Abstract
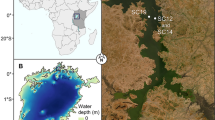
Anthropogenic perturbations and climate change have altered the zooplankton community structure in the Klang Strait during the past 30 years, in that the taxa of large-bodied crustaceans (Acartiidae, Calanidae, Pseudodiaptomidae) are being replaced by those of small-bodied crustaceans (Oithonidae, Ectinosomatidae), gelatinous jellyfish, and appendicularians. Since zooplankton constitutes the main larval food, we questioned: have bottom-up effects impacted larval fish feeding via the food chain? Larval fish that were sampled previously (1985–1986) and nearly three decades thereafter (2013–2014) were analysed for their dietary composition. Despite the dramatic replacements of zooplankton taxa due to escalating anthropogenic disturbances, the dominant copepod families, Paracalanidae, Oithonidae and Euterpinidae, remain the major prey for fish larvae. Dietary shifts in prey composition from before to after impact depend on the larval fish family and their ontogenetic stage. Dietary changes are observed in the Bregmacerotidae, Engraulidae, Gobiidae and Sciaenidae that opportunistically feed on the small-bodied copepods (oithonids and Parvocalanus crassirostris), whereas the Callionymidae, Clupeidae and Cynoglossidae naturally feed on these copepods even before these prey become numerically dominant with anthropogenic disturbance. There is no dietary shift in the Leiognathidae, exceptional in that they are specialists feeding mainly on detritus and polychaete larvae. Since the bottom-up effects are not comprehensive among fish families and dietary plasticity is evident, it is postulated that only the intolerant or non-adaptable larval species are adversely affected by the environmental perturbations.






Similar content being viewed by others
Data availability
Additional data related to this study are available upon reasonable request.
References
Anderson, MJ, Gorley RN, Clarke KR (2008) PERMANOVA for PRIMER: Guide to Software and Statistical Methods. PRIMER-E, Plymouth, UK
Ara R, Arshad A, Amin SMN, Mazlan AG, Rahman MA (2013) Feeding ecology and nursery of marine larval fishes. Asian Journal of Animal and Veterinary Advances 8:266–274. https://doi.org/10.3923/ajava.2013.266.274
Arthur DK (1976) Food and feeding of larvae of three fishes occurring in the California current, Sardinops sagax, Engraulis mordax, and Trachurus symmetricus. Fish Bull 74:517–530
Azila A, Chong VC (2010) Multispecies impingement in a tropical power plant, Straits of Malacca. Mar Environ Res 70:13–25. https://doi.org/10.1016/j.marenvres.2010.02.004
Baum JK, Worm B (2009) Cascading top-down effects of changing oceanic predator abundances. J Anim Ecol 78:699–714. https://doi.org/10.1111/j.1365-2656.2009.01531.x
Beaugrand G, Reid PC, Ibanez F, Lindley JA, Edwards M (2002) Reorganization of North Atlantic marine copepod biodiversity and climate. Science 296:1692–1694. https://doi.org/10.1126/science.1071329
Beaugrand G, Brander K, Souissi S, Reid P (2003) Plankton effect on cod recruitment in the North Sea. Nature 426. https://doi.org/10.1038/nature02164
Boglione C, Gisbert E, Gavaia P, Witten PE, Moren M, Fontagné S, Koumoundouros G (2013) Skeletal anomalies in reared European fish larvae and juveniles. Part 2: main typologies, occurrences and causative factors. Rev Aquac 5:S121–S167. https://doi.org/10.1111/raq.12016
Bowles RR, Merriner JV, Grant GC (1978) Factors associated with accuracy in sampling fish eggs and larvae. Special scientific report; no 89. Virginia Institute of Marine Science, College of William and Mary. https://doi.org/10.21220/m2-5hjz-km96
Boxshall GA, Hasley SH (2004) An introduction to copepod diversity. Ray Society, London
Bray RJ, Curtis JT (1957) An ordination of the upland forest communities of southern Wisconsin. Ecol Monogr 27:325–349. https://doi.org/10.2307/1942268
Campfield PA, Houde ED (2011) Ichthyoplankton community structure and comparative trophodynamics in an estuarine transition zone. Fishery Bulletin 109:1–19. http://aquaticcommons.org/id/eprint/8720
Chargulaf CA, Kruck NC, Tibbetts IR (2011) Does sympatry affect trophic resource use in congeneric tidepool fishes? A tale of two gobies Favonigobius lentiginosus and Favonigobius exquisitus. J Fish Biol 79:1968–1983. https://doi.org/10.1111/j.1095-8649.2011.03132.x
Chew LL, Chong VC, Tanaka K, Sasekumar A (2012) Phytoplankton fuel the energy flow from zooplankton to small nekton in turbid mangrove waters. Mar Ecol Prog Ser 469:7–24. https://doi.org/10.3354/meps09997
Chew LL, Chong VC, Wong RCS, Lehette P, Ng CC, Loh KH (2015) Three decades of sea water abstraction by Kapar power plant (Malaysia): what impacts on tropical zooplankton community? Mar Pollut Bull 101:69–84. https://doi.org/10.1016/j.marpolbul.2015.11.022
Chew LL, Chong VC (2016) Response of marine copepods to a changing tropical environment: winners, losers and implications. PeerJ 4:e2052. https://www.ncbi.nlm.nih.gov/pubmed/27257540
Chu C, Chew LL, Chong VC, Ng CC, Ooi AL, Loh KH (2021) Effect of coastal development on larval fish abundance in Klang Strait (Malaysia). Regional Studies in Marine Science 46:101889
Cushing DH (1975) Marine ecology and fisheries. Cambridge University Press, New York
Duque G, Acero PA (2003) Feeding selectivity of Anchovia clupeoides (Pisces: Engraulidae) in the Cienaga Grande de Santa Marta, Colombian Caribbean. Gulf and Caribbean Research 15:21–26. https://doi.org/10.18785/gcr.1501.04
Durant JM, Molinero J, Ottersen G, Reygondeau G, Stige LC, Langangen O (2019) Contrasting effects of rising temperatures on trophic interactions in marine ecosystems. Scientific Reports 9:15213. https://www.nature.com/articles/s41598-019-51607-w
Edwards M, Richardson AJ (2004) The impact of climate change on the phenology of the plankton community and trophic mismatch. Nature 430:881–884
FRI (2018) Fisheries Resources Surveys in Malaysian Waters 2013–2016 Volume I: Demersal. Department of Fisheries Malaysia. Ministry of Agriculture & Agro-Based Industry Malaysia
Harold AS, Johnson RK (2016) Bregmacerotidae. In: Carpenter KE, Angelis ND (ed) The living marine resources of the Eastern Central Atlantic. Vol. 3: Bony fishes part 1 (Elopiformes to Scorpaeniformes. FAO Species Identification Guide for Fishery Purposes. FAO, Rome, pp 1957–1958
Haroon AKY, Pittman KA (1999) Development of a new model of feeding strategy analysis of fish incorporating resource availability and use data. Bangladesh Journal of Fisheries Research 3:201–208
Hernandez JFJ, Carassou L, Graham WM, Powers SP (2013) Evaluation of the taxonomic sufficiency approach for ichthyoplankton community analysis. Mar Ecol Prog Ser 491:77–90. https://doi.org/10.3354/meps10475
Herwig BR, Zimmer KD (2007) Population ecology and prey consumption by fathead minnows in prairie wetlands: importance of detritus and larval fish. Biology of Freshwater Fish 16:282–294. https://doi.org/10.1111/j.1600-0633.2006.00220.x
Hunter JR (1980) The feeding behavior and ecology of marine fish larvae. In Bardach JE, Magnuson JJ, May RC, Reinhart JM (ed) Fish behavior and its use in the capture and culture of fishes, ICLARM Conference Proceedings, pp. 87–330
Hyslop EJ (1980) Stomach content analysis—a review of methods and their application. J Fish Biol 17:411–429. https://doi.org/10.1111/j.1095-8649.1980.tb02775.x
Ivlev VS (1961) Experimental ecology of the feeding of fishes. Yale University Press, New Haven, Connecticut
Kimmerling N, Zuquet O, Gurevich T, Armoza-Zvuloni A, Kolesnikov I, Berenshtein I, Melamed S, Gilad S, Benjamin S, Rivlin A, Ohavia M, Paris CB, Holzman R, Kiflawi M, Sorek R (2018) Quantitative species-level ecology of reef fish larvae via metabarcoding. Nature Ecology and Evolution 2:306–316. https://www.nature.com/articles/s41559-017-0413-2
Kulbicki M, Morize E, Wantiez L (2009) Sypnosis of the biology and ecology of Pomadasys argenteus (Haemulidae) in New Caledonia. Cybium: International Journal of Ichthyology 33:45–59. https://doi.org/10.26028/cybium/2009-331-006
Kume G, Furumitsu K, Nakata H, Suzuki T, Handa M, Yamaguchi A (2015) Spatiotemporal occurrence and feeding habits of tonguefish, Cynoglossus lighti Norman, 1925, larvae in Ariake Bay, Japan. J Appl Ichthyol 31:276–281. https://doi.org/10.1111/jai.12498
Lee CW, Bong CW (2006) Carbon flux through bacteria in a eutrophic tropical environment: Port Klang waters. In Wolanski E (ed) The environment in Asia Pacific Harbour, chapter 20. Dordrecht: Springer, pp. 329–345. https://doi.org/10.1007/1-4020-3655-8_20
Lee SL, Chong VC, Then AY (2019) Fish trophodynamics in tropical mudflats: a dietary and isotopic perspective. Estuaries Coasts 42:868–889. https://doi.org/10.1007/s12237-019-00519-z
Leis JM, Carson-Ewart BM, (2004) The larvae of Indo-Pacific coastal fishes: a guide to identification. Fauna Malesiana Handbook 2. Brill: Leiden.
Leis JM (2015) Taxonomy and systematics of larval Indo-Pacific fishes: a review of progress since 1981. Ichthyol Res 62:9–28. https://doi.org/10.1007/s10228-014-0426-7
Lim JH, Lee CW (2017) Effects of eutrophication on diatom abundance, biovolume and diversity in tropical coastal waters. Environmental monitoring and assessment 189. https://www.x-mol.com/paperRedirect/1212917492580425751
Lima ARA, Barletta M (2016) Lunar influence on prey availability, diet shifts and niche overlap between Engraulidae larvae in tropical mangrove creeks. J Fish Biol 89:2133–2152. https://doi.org/10.1111/jfb.13121
Llopiz JK (2013) Latitudinal and taxonomic patterns in the feeding ecologies of fish larvae: a literature synthesis. J Mar Syst 109:69–77. https://doi.org/10.1016/j.jmarsys.2012.05.002
Lynam CP, Llope M, Mollmann C, Helaouet P, Bayliss-Brown GA, Stenseth NC (2017) Interaction between top-down and bottom-up control in marine food webs. PNAS 114:1952–1957. https://doi.org/10.1073/pnas.1621037114
Morote E, Olivar MP, Villate F, Uriarte I (2010) A comparison of anchovy (Engraulis encrasicolus) and sardine (Sardina pilchardus) larvae feeding in the Northwest Mediterranean: influence of prey availability and ontogeny. ICES J Mar Sci 67:897–908. https://doi.org/10.1093/icesjms/fsp302
MPP-EAS (1999) Total economic valuation: coastal and marine resources in the Straits of Malacca. MPP-EAS Technical Report No. 24 Philippines: GEF/UNDP/IMO.
Nevejan N, Schryver PD, Willie M, Dierckerns K, Baruah K, Stappen GV (2016) Bacteria as food in aquaculture: do they make a difference? Rev Aquac 10:180–212. https://doi.org/10.1111/raq.12155
Nichols JH, Thompson AB (1991) Mesh selection of copepodite and nauplius stages of four calanoid copepod species. J Plankton Res 13:661–671. https://doi.org/10.1093/plankt/13.3.661
Nogales B, Lanfranconi MP, Pina-Villalonga JM, Bosch R (2011) Anthropogenic perturbations in marine microbial communities. FEMS Microbiol Rev 35:275–298. https://doi.org/10.1111/j.1574-6976.2010.00248.x
Nunn AD, Harvey JP, Cowx IG (2007) The food and feeding relationships of larval and 0+ year juvenile fishes in lowland rivers and connected waterbodies. I. Ontogenetic shifts and interspecific diet similarity. J Fish Biol 70:726–742. https://doi.org/10.1111/j.1095-8649.2007.01335.x
Ocana-Luna A, Sanchez-Ramirez M (1998) Feeding of Sciaenid (Pisces: Sciaenidae) larvae in two coastal lagoons of the Gulf of Mexico. Gulf Research Reports 10:1–9. https://doi.org/10.18785/grr.1001.01
Okiyama M (2014) An atlas of early stage fishes in Japan, 2nd edn. Tokai University Press, Hadano
Ooi AL, Chong VC (2011) Larval fish assemblages in a tropical mangrove estuary and adjacent coastal waters: offshore-inshore flux of marine and estuarine species. Cont Shelf Res 31:1599–1610. https://doi.org/10.1016/j.csr.2011.06.016
Østergaard P, Munk P, Janekarn V (2005) Contrasting feeding patterns among species of fish larvae from the tropical Andaman Sea. Mar Biol 146:595–606. https://doi.org/10.1007/s00227-004-1458-8
Peck MA, Huebert KB, Llopiz JK (2012) Intrinsic and extrinsic factors driving match-mismatch dynamics during the early life history of marine fishes. Adv Ecol Res 47:177–302. https://doi.org/10.1016/B978-0-12-398315-2.00003-X
Peters KM, McMichael RH Jr(1987) Early life history of the red drum, Sciaenops ocellatus (Pisces: Sciaenidae) in Tampa Bay, Florida. Estuaries 10:92–107. https://doi.org/10.2307/1352173
Philips NW (1984) Role of different microbes and substrates as potential suppliers of specific, essential nutrients to marine detritivores. Bull Mar Sci 35:283–298
Pinkas L, Oliphant MS, Inverson ILK (1971) Food habits of albacore, bluefin tuna and bonito in California waters. Fish Bull 152:11–105
Przeslawski R, Byrne M, Mellin C (2015) A review and meta-analysis of the effects of multiple abiotic stressors on marine embryos and larva. Glob Change Biol 21:2122–2140. https://doi.org/10.1111/gcb.12833
Rice E, Dam HG, Stewart G (2014) Impact of climate change on estuarine zooplankton: surface water warming in Long Island Sound is associated with changes in copepod size and community structure. Estuaries Coasts 38:13–23. https://doi.org/10.1007/s12237-014-9770-0
Robert D, Murphy HM, Jenkins GP, Fortier L (2014) Poor taxonomical knowledge of larval fish prey preference is impeding our ability to assess the existence of a “critical period” driving year-class strength. ICES J Mar Sci 71:2042–2052. https://doi.org/10.1093/icesjms/fst198
Sampey A, McKinnon AD, Meekan MG, McCormick MI (2007) Glimpse into guts: overview of the feeding of larvae of tropical shorefishes. Mar Ecol Prog Ser 339:243–257. https://doi.org/10.3354/meps339243
Sánchez-Velasco L (1998) Diet composition and feeding habits of fish larvae of two co-occurring species (Pisces: Callionymidae and Bothidae) in the North-western Mediterranean. ICES J Mar Sci 55:299–308. https://doi.org/10.1006/jmsc.1997.0278
Sany SBT, Salleh A, Sulaiman AH, Sasekumar A, Rezayi M, Tehrani GM (2013) Heavy metal contamination in water and sediment of the Port Klang coastal area, Selangor, Malaysia. Environmental Earth Sciences 69:2013–2025. https://doi.org/10.1007/s12665-012-2038-8
Sasekumar A, Chong VC, Leh MU, D’Cruz R (1992) Mangroves as a habitat for fish and prawns. Hydrobiologia 247:195–207. https://doi.org/10.1007/BF00008219
Sasekumar A, Then AYH, Moh HH (2012) Vegetation, forestry and conservation of Selangor mangrove forests. In Sasekumar A, Chong VC Mangrove and coastal environment of Selangor, Malaysia. Monograph Series, 14. Institute of Ocean and Earth Science, University of Malaya, pp 55–65
Sinclair M, Tremblay MJ (1984) Timing and spawning of Atlantic herring (Clupea harengus harengus) populations and the match-mismatch theory. Can J Fish Aquat Sci. https://doi.org/10.1139/f84-123
Smith PE, Richardson SL (1977) Standard techniques for pelagic fish eggs and larvae survey. FAO, Rome
Stoecker D, Pierson J (2019) Predation on protozoa: its importance to zooplankton revisited. J Plankton Res 41:367–373. https://doi.org/10.1093/plankt/fbz027
Sutton TM, Bowen SH (1994) Significance of organic detritus in the diet of larval lampreys in the Great Lakes Basin. Canadian Journal of Fisheries and Aquatic Science 51:2380–2387. https://doi.org/10.1139/f94-239
Tanaka K, Hanamura Y, Chong VC, Watanabe S, Man A, Faizul MK, Kodama M, Ichikawa T (2011) Stable isotope analysis reveals ontogenetic migration and the importance of a large mangrove estuary as a feeding ground for juvenile John’s snapper Lutjanus johnii. Fish Sci 77:809–816. https://doi.org/10.1007/s12562-011-0396-x
Ter Braak CJF, Smilauer P (2002) CANOCO reference manual and CanoDraw for Windows user’s guide: software for canonical community ordination (Version 4.5). Microcomputer Power, Ithaca, New York, 500 pp
Teoh HW, Lee SL, Chong VC, Yurimoto T (2016) Nutrient (N, P, Si) concentration and primary production on a perturbed tropical coastal mudflat. Environmental Earth Sciences 75:1147. https://doi.org/10.1007/s12665-016-5953-2
Terlizzi A, Benedetti-Cecchi L, Bevilacqua S, Fraschetti S, Guidetti P, Anderson MJ (2005) Multivariate and univariate asymmetrical analyses in environmental impact assessment: a case study of Mediterranean subtidal sessile assemblages. Mar Ecol Prog Ser 289:27–42. https://doi.org/10.3354/meps289027
Turner JT (2004) The importance of small planktonic copepods and their roles in pelagic marine food webs. Zoological Studies 43:255–266
Vargas CA, Martinez RA, Cuevas LA, Pavez MA, Cartes C, González HE, Escribano R, Daneri G (2017) The relative importance of microbial and classical food webs in a highly productive coastal upwelling area. Limnol Oceanogr 52:1495–1510. https://doi.org/10.4319/lo.2007.52.4.1495
Walsh HJ, Richardson DE, Marancik KE, Hare JA (2015) Long-term changes in the distributions of larval and adult fish in the northeast U.S. shelf ecosystem. PloS ONE 10. https://doi.org/10.1371/journal.pone.0137382
Wilson SK, Bellwood DR, Choat HJ, Furnas MJ (2003) Detritus in the epilithic algal matrix and its use by coral reef fishes. Oceanogr Mar Biol 41:279–309
Young CM, Sewell MA, Rice ME (2002) Atlas of Marine Invertebrate Larvae. Academic Press, California
Acknowledgements
We thank the University of Malaya for providing research facilities. We would also like to thank Dr. Konishi Yoshinobu for helping with the identification of some of the larval fish specimens. We acknowledge Goh Hao Chin for microtechnique assistance.
Funding
This study was funded by research grants from the Universiti Tunku Abdul Rahman awarded to Ooi AL (Research Fund (UTARRF) Cycle 2016/1), and the Ministry of Education Malaysia awarded to Chong VC (HIR grant no. H-21001–00-F000023).
Author information
Authors and Affiliations
Contributions
Ooi AL conceived study conception and design. Ooi AL and Chong VC obtained funding. Ooi AL and Teoh CY supervised the findings of this work. Chew LL and Chong VC carried out the field collection. Chew LL and Chu C did taxonomy identification. Chu C and Quah WC did the laboratory examinations. Quah WC performed data collection and analysis, and wrote the first draft of the manuscript. Chew LL, Chong VC and Ooi AL helped improved the manuscript. All authors discussed the results and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study is based on archived, formalin-preserved samples that do not need the approval of an animal care or ethics committee.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Quah, W.C., Chew, L.L., Chong, V.C. et al. Does structural change in the zooplankton community affect larval fish feeding in anthropogenically disturbed tropical waters?. Environ Biol Fish 105, 55–76 (2022). https://doi.org/10.1007/s10641-021-01189-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-021-01189-2