Abstract
Smallmouth bass (Micropterus dolomieu) predation on subyearling fall Chinook salmon (Oncorhynchus tshawytscha) was examined in the Snake River (USA) to identify seasonal and habitat-related changes in bass diets, and associated subyearling consumption and loss in various riverine and impounded reaches. Smallmouth bass diets reflected opportunistic foraging that at times showed predation on subyearlings is influenced by the consumption of other prey such as crayfish, sand roller (Percopsis transmontana), and smaller invertebrates. Estimated loss of subyearlings was influenced by bass abundance and consumption rates. The highest bass abundances (> 1,000 bass/river kilometer) were observed in the upper reach of Hells Canyon early in April and May, and in Lower Granite Reservoir. Peak consumption rates of subyearlings (≥ 0.12 subyearlings/bass/day) occurred in the upper reach of Hells Canyon during May and in most reservoir reaches in June. Predation losses accumulated evenly along the river continuum from riverine to reservoir habitats. We estimated that 869,371 subyearlings could be lost to smallmouth bass predation between riverine production areas and Lower Granite Dam in a given year. To provide a context for this estimated loss, we provide an illustration of its potential effect on the adult population. Assuming no juvenile mortality occurred downstream of the dam and depending on smolt-to-adult return rates, this represented up to 3.9–16.0% of the potential adult run that could have returned to Lower Granite Dam had no subyearling predation by smallmouth bass occurred upstream of the dam. Although this study was limited by a number of assumptions and constraints, it does provide an illustration of how predation affects juvenile and adult salmon loss over a broad, changing river landscape.
Similar content being viewed by others
Introduction
Predation is a significant source of mortality for juvenile salmon and is commonly cited as a contributing factor to the decline of anadromous salmonids in the Pacific Northwest of the United States (Sanderson et al. 2009; Carey et al. 2011). Predation on juvenile salmonids by both piscine and avian predators has been studied extensively in anadromous fish-bearing waters along the west coast of the United States and Canada (Rieman et al. 1991; Gregory and Levings 1998; Furey et al. 2016; Grossman 2016; Sabal et al. 2016; Evans et al. 2012), but past efforts have generally focused on specific areas like river transitional areas (Tabor et al. 1993; Shively et al. 1996; Erhardt et al. 2018a), river reaches (Fritts and Pearsons 2004), or to address ecological questions [e.g., binge feeding (Collis et al. 1995; Furey et al. 2016; Erhardt and Tiffan 2018)]. One reason for this is likely the cost and effort required to estimate predation losses at larger spatial scales. Because many juvenile salmonids emigrate across great distances and through varied habitats during seaward emigration, localized predation assessments may not capture the variation in predation risk and loss across larger spatial scales. A notable exception, however, is the work of Ward et al. (1995) who developed indices of northern pikeminnow (Ptychocheilus oregonensis) predation on juvenile salmonids for much of the impounded Snake and Columbia rivers.
Predation likely varies along a river continuum in which habitats, food webs, and predator abundance change resulting in variable predation risk and loss for prey moving along that continuum. For example, Rieman et al. (1991) found that northern pikeminnow abundance was highest in the upper end of John Day Reservoir below McNary Dam on the Columbia River and exacted heavy predation losses on juvenile salmon (Oncorhynchus spp.) in that area, but predator abundance and estimated losses were lower elsewhere in the reservoir. Similarly, abundances of smallmouth bass (Micropterus dolomieu), channel catfish (Ictalurus punctatus), walleye (Sander vitreus), and northern pikeminnow varied spatially within the Snake and Columbia rivers that resulted in variable predation rates on juvenile salmonids (Ward et al. 1995; Zimmerman and Parker 1995). Food webs also change with river hydrology, particularly between lotic and lentic habitats (e.g., Vannote et al. 1980), and changes in prey assemblages will influence predation on target species by opportunistic predators (Poe et al. 1991; Hemingway et al. 2019). Given the fact that the Snake River encompasses such landscape-level changes, we believed this system would be well-suited to studying how predation on juvenile salmon changes along a diverse river continuum.
