soil nematode Caenorhabditis elegans. Chemosphere 84:1356–1361
Roh JY, Lee J, Choi J (2006) Assessment of stress—related gene expression in the heavy metal—exposed nematode Caenorhabditis elegans: a potential biomarker for metal-induced toxicity monitoring and environmental risk assessment. Environ Toxicol Chem 25:2946–2956
Selkirk ME, Lazari O, Hussein AS, Matthewsd JB (2005) Nematode acetylcholinesterases are encoded by multiple genes and perform non-overlapping functions. Chem Biol Interact 157:263–268
Song SJ, Zhang XY, Wu HH, Han Y, Zhang JZ, Ma EB, Guo YP (2014) Molecular basis for antioxidant enzymes in mediating copper detoxification in the nematode Caenorhabditis elegans. PLoS ONE 9(9):e107685
Tatara CP, Newman MC, McCloskey JT, Williams PL (1998) Use of ion characteristics to predict relative toxicity of mono-, di- and trivalent metal ions: Caenorhabditis elegans LC50. Aquat Toxicol 42:255–269
Toumi H, Burga-Perez KF, Ferard JF (2016) Acute and chronic ecotoxicity of carbaryl with a battery of aquatic bioassays. J Environ Sci Health B 51:57–62
Tsalik EL, Hobert O (2003) Functional mapping of neurons that control locomotory behavior in Caenorhabditis elegans. J Neurobiol 56:178–197
Von Stetina SE, Mango SE (2015) PAR-6, but not E-cadherin and beta-integrin, is necessary for epithelial polarization in C. elegans. Dev Biol 403:5–14
Wang DY, Wang Y (2008) Nickel sulfate induces numerous defects in Caenorhabditis elegans that can also be transferred to progeny. Environ Pollut 151:585–592
Wang DY, Xing XJ (2008) Assessment of locomotion behavioral defects induced by acute toxicity from heavy metal exposure in nematode Caenorhabditis elegans. J Environ Sci 20:1132–1137
Wang YH, Wang JM, Peng GD, Sun BX, Li B, Shen WD (2011) Gene expression analysis from phoxim-induced domesticated silkworm (Bombyx mori) by whole-genome oligonucleotide microarray. Pestic Biochem Physiol 101:48–52
Wang BB, Li FC, Ni M, Zhang H, Xu KZ, Tian JH, Hu JS, Shen WD, Li B (2015) Molecular signatures of reduced nerve toxicity by CeCl3 in phoxim exposed silkworm brains. Sci Rep 5:1–9
Ware GW, Whitacre DM (2004) The pesticide book, 6th edn. Meister Media Worldwide, Willoughby
Xie Y, Wang B, Li F, Ma L, Ni M, Shen W, Hong F, Li B (2014) Molecular mechanisms of reduced nerve toxicity by titanium dioxide nanoparticles in the phoxim-exposed brain of Bombyx mori. PLoS ONE 9(6):e101062
Zhu KY, Clark JM (1994) Purification and characterization of acetyl-cholinesterase from the Colorado potato beetle, Leptinotarsa decemlineata (Say). Insect Biochem Mol Biol 24:453–461
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31320103921, 31172161) and the Outstanding Graduate Science Innovation Foundation of Shanxi Province (Grant No. 021852901008).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yan Han and Shaojuan Song have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Han, Y., Song, S., Guo, Y. et al. ace-3 plays an important role in phoxim resistance in Caenorhabditis elegans . Ecotoxicology 25, 835–844 (2016). https://doi.org/10.1007/s10646-016-1640-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-016-1640-z