Abstract
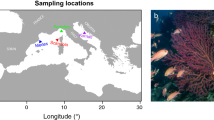
For an introduced species to expand its range, individuals must be able to adapt to novel stressors (biotic and abiotic) that they may encounter. Florida has a distinct temperature gradient and rapid climate zone transitions from warmer subtropical regions to cooler temperate regions. Mean minimum temperatures are thought to prohibit northward movement of introduced fish species in south Florida. As the introduced Belonesox belizanus (pike killifish) expands its introduced range northward, it may be gaining cold tolerance. This study sought to compare thermal tolerances and cessation of feeding between adult B. belizanus from southern (initial, introduced population) and northern (spreading, fringe edge population) populations from its introduced range. To compare thermal tolerances between locations, southern individuals and northern individuals were acclimated to three temperatures (20, 25, and 30 °C) and their upper and lower lethal temperatures and cessation of feeding temperatures were measured. Two-way ANOVAs revealed that lower lethal temperatures and cessation of feeding temperatures were significantly different between northern and southern populations, with northern individuals having a lower lethal temperature approximately 0.5 °C lower and a cessation of feeding temperature approximately 2 °C lower compared with southern individuals. Directional selection may be influencing this non-native species’ thermal tolerance and feeding ability benefitting their continued effort to disperse from subtropical to temperate environments within Florida. This study indicates that B. belizanus may be gaining cold tolerance as it continues to spread northward in its introduced range.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Allan BJM, Domenici P, Munday PL, McCormick MI (2015) Feeling the heat: the effect of acute temperature changes on predator-prey interactions in coral reef fish. Conserv Physiol 3:cov011–cov011. https://doi.org/10.1093/conphys/cov011
Al-Habbib OAM, Yacoob MP (1993) Effects of acclimation and experience to changing heat and cold shock temperature on lethal temperature and thermal tolerance of Gambusia affinis (Baird & Girard) (Poeciliidae). Cybium 17:265–272
Beaver JR, Crisman TL, Bays JS (1981) Thermal regimes of Florida Lakes. Hydrobiologia 83:267–273. https://doi.org/10.1007/BF00008277
Beitinger TL, Bennett WA, McCauley RW (2000) Temperature tolerances of North American freshwater fishes exposed to dynamic changes in temperature. Environ Biol Fish 58:237–275. https://doi.org/10.1023/A:1007676325825
Belshe JF (1961) Observations on an introduced tropical fish, Belonesox belizanus, in southern Florida. Master’s Thesis, University of Miami
Bonduriansky R, Crean AJ, Day T (2012) The implications of nongenetic inheritance for evolution in changing environments. Evolutionary Applications 5:192–201. https://doi.org/10.1111/j.1752-4571.2011.00213.x
Brett JR (1956) Some principles in the thermal requirements of fishes. Q Rev Biol 31:75–87. https://doi.org/10.1086/401257
Capps KA, Nico LG, Mendoza-Carranza M, Arévalo-Frías W, Ropicki AJ, Heilpern SA, Rodiles-Hernández R (2011) Salinity tolerance of non-native suckermouth armoured catfish (Loricariidae: Pterygoplichthys) in south-eastern Mexico: implications for invasion and dispersal. Aquat Conserv 21:528–540. https://doi.org/10.1002/aqc.1210
Carveth CJ, Widmer AM, Bonar SA (2006) Comparison of upper thermal tolerances of native and nonnative fish species in Arizona. Trans Am Fish Soc 135:1433–1440. https://doi.org/10.1577/T05-025.1
Courtenay WR Jr (1997) Nonindigenous fishes. In: Simberloff DS, Schmitz DC, Brown TC (eds) Strangers in Paradise. Island Press, Washington DC, pp 109–122
Dunker KJ (2003) Non-indigenous fishes in restored and natural wetlands on the Big Cypress Seminole Indian Reservation. Master’s Thesis, Florida Atlantic University
Eliason EJ, Clark TD, Hague MJ, Hanson LM, Gallagher ZS, Jeffries KM, Gale MK, Patterson DA, Hinch SG, Farrell AP (2011) Differences in Thermal Tolerance among sockeye salmon populations. Science 332:109–112. https://doi.org/10.1126/science.1199158
Feminella JW, Matthews WJ (1984) Intraspecific differences in thermal tolerance of Etheostoma spectabile (Agassiz) in constant versus fluctuating environments. J Fish Biol 25:455–461. https://doi.org/10.1111/j.1095-8649.1984.tb04892.x
Greenwood MFD (2012) Assessing effects of the nonindigenous Pike Killifish on indigenous fishes in Tampa Bay, Florida, using a weighted-evidence approach. T Am Fish Soc 141:84–99. https://doi.org/10.1080/00028487.2011.651552
Healy T, Schulte PM (2012) Factors affecting plasticity in whole-organism thermal tolerance in common killifish (Fundulus heteroclitus). J Comp Physiol 182:49–62. https://doi.org/10.1007/s00360-011-0595-x
Hickman GD, Dewey MR (1973) Notes on the upper lethal temperature of the duskystripe shiner, Notropis pilsbryi, and the bluegill, Lepomis macrochirus. Ttans Am Fish Soc 102:838–840. https://doi.org/10.1577/1548-8659(1973)102<838:NOTULT>2.0.CO;2
Kerfoot JR Jr (2012) Thermal tolerance of the invasive Belonesox belizanus, pike killifish, throughout ontogeny. J Exp Zool Part A 317:266–274. https://doi.org/10.1002/jez.1720
Kerfoot JR Jr, Lorenz JJ, Turingan RG (2011) Environmental correlates of the abundance and distribution of Belonesox belizanus in a novel environment. Environ Biol Fish 92:125–139. https://doi.org/10.1007/s10641-011-9822-2
Johnson T, Bennett A (1995) The thermal acclimation of burst escape performance in fish: an integrated study of molecular and cellular physiology and organismal performance. J Exp Biol 198:2165–2175. https://doi.org/10.1242/jeb.198.10.2165
Johnston IA, Temple GK (2002) Thermal plasticity of skeletal muscle phenotype in ectothermic vertebrates and its significance for locomotory behavior. J Exp Biol 205:2305–2322. https://doi.org/10.1242/jeb.205.15.2305
Jonsson B, Jonsson N (2014) Early environment influences later performance in fishes. J Fish Biol 85:151–188. https://doi.org/10.1111/jfb.12432
Jonsson B, Jonsson N (2019) Phenotypic plasticity and epigenetics of fish: embro temperature affects later-developing life-history traits. Aquat Biol 28:21–32. https://doi.org/10.3354/ab00707
Lighten J, Incarnato D, Ward BJ, van Oosterhout C, Bradbury I, Hanson M, Bentzen P (2016) Adaptive phenotypic response to climate enabled by epigenetics in a K-strategy species, the fish Leucoraja ocellate (Rajidae). Roy Soc Open Sci 3:160299. https://doi.org/10.1098/rsos.160299
Lockwood JL, Hoopes MF, Marchetti MP (2007) Chap. 1: An Introduction to Invasion Ecology. In: Lockwood JL, Hoopes MF, Marchetti MP (eds) Invasion Ecology. Blackwell Publishing, Malden, pp 1–18
Loftus WF, Ellis G, Zokan M, Lorenz J (2004) Inventory of freshwater fish species within Big Cypress National Preserve: The basis for a long-term sampling program (USGS Fact Sheet 2004–3131). USGS, U.S. Department of the Interior (US)
Marchetti MP, Moyle PB, Levine R (2004) Invasive species profiling? Exploring the characteristics of non-native fishes across invasion stages in California. Freshw Biol 49:646–661. https://doi.org/10.1111/j.1365-2427.2004.01202.x
Özbilgin H, Wardle CS (2002) Effect of seasonal temperature changes on the escape behavior of haddock, Melanogrammus aeglefinus, from the cod end. Fish Res 58:323–331. https://doi.org/10.1016/S0165-7836(01)00394-0
Podrabsky J, Riggs C, Wagner J (2015) Tolerance of environmental stress. In: Garcia G, Berois B, de Sa R (eds) Annual fishes: life history strategy, diversity, and evolution. CRC Taylor & Francis, Boca Raton, pp 159–184
Quinn GP, Keough MJ (2002) Chap. 9: Multifactor analysis of variance. In: Quinn GP, Keough MJ (eds) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge, pp 208–261
Randall D, Brauner C (1991) Effects of environmental factors on exercise in fish. J Exp Biol 160:113–126. https://doi.org/10.1242/jeb.160.1.113
Robbins LW, Hartman GD, Smith MH (1987) Dispersal, reproductive strategies, and the maintenance of genetic variability in mosquitofish (Gambusia affinis). Copeia 1:156–164. https://doi.org/10.2307/1446047
Sakai AK, Allendorf FW, Holt JS, Lodge DM, Molofsky J, With KA, Baughman S, Cabin RJ, Cohen JE, Ellstrand NC, McCauley DE, O’neil P, Parker IM, Thompson JN, Weller SG (2001) The Population Biology of Invasive Species. Annu Rev Ecol Syst 32:305–332. https://doi.org/10.1146/annurev.ecolsys.32.081501.114037
Sanford MT (2003) Florida’s climate and its beekeeping (University of Florida Fact Sheet ENY-134). University of Florida, Gainesville, FL
Schaefer JF (2006) A simple and economical method for precise, programmable thermal regimes in recirculating aquatic mesocosms. N Am J Aquacult 68:240–244. https://doi.org/10.1577/A05-069.1
Schofield PJ, Loftus WF, Kobza RM, Cook MI, Slone DH (2010) Tolerance of nonindigenous cichlid fishes (Cichlasoma urophthalmus, Hemichromis letoureuxi) to low temperature: laboratory and field experiments in south Florida. Biol Invasions 12:2441–2457. https://doi.org/10.1007/s10530-009-9654-6
Shafland PL, Pestrak JM (1982) Lower lethal temperatures for fourteen non-native fishes in Florida. Environ Biol Fish 7:149–156. https://doi.org/10.1007/BF00001785
Stauffer JR Jr (1984) Chap. 2: Colonization theory relative to introduced populations. In: Courtenay WR Jr, Stauffer JR Jr (eds) Distribution, biology and management of exotic fishes. Johns Hopkins University Press, Baltimore, pp 8–21
Stitt BC, Burness G, Burgomaster KA, Currie S, McDermid JL, Wilson CC (2014) Intraspecific variation in thermal tolerance and acclimation capacity n brook trout (Salvelinus fontinalis): Physiological implication for climate change. Physiol Biochem Zool 87:15–29. https://doi.org/10.1086/675259
Strange KT, Vokoun JC, Noltie DB (2002) Thermal tolerance and growth differences in Orangethroat darter (Etheostoma spectabile) from thermally contrasting adjoining streams. Am Midl Nat 148:120–128. https://doi.org/10.1674/0003-0031(2002)148[0120:TTAGDI]2.0.CO;2
Taylor JN, Courtenay WR Jr, McCann JA (1984) Chap. 16: Known impacts of exotic fishes in the continental United States. In: Courtenay WR Jr, Stauffer JR Jr (eds) Distribution, biology and management of exotic fishes. Johns Hopkins University Press, Baltimore, pp 322–373
Trexler JC, Loftus WF, Jordan F, Lorenz JJ, Chick JH, Kobza RM (2000) Empirical assessment of fish introductions in a subtropical wetland: an evaluation of contrasting views. Biol Invasions 2:265–277. https://doi.org/10.1023/A:1011488118444
Trexler JC, Loftus WF, Perry S (2005) Disturbance frequency and community structure in a twenty-five-year intervention study. Oecologia 145:140–152. https://doi.org/10.1007/s00442-005-0094-4
Turner JS, Snelson FF (1984) Population structure, reproduction, and laboratory behavior of the introduced Belonesox belizanus (Poeciliidae) in Florida. Environ Biol Fish 10:89–100. https://doi.org/10.1007/BF00001665
Wolter C, Röhr F (2010) Distribution history of non-native freshwater fish species in Germany: how invasive are they? J Appl Ichthyol 26:19–27. https://doi.org/10.1111/j.1439-0426.2010.01505.x
Wood ZT, Shepard ID, Hurley ST, Paisker MR, Hensley VR, Kinnison MT (2020) Sex-dependent cold tolerance at the northern invasive range limit of Gambusia affinis on Cape Cod, Massachusetts. Copeia 108:670-678. https://doi.org/10.1643/CE-19-332
Acknowledgements
I would like to thank Matt Wittenrich, Ronald Maliao, and Paul Sexton for their assistance in collecting B. belizanus from the Everglades and Big Cypress National Parks. I would also like to recognize Arthur Kunkle for his assistance in designing the computer-controlled temperature environment used for the experiments. I appreciate the valuable input that Ralph Turingan offered in implementing the experiments and the use of his laboratory space. Finally, I would like to thank Charley Kate Barcroft, Mark Bolyard, Gabi Mirabella, Darius Mullin, Dylan Parmely, Sam Ross, Tayler Schudel, and anonymous reviewers for their valuable comments on earlier drafts of this manuscript. This study was completed under Florida Institute of Technology’s Animal Care and Use Committee guidelines (IACUC Permit No. 99 − 01).
Funding
Funding in part from the Florida Institute of Technology, Graduate Research Grant.
Author information
Authors and Affiliations
Contributions
Experimentation, data collection, analysis, and manuscript writing were done by J. Kerfoot.
Corresponding author
Ethics declarations
Ethics approval
This study was completed under Florida Institute of Technology’s Animal Care and Use Committee guidelines (IACUC Permit No. 99 − 01).
Consent to participate
Not applicable.
Consent for publication
J. Kerfoot consents for publication of this manuscript.
Conflicts of interest/Competing interests
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kerfoot, J.R. Northward expansion leads to cold tolerance? Investigating thermal adaptation of the non-native pike killifish (Belonesox belizanus) in Florida. Environ Biol Fish 105, 487–497 (2022). https://doi.org/10.1007/s10641-022-01248-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-022-01248-2