The Snake River within portions of Idaho, Oregon, and Washington changes from an unimpounded, but regulated, state within Hells Canyon to being fully impounded by Lower Granite Dam (Fig. 1). The river supports an Endangered Species Act (ESA)-listed population of fall Chinook salmon (O. tshawytscha) that is also supplemented with annual releases of hatchery fish. During rearing, juveniles continually disperse downstream along mainstem shorelines through both unimpounded and impounded habitats for extended periods before moving offshore to emigrate seaward as age-0 fish (hereafter, subyearlings) by early summer (Connor et al. 2003). Their relatively small size and extended shoreline rearing makes subyearlings vulnerable to predation by smallmouth bass, which are present throughout the river system. Although several past studies have examined smallmouth bass predation on juvenile salmon in specific locations in the Snake River (Nelle 1999; Anglea 1997; Naughton et al. 2004; Erhardt and Tiffan 2018; Erhardt et al. 2018a), none has estimated predation losses over a broader landscape to identify how changes in river habitats and prey assemblages affect predation losses. Furthermore, these and other studies typically report numbers of juvenile salmon lost to predation but rarely is any effort made to put those losses in a population-level context. Because declines in salmon populations are often measured by the number of returning adults, expressing juvenile predation losses in terms of foregone adult production provides an indication of the effect of predation on a population.
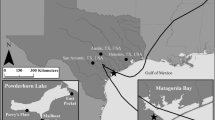
A map of the Snake River and reaches sampled to describe smallmouth bass diets and estimate predation losses of subyearling fall Chinook salmon during 2013–2018. Reach boundaries are denoted by circles with associated river kilometer (rkm), and reaches are abbreviated as Upper HC (upper Hells Canyon, rkm 323–369), Middle HC (middle Hells Canyon, rkm 272–303), Lower HC (lower Hells Canyon, rkm 234–272), SRTZ (Snake River transition zone, rkm 224–234), CON (confluence, rkm 217–224), Upper Res (upper Lower Granite Reservoir, rkm 201–217), Middle Res (middle Lower Granite Reservoir, rkm 187–201), and Lower Res (lower Lower Granite Reservoir, rkm 173–187). The flow of the Snake River is from south to north
In this paper, we pooled information from a number of our recent studies to examine smallmouth bass predation on subyearling fall Chinook salmon along an unimpounded to impounded river continuum. Specifically, we sought to link seasonal, habitat-related changes in smallmouth bass diets to the incidence of subyearling consumption by bass. Second, we estimated cumulative subyearling predation mortality from natal rearing areas to Lower Granite Dam to identify areas and times of greatest predation. Finally, we expressed the total cumulative loss of subyearlings in terms of the number of adults that would be lost to production to provide population-level context for our estimate of predation loss.
Methods
Study area
The study was conducted on the Snake River between Lower Granite Dam (river kilometer [rkm] 173, as measured from the mouth of the Snake River) to rkm 369 within Hells Canyon; an unimpounded, but regulated, river reach that forms the Idaho-Oregon and Idaho-Washington state borders, USA (Fig. 1). The Snake River within Hells Canyon supports a spawning aggregate of Snake River fall Chinook salmon. Naturally produced fry emerge from the gravel during spring (Connor et al. 2002) and are supplemented annually by releases of ~ 2.4 million hatchery-reared fish (typically in May and June; DART 2019). Hells Canyon was divided into three reaches (upper HC [rkm 323–369], middle HC [rkm 272–303], lower HC [rkm 234–272]) because of geomorphological differences along the river continuum (Miller et al. 2002) and because main tributaries (Salmon River, Grande Ronde River) that enter the Snake River alter temperatures, increase flows, and contribute additional emigrating natural and hatchery salmonids in the system.
At Asotin, Washington (rkm 234), the Snake River transitions to an impounded state by the time it reaches the Clearwater River confluence at rkm 224 (Fig. 1). We refer to this reach as the Snake River transition zone (SRTZ) because water velocities decline rapidly and subyearling emigration rates slow dramatically (Tiffan et al. 2009). We refer to the 7-km section of the Snake River from the confluence of the Snake and Clearwater rivers downstream to the Port of Wilma (rkm 217) as the confluence (CON) reach where fish emigration rates are slow and smallmouth bass predation is high (Tiffan et al. 2009; Erhardt et al. 2018a). The remainder of the reservoir from the Port of Wilma downstream to Lower Granite Dam was divided into three roughly equal sections (upper Res [rkm 201–217], middle Res [rkm 187–201], and lower Res [rkm 173–187]) based on reservoir morphology and subyearling habitat suitability and availability (Tiffan et al. 2016).
Data collection
Smallmouth bass were collected from 2013 to 2018 to describe diet changes and estimate subyearling loss to predation in each reach, but not all reaches were sampled every year. Data for this study were collected and combined with data from previously published studies (Erhardt et al. 2018a; Erhardt and Tiffan 2018) to derive monthly estimates of predation metrics for each study reach. Study designs varied between reaches because of potential differences in habitat-related sampling efficiencies, safety, and logistical reasons (e.g., limited access points within Hells Canyon).
Sampling was conducted in the three Hells Canyon reaches from late March through June during 2013–2014. Reaches were sampled with a combination of angling (HC upper reach) and boat electrofishing (HC middle and lower reaches) for logistical and safety reasons; the 20-km section of river between the upper and middle Hells Canyon reaches was not sampled for the same reasons. Within each reach, four sites (200–800 m in length) were randomly chosen and fixed for the duration of the study. Most sites covered multiple habitat types (i.e., pool, riffle, glide, fan, bar), and sampling occurred on both banks of the river. The start and end points of each site typically occurred at habitat breaks (see Erhardt and Tiffan 2018 for details on habitat delineation) and when possible extended between two rapids. Preliminary analyses showed that the proportions of habitat surveyed in the fixed sampling sites, along with smallmouth bass relative abundances, were representative of the entire reach. Angled sites were fished for 75 min by 2–3 people on each of two boats that moved periodically along the shore to sample the entire shoreline. For sites that were electrofished, both shorelines were sampled using one dipnetter. We supplemented the fixed sampling sites with randomly chosen sites (without replacement) from a pool of river kilometer starting locations delineated to 0.1 km. Random sites were fixed points that were angled for 20 min, and sampling typically occurred in a different reach during each sampling interval.
The SRTZ and CON reaches of Lower Granite Reservoir were sampled with boat electrofishing biweekly from April through June 2013–2015; detailed methodologies can be found in Erhardt et al. (2018a). In brief, four random sites (500–800 m of shoreline) were selected per reach and fixed for the duration of the study. Sampling was stratified by two predominant shoreline habitat types: those modified by riprap revetments (riprap), and unaltered, low-gradient (‘suitable’, discussed below) shorelines that are preferred by rearing subyearlings (Tiffan et al. 2016).
The upper, middle, and lower reaches of Lower Granite Reservoir were sampled with electrofishing biweekly from April through June 2016–2018. We used a stratified-random sampling design based on the habitat suitability delineations identified by Tiffan et al. (2016). In brief, habitat deemed “suitable” to subyearlings was generally shallow with low lateral bed slope and small substrate, whereas unsuitable habitat was deeper with steep lateral bed slopes. Unsuitable habitat was further divided by substrate as “natural”, which contained large natural substrate (e.g., talus) and “riprap”, which was characterized by artificially placed boulders. Next, we used a geographic information system to characterize the shoreline by habitat type and calculate distances (m) for all continuous habitat patches. Within habitat patches, we established sampling transects along the shore that ranged from 100 to 200 m depending on total patch length; no transects were established in habitat patches < 100 m. For each two-week sampling interval, we randomly selected 16 transects per habitat without replacement from the pool of 100–200-m transects using proportional allocation methods based on the amount of available habitat type within a reach (Scheaffer et al. 1996).
All collected smallmouth bass ≥ 150 mm total length (TL) were measured, and weights were estimated using the following regression: Weight = 0.000007TL3.09 (n = 3,350, r2 = 0.93; Erhardt et al. 2018a). Stomach contents from smallmouth bass were collected using a nonlethal lavage technique modified from Seaburg (1957) that removed 99.8% of diet items (Erhardt et al. 2014). The lavage instrument consisted of a 6-mm diameter tube connected to a common garden spray nozzle that supplied filtered river water via a wash-down pump installed on the boat. Stomach contents were collected in a 42-µm sieve and preserved in 90% ethanol.
Smallmouth bass abundances, which were necessary to estimate predation losses, were estimated with methods described by Erhardt and Tiffan (2018) and Erhardt et al. (2018a). In brief, for Hells Canyon reaches, collected bass were tagged with anchor tags and recapture data were used to estimate abundance for the fixed sites in each reach for each biweekly sampling interval using open-population models (Jolley-Seber type; Schwarz and Arnason 1996). Because estimates could not be derived for the first and last sampling occasion within a year, we estimated abundances for these periods in each sample site by regressing weekly angling or electrofishing catch-per-unit-effort (CPUE) against weekly abundances (for the weeks where they could be derived) and predicting the first and last abundances from the CPUE estimates. Abundance estimates were then extrapolated to the entire reach. The number of bass per river kilometer in the unsampled section of river between the upper and middle Hells Canyon reaches was assumed to be the same as in the upper reach based on similar habitats and river morphology. Absolute abundance estimates of bass in the SRTZ and CON reaches were estimated by Erhardt et al. (2018a). In brief, catchability models were used to predict absolute density (fish/m) from electrofishing catch (also fish/m) for each habitat type (riprap and suitable) with the Chapman estimator of the Petersen index (Seber 1982). Recapture events of anchor-tagged bass were conducted 1–2 days following marking events at each site. Estimates were then extrapolated to the amount of available habitat within each reach for each sampling interval. We further developed these relationships during 2017–2018 by conducting additional mark-recapture sampling in the reservoir and deriving a new relationship for ‘natural’ habitat.
Data analysis
Diet
Laboratory analyses of bass stomach contents followed those of Erhardt et al. (2018a). All diet items were identified to the lowest practical taxon and weighed (± 0.001 g, wet); prey fish were identified to species using diagnostic bones (i.e., dentary, cleithrum, opercle; Parrish et al. 2006) when necessary. Size at ingestion of partially digested prey fish was estimated using regressions between fish length and various body parts or diagnostic bone measurements (Vigg et al. 1991; Parrish et al. 2006; Erhardt et al. 2014). Subyearlings were distinguished from yearling Chinook salmon based on differences in length-frequency distributions and emigration timing (Erhardt et al. 2018a). We accounted for a 21.3% weight loss of all diet items associated with preservation in 90% ethanol (Shields and Carlson 1996). Ingested prey were pooled by reach and sampling month to calculate frequencies of occurrence ([number of prey in bass stomachs / total number of bass examined] × 100) and percentages by weight ([weight of prey in bass stomachs / total weight of all prey in bass stomachs] × 100) of predominant prey. A more detailed description of how prey varied through time and by bass size can be found in Connor et al. (2015) and Erhardt et al. (2018a).
Consumption of subyearlings
We calculated the consumption rate, C (number of subyearlings/smallmouth bass/day), in a series of steps similar to Fritts and Pearsons (2004). First, we calculated the original weight of each prey fish at ingestion for each smallmouth bass using length-weight regressions (Vigg et al. 1991; Parrish et al. 2006) with the back-calculated length at ingestion described above. These were summed with other diet items (if present) to derive a meal weight (MW) for each smallmouth bass (Vigg et al. 1991). Next, we input MW into an evacuation rate model of smallmouth bass digestion of salmonids developed by Rogers and Burley (1991) and modified by Fritts and Pearsons (2004) that predicts time (in hours) to 90% evacuation (ET90):
where W is smallmouth bass weight (g; measured or estimated) and T is temperature. Finally, we calculated C for each individual smallmouth bass using the equation presented by Ward et al. (1995):
where n is the number of subyearlings found in the smallmouth bass gut. The mean C from all fish that were examined (including bass with empty stomachs) was calculated for each sampling site and a grand mean C was calculated for each reach across years. More detailed descriptions of how C varied seasonally and spatially can be found in Erhardt et al. (2017), Erhardt and Tiffan (2018), Erhardt et al. (2018a, b), Tiffan et al. (2019). Consumption in the unsampled section of river between the upper and middle Hells Canyon reaches was assumed to be the same as in the upper reach.
Loss estimation
We estimated the daily loss of subyearlings to predation by smallmouth bass by multiplying C by the corresponding bass abundance estimate within each reach for a given sampling interval. Loss within a sampling interval was estimated by expanding the daily loss by the number of days within the interval (typically 14 or 21). Interval losses were then summed to estimate monthly and total losses within each reach. We then calculated the mean monthly and total loss for each reach across years as an index of the magnitude of predation in each reach. To account for differences in reach lengths, monthly loss in each reach was divided by the reach length to express mortality on a per river kilometer basis. Estimates for the unsampled reach between the upper and middle Hells Canyon reaches were combined with those of the upper reach. In the upper Hells Canyon Reach, loss estimates during 2013–2014 (reported in Connor et al. 2014, 2015) likely underestimated bass consumption of hatchery-reared fish that were released each May because sampling did not occur directly following the hatchery fish releases when consumption was high (Erhardt and Tiffan 2018). In those years, predation losses were only estimated for natural subyearlings, which were distinguishable from hatchery fish based on length-frequency differences (see Erhardt and Tiffan 2018 for details). Erhardt and Tiffan (2018) subsequently conducted a study within the upper reach during 2015–2016 and estimated that 348,513 hatchery subyearlings were consumed in 1–2 d directly following the May hatchery releases of 1.4 million fish. Therefore, we expanded their hatchery loss estimate to the entire upper river reach and then added it to our loss estimate of natural-origin Chinook salmon. We assumed the pulse of hatchery fish dispersed enough in downstream reaches to not elicit a strong feeding response by predators. Finally, we plotted the cumulative predation loss of subyearlings along the river continuum from Hells Canyon Dam to Lower Granite Dam to show spatial changes in predation.
To provide perspective for our estimate of cumulative predation loss to Lower Granite Dam, we estimated the number of adult fall Chinook salmon that would have returned back to the dam had no predation on juveniles occurred. Our purpose was merely to illustrate the potential effects of juvenile predation losses at the adult population level and not to draw absolute inferences about predation consequences. This was because we had to make numerous assumptions for this analysis that were probably not realistic but served the illustrative nature of our purpose. First, we assumed our cumulative loss estimate was representative across years and then multiplied it by the post-harvest smolt-to-adult return (SAR) rates calculated for adults returning to Lower Granite Dam (DART 2019). Of our study years, we only considered SARs from brood years 2012–2015 (2013–2016 juvenile outmigration years) that were calculated from returns of one- to three-ocean age fish, which comprised the bulk (> 95%) of returning adults. Second, estimates of the number of adults forfeited to predation for each return year were then added to the actual adult return in that year to obtain an estimate of the potential number of adults that could have returned had no predation occurred. Adult loss was then expressed as a percentage of this number within each year.
Results
Diet
We examined the diets of 22,861 smallmouth bass which showed marked changes along the Snake River continuum from the unimpounded Hells Canyon to Lower Granite Reservoir (Figs. 2 and 3). Predominant prey in smallmouth bass diets included three main fish taxa (subyearlings, sculpin [Cottus spp.], sand roller [Percopsis transmontana]) and invertebrates (signal crayfish [Pacifastacus leniusculus], opossum shrimp [Neomysis mercedis], insects). The prevalence of subyearlings in smallmouth bass diets varied both seasonally and spatially, but typically composed < 25% of diets by both weight and frequency of occurrence (Figs. 2 and 3). Consumption of subyearlings was generally lowest in April but increased seasonally and varied by reach. The highest subyearling prevalence in bass diets was observed in May and June in the upper and lower Hells Canyon reaches, respectively. In the lower Hells Canyon reach, the percent by weight of subyearlings in bass diets was inversely proportional to that of sand roller (described below). In April and May, consumption of subyearlings was low whereas consumption of sand roller was high; the opposite was observed during June. Similarly, the consumption of subyearlings in the upper Hells Canyon reach was low during April when the consumption of sculpin was high (Fig. 2). However, this relationship was inconsistent for other prey, reaches, and months. Bass consumption of subyearlings increased slightly in a downstream direction in the upper, middle, and lower reservoir reaches during April and June as sand roller consumption sharply declined, but the same trend was not observed during May.
Seasonal and longitudinal changes in the frequency of occurrence of predominant prey in smallmouth bass diets in the Snake River from upper Hells Canyon to Lower Granite Dam during 2013–2018. Sampling reaches are abbreviated as Up HC (upper Hells Canyon), Mid HC (middle Hells Canyon), Low HC (lower Hells Canyon), SRTZ (Snake River transition zone), CON (confluence), Up Res (upper Lower Granite Reservoir), Mid Res (middle Lower Granite Reservoir), and Low Res (lower Lower Granite Reservoir)
Seasonal and longitudinal changes of predominant prey in smallmouth bass diets (percent by weight) in the Snake River from upper Hells Canyon to Lower Granite Dam during 2013–2018. Sampling reaches are abbreviated as Up HC (upper Hells Canyon), Mid HC (middle Hells Canyon), Low HC (lower Hells Canyon), SRTZ (Snake River transition zone), CON (confluence), Up Res (upper Lower Granite Reservoir), Mid Res (middle Lower Granite Reservoir), and Low Res (lower Lower Granite Reservoir)
Sculpin were mainly consumed in Hells Canyon reaches and few were observed in bass diets in reservoir reaches (Figs. 2 and 3). Sculpin were typically only present in about 20% of bass diets but at times composed as significant portion of bass diets by weight, particularly in the upper and middle reaches of Hells Canyon. In riverine reaches, bass consumption of sculpin was generally higher in April and May than in June.
Sand roller was the predominant prey consumed by smallmouth bass in lower Hells Canyon and upper reservoir reaches. They were absent in the upper and middle reaches of Hells Canyon but constituted 60–80% of all bass diets by weight in the lower Hells Canyon, SRTZ, and CON reaches during all months in those reaches (Fig. 2). Below the SRTZ reach, the consumption of sand roller by bass generally decreased steadily in a downstream direction to reach lows of about 10% of the diet by weight and about 5% frequency of occurrence during all months (Figs. 2 and 3).
Smallmouth bass consumption of crayfish showed an inverse relationship with the consumption of sand roller by both percent by weight and frequency of occurrence (Figs. 2 and 3). During all months, consumption of crayfish was highest in the upper and middle Hells Canyon reaches, declined to low levels in the lower Hells Canyon, SRTZ, and CON reaches, then increased in a downstream direction in the reservoir; the opposite trend was observed for sand roller. Although crayfish were generally found in less than 40% of bass diets (Fig. 3), they often composed 60–80% of diets by weight at times in the middle and upper reach of Hells Canyon and the middle and lower reservoir reaches (Fig. 2). The consumption of crayfish by bass tended to increase seasonally in most reaches.
Neomysis was absent in riverine reaches but was prevalent in smallmouth bass diets in reservoir reaches (Figs. 2 and 3). Neomysis occurred in relatively high percentages of bass diets (55–80% [frequency of occurrence]), particularly in April and May, but being relatively small prey, they composed lower percentages of diets by weight (Fig. 2). However, in the CON reach during April they constituted 35% of the diet by weight and in the middle reservoir reach during May they composed 29% of the diet by weight (Fig. 2). Although the frequency of occurrence of Neomysis in bass diets increased in a downstream direction to a high of 40% during June, they never represented more than 7% of the diet by weight (Fig. 3).
Smallmouth bass consumed a variety of terrestrial and aquatic insects in all reaches and during all months. The frequency of insects in bass diets increased seasonally within each reach, but occurrence decreased steadily from the upper Hells Canyon reach to the SRTZ and CON reaches then increased steadily again in lower reservoir reaches (Fig. 3). Although the frequency of occurrence of insects in bass diets was often high, they contributed little to the weight of diets except in the upper Hells Canyon reach during May and in the middle Hells Canyon reach during April and June (Fig. 2).
Smallmouth bass abundance and consumption
Smallmouth bass abundance varied seasonally and spatially within our study area. Bass abundance increased seasonally in all but the middle reach of Hells Canyon (Table 1). In riverine reaches, bass abundance tended to decrease in a downstream direction, whereas the opposite trend was observed in reservoir reaches except for the CON reach in June. Bass were generally most abundant (> 1,000 bass/rkm) in the upper reach of Hells Canyon and in the middle and lower reaches of the reservoir, but the CON reach also contained a high number of bass in June (Table 1).
Smallmouth bass consumption of subyearlings was variable over reaches and months but generally showed similar patterns to bass abundance. In riverine reaches, C peaked in the upper reach in May (0.130 subyearlings/bass/day), was somewhat consistent across months in the middle reach, and increased monthly in the lower reach (Table 1). In reservoir reaches, C varied by reach within months and no clear patterns were evident. Seasonally, C generally increased within reservoir reaches. The highest Cs (> 0.12 subyearlings/bass/day) were observed in the upper reach of Hells Canyon during May, and in all reservoir reaches except the SRTZ reach during June (Table 1).
Predation loss
Estimated total loss of subyearlings to smallmouth bass predation was highest in the upper Hells Canyon reach in April and May and declined rapidly in a downstream direction during those months (Table 2; Fig. 4). Losses in reservoir reaches during April and May were relatively low, ranging from 0 to < 15,000 fish (Table 2). During June, estimated total losses of subyearlings increased from a low of 7,535 fish in the upper Hells Canyon reach to a high of 67,962 fish in lower Hells Canyon then declined and were relatively stable in most reservoir reaches (range, 17,021–42,323; Table 2; Fig. 4). Downstream of the middle reach of Hells Canyon, losses were always higher in the June than during April and May. However, because reaches differed in length, expressing loss per river kilometer showed where the most intense predation occurred.
Seasonal and longitudinal estimates of mean number (panel a) and mean number/km (panel b) of subyearling Chinook salmon lost to smallmouth bass predation in the Snake River from upper Hells Canyon to Lower Granite Dam during 2013–2018. Sampling reaches are abbreviated as Up HC (upper Hells Canyon), Mid HC (middle Hells Canyon), Low HC (lower Hells Canyon), SRTZ (Snake River transition zone), CON (confluence), Up Res (upper Lower Granite Reservoir), Mid Res (middle Lower Granite Reservoir), and Low Res (lower Lower Granite Reservoir)
During April, the loss of subyearlings per river kilometer generally showed the same longitudinal trend as was observed for total loss. The loss per river kilometer was highest in the upper and middle Hells Canyon reaches and declined to low levels in downstream reaches (Table 2; Fig. 4). During May, subyearling loss per river kilometer declined from the upper to lower Hells Canyon reaches but increased in the SRTZ then declined in a downstream direction. During June, loss increased from a low of 114 fish/rkm in the upper Hells Canyon reach to about 1,750 fish/rkm in the lower Hells Canyon and SRTZ reaches. Loss then increased over 3-fold in the CON reach, declined again in the upper reservoir, and then increased to a high of 3,023 fish/rkm in the lower-most reach of the reservoir (Table 2; Fig. 4).
Cumulative loss of subyearlings to smallmouth bass predation increased steadily in a downstream direction from upper Hells Canyon to Lower Granite Dam (Table 2; Fig. 5). Three-quarters (654,710 fish) of the total cumulative loss (869,371 fish) of subyearlings occurred in the riverine reaches of Hells Canyon (Table 2). Our estimated mean annual total loss of 869,371 subyearlings to smallmouth bass predation equated to adult losses that ranged from 2,434 fish in 2014 to 10,780 fish in 2013 (Table 3). This represented 3.9% and 16.0% of the potential adult run that could have returned to Lower Granite Dam had no subyearling predation by smallmouth bass occurred (Table 3). This assumes that no compensatory mortality of salmonids would have taken place downstream of Lower Granite Dam before fish returned as adults.
Estimated cumulative number of subyearling Chinook salmon lost to smallmouth bass predation in the Snake River from upper Hells Canyon to Lower Granite Dam during 2013–2018. Sampling reaches are abbreviated as Up HC (upper Hells Canyon), Mid HC (middle Hells Canyon), Low HC (lower Hells Canyon), SRTZ (Snake River transition zone), CON (confluence), Up Res (upper Lower Granite Reservoir), Mid Res (middle Lower Granite Reservoir), and Low Res (lower Lower Granite Reservoir). The x-axis has been adjusted for reach length
Discussion
The seasonal and spatial variation in prey consumption by smallmouth bass likely reflected prey habitat preferences and availability, which was a function of abundance and behavior. Sculpin was the predominant nonsalmonid prey fish in riverine habitats in the upper and middle reaches of Hells Canyon and were rarely consumed in the reservoir. Sculpin was replaced by sand roller in bass diets in the lower reach of Hells Canyon and in most reservoir reaches. The sand roller, whose population has increased in recent years, is most abundant in shoreline habitats when they spawn during April and May after which they move offshore into deeper water where they are probably less vulnerable to smallmouth bass predation (Tiffan et al. 2017a). This explains their greater prominence in bass diets during these months. Furthermore, the abundance of sand roller decreased in lower reservoir reaches (Erhardt et al. 2017, 2018b) and were replaced mainly by crayfish in bass diets, particularly later in the season.
Crayfish, although present in every river reach, were most prevalent in smallmouth bass diets when other prey fish decreased. In the upper and middle reaches of Hells Canyon, crayfish composed a smaller portion of bass diets by weight when sculpin consumption was greater, and the same trend was observed with sand roller in reaches where they were present. This suggests that smallmouth bass may select fishes over crayfish when prey fish are abundant (e.g., Carter et al. 2010), but there was no obvious relationship between bass consumption of subyearlings and crayfish.
It is noteworthy that the other predominant invertebrate in smallmouth bass diets was Neomysis mercedis—a relatively recent addition to the reservoir food web (Tiffan et al. 2017b). Like the sand roller, Neomysis has become very abundant in Lower Granite Reservoir and is preyed heavily upon by bass mainly during April and May when many mysids use shoreline habitats for reproduction (Tiffan et al. 2017b). Although Neomysis may not contribute to bass diets to the same extent as fishes and crayfish because of their small size (~ 14 mm TL), they may be providing a ready food source for smaller bass and contributing to their survival and recruitment.
The greatest losses of subyearlings to smallmouth bass predation in riverine habitats occurred during April and May in the upper Hells Canyon reach where bass abundance and consumption rates were high. This reach is the warmest of all contemporary production areas and subyearlings emerge early there and are most abundant in this reach early in the year (Connor et al. 2002). The warm temperatures also likely increased bass metabolism and feeding. Most subyearlings produced in this reach disperse downstream by June which explains why few fish were consumed during that month. In addition, the greater bass abundance in the upper and middle reaches, and hence greater subyearling loss, may be the result of more suitable smallmouth bass habitat in those reaches compared with the lower reach where bass abundance and subyearling loss were generally lower except during June (Erhardt and Tiffan 2018). Subyearling losses were also highest in June in all reservoir reaches.
The high subyearling predation losses in reservoir reaches occurred because bass abundances and consumption rates were high. This is not unexpected because seasonal increases in temperature should increase bass metabolism and feeding (Whitledge et al. 2002). In addition, many bass move into shoreline habitats during June to spawn (Carlander 1977), which increases their apparent abundance and habitat overlap with subyearlings. Thus, predation risk is highest in June for subyearlings that use reservoir shoreline habitats. This risk increases in a downstream direction as habitats become less suitable for subyearlings (Tiffan et al. 2016) and smallmouth bass habitat suitability, consumption rate, and abