Abstract
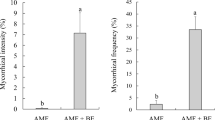
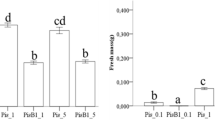
Effects of mycorrhization with Amanita rubescens or Hebeloma sinapizans and dual inoculation with the fungi and ectomycorrhiza associated bacteria (EMAB) Pseudomonas putida or Bacillus cereus on seedling growth and accumulation of Cd(II) in Pinus sylvestris were studied. Both fungal and bacterial species were isolated from roots of pines growing in an industrial area polluted with high concentrations of heavy metals. During mycorrhization, A. rubescens colonized higher number of pine seedlings than H. sinapizans, especially when EMAB were co-inoculated. In addition, the seedling biometric characteristics (i.e. root and shoot lengths and biomass) were stimulated by treatment with the fungal species alone and dual inoculation with the fungi and EMAB. Amanita rubescens was more efficient in this stimulation than H. sinapizans. The increased growth of pine seedlings was especially seen for co-inoculation with P. putida. Furthermore, elevated accumulation of Cd(II), ranging from 56 μg g−1 to 72 μg g−1 dry weight, in underground parts of the inoculated seedlings was found. The seedlings treated with A. rubescens accumulated higher concentrations of the metal than those inoculated with H. sinapizans. Additional treatment of pine seedlings with P. putida resulted in the higher accumulation of Cd(II) in the roots as compared with those inoculated with B. cereus. The results suggest that the growth of pine seedlings in Cd(II)-polluted soil may depend on fungal species forming ectomycorrhizae, species-specific co-inoculation with EMAB and specificity of fungal-EMAB interactions.


Similar content being viewed by others
References
Agerer R (2003) Color atlas of ectomycorrhiza. Einhorn-Verlag, Germany
Adriaensen K, van der Lelie D, van Laere A, Vangronsveld J, Colpaert JV (2003) A zinc-adapted fungus protect pines from zinc stress. New Phytol 161:549–555
Barea JM, Azcón R, Azcón-Aguilar C (2002) Mycorrhizosphere interactions to improve plant fitness and soil quality. Antonie van Leeuwenhoek 81:343–351
Bruins MR, Kapil S, Oehme FW (2000) Microbial resistance to metals in the environment. Ecotoxicol Environ Saf 45:198–207
Brule C, Frey-Klett P, Pierrat JC, Courrier S, Gerard F, Lemoine MC, Rousselet JL, Sommer G, Garbaye J (2001) Survival in the soil of the ectomycorrhizal fungus Laccaria bicolor and the effects of a mycorrhiza helper Pseudomonas fluorescens. Soil Biol Biochem 33:1683–1694
Collins YE, Stotzky G (1989) Factors affecting the toxicity of heavy metals to microbes. In: Beveridge TJ, Doyle RJ (eds) Metal ions and bacteria. John Willey & Sons Inc, New York pp 31–90
Colpaert JV, Van Assche JA (1992) Zinc toxicity in ectomycorrhizal Pinus sylvestris. Plant Soil 143:201–211
Colpaert JV, van Assche JA (1993) The effects of cadmium on ectomycorrhizal Pinus sylvestris L. New Phytol 123:325–333
Fomina MA, Alexander IJ, Colpaert JV, Gadd GM (2005) Solubilization of toxic metal minerals and metal tolerance of mycorrhizal fungi. Soil Biol Biochem 37:851–866
Frey-Klett P, Pierrat JC, Garbaye L (1997) Localization and survival of mycorrhizal helper Pseudomonas fluorescens during establishment of ectomycorrhizal symbiosis between Laccaria bicolor and Douglas-fir. Appl Environ Microbiol 63:139–144
Frey-Klett P, Churin JL, Pierrat JC, Garbaye J (1999) Dose effect in the dual inoculation of an ectomycorrhizal fungus and a mycorrhiza helper bacterium in two forest nurseries. Soil Biol Biochem 31:555–1562
Frey-Klett P, Chavatte M, Clausse ML, Courrier S, Le Roux C, Raaijmakers J, Martinotti MG, Pierrat JC, Garbaye J (2005) Ectomycorrhizal symbiosis affects functional diversity of rhizosphere fluorescent pseudomonads. New Phytol 165:317–328
Gadd GM (1996) Influence of microorganisms on the environmental fate of radionuclides. Endeavour 20:150–156
Galli U, Schuepp H, Brunold C (1994) Heavy metal binding by mycorrhizal fungi. Physiol Plant 92:364–368
Gamalero E, Fracchia L, Cavaletto M, Garbaye J, Frey-Klett P, Varese GC, Martinotti MG (2003) Characterization of functional traits of two fluorescent pseudomonads isolated from basidiomes of ectomycorrhizal fungi. Soil Biol Biochem 35:55–65
Garbaye J (1994) Helper bacteria: a new dimension to the mycorrhizal symbiosis. New Phytol 128:197–210
Germida JJ, Siciliano SD (2001) Taxonomic diversity of bacteria associated with the roots of modern, recent and ancient wheat cultivars. Biol Fertil Soils 33:410–415
Giller KE, Witter E, McGrath SP (1998) Toxicity of heavy metals to microorganisms and microbial processes in agricultural soils: a review. Soil Biol Biochem 30:1389–1414
Haack SK, Garchow H, Odelson DA, Forney LJ, Klug MJ (1994) Accuracy, reproducibility, and interpretation of fatty acid methyl ester profiles of model bacterial communities. Appl Environ Microbiol 60:2483–2493
Hartley JW, Cairney G, Meharg AA (1997) Do ectomycorrhizal fungi exhibit adaptive tolerance to potentially toxic metals in the environment? Plant Soil 189:303–319
Hartley JW, Cairney G, Meharg AW (1999) Cross-colonization of Scots pine (Pinus sylvestris) seedlings by the ectomycorrhizal fungus Paxillus involutus in the presence of inhibitory levels of Cd and Zn. New Phytol 142:141–149
Hartley-Whitaker JW, Cairney G, Meharg AA (2000) Toxic effects of cadmium and zinc on ectomycorrhizal colonization of Scots pine (Pinus sylvestris L.) from soil in oculum. Environ Toxicol Chem 19:694–699
Heim R (1957) Les champignons D’Europe. N. Boubée et Cie, Paris
Jentschke G, Winter S, Godbold DL (1999) Ectomycorrhizas and cadmium toxicity in Norway spruce seedlings. Tree Physiol 19:23–30
Jentschke G, Godbold DL (2000) Metal toxicity and ectomycorrhizas. Physiol Plant 109:107–116
Kawai M (1997) Artificial ectomicorrhiza formation on rots of air-layered Pinus densiflora samplings by inoculation with Lycophyllum shimei. Mycologia 89:228–232
Kim CG, Power SA, Bell JNB (2004) Response of Pinus sylvestris seedlings to cadmium and mycorrhizal colonization. Water Air Soil Pollut 155:189–203
Krupa P (2003) The role of ectomycorrhizal fungi in heavy metal polluted areas. Pol J Nat Sci 15:629–636
Krupa P, Kozdrój J (2004) Accumulation of heavy metals by ectomycorrhizal fungi colonizing birch trees growing in an idustrial desert soil. World J Microbiol Biotechnol 20:427–430
Kuek C (1996) Shake flask culture of Laccaria laccata an ectomycorrhizal basidiomycete. Microbiol Biotechnol 45:319–326
Lafranco L, Perotto S, Longato S, Mello A, Cometti V, Bonfante P (1998) Molecular approaches to investigate biodiversity in mycorrhizal fungi. In: Varma A (ed). Mycorrhizal manual, Springer-Verlag, Berlin-Heidelberg-New York, pp 353–372
Lendin M (2000) Accumulation of metals by microorganisms—processes and importance for soil systems. Earth Sci Rev 51:1–31
Leyval C, Turnau K, Haselwandter K (1997) Effect of heavy metal pollution on mycorrhizal colonization and function: physiological, ecological and applied aspects. Mycorrhiza 7:139–153
Marx DH (1969) The influence of ectotrophic mycorrhizal fungi on the resistance of pine roots to pathogenic infections. I. Antagonism of mycorrhizal fungi to root pathogenic fungi and bacteria. Phytopathology 59:153–163
Microbial ID Inc. (1999) Microbial identification system operating manual, Version 7, Newark, DE
Mogge B, Loferer C, Agerer R, Huzler P, Hartman A (2000) Bacterial community structure and colonisation patterns of Fagus sylvatica L. ectomycorrhizospheres as determinated by fluorescence in situ hybridization and confocal laser scanning microscopy. Mycorrhiza 9:271–278
Nurmiaho-Lassila EL, Timonen S, Haahtela K, Sen R (1997) Bacterial colonisation patterns of intact Pinus sylvestris mycorrhizospheres in dry pine forest soil: an electron microscopy study. Can J Microbiol 43:1017–1035
O’Neill GA, Chanway CP, Axelrood PE, Radley RA, Holl FB (1992) An assessment of spruce growth response specificity after inoculation with coexistent rhizosphere bacteria. Can J Bot 70:2347–2353
Poole EJ, Bending GD, Whipps JM, Read DJ (2001) Bacteria associated with Pinus sylvestris—Lactarius rufus ectomycorrhizas and their effects on mycorrhiza formation in vitro. New Phytol 151:743–751
Probanza A, Lucas García JA, Ruiz Palomino M, Ramos B, Gutiérrez Mañero FJ (2002) Pinus pinea L. seedling growth and bacterial rhizosphere structure after inoculation with PGPR Bacillus (B. licheniformis CECT 5106 and B. pumilus CECT 5105). Appl Soil Ecol 20:75–84
Shishido M, Massicotte HB, Chanway CP (1996) Effect of plant growth promoting Bacillus strains on pine and spruce seedling growth and mycorrhizal infection. Ann Bot 77:433–441
Van Tichelen KK, Colpaert JV, Vangronsveld J (2001) Ectomycorrhizal protection of Pinus sylvestris against copper toxicity. New Phytol 150:203–213
Vivas A, Vörös I, Biró B, Barea JM, Ruiz-Rozano JM, Azcón R (2003a) Beneficial effects of indigenous Cd-tolerant and Cd-sensitive Glomus mosseae associated with a Cd-adapted strain of Brevibacillus sp. in improving plant tolerance to Cd contamination. Appl Soil Ecol 24:177–186
Vivas A, Vörös I, Biró B, Campos E, Barea JM, Azcón R (2003b) Symbiotic efficiency of autochthonous arbuscular mycorrhizal fungus (G. mosseae) and Brevibacillus sp. isolated from cadmium polluted soil under increasing cadmium levels. Environ Pollut 126:179–189
Willenborg AD, Schmotz YJ, Lelley A (1990) Effects of environmental stress factors on ectomycorrhizal fungi in vitro. Can J Bot 68:1741–1746
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kozdrój, J., Piotrowska-Seget, Z. & Krupa, P. Mycorrhizal fungi and ectomycorrhiza associated bacteria isolated from an industrial desert soil protect pine seedlings against Cd(II) impact. Ecotoxicology 16, 449–456 (2007). https://doi.org/10.1007/s10646-007-0149-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-007-0149-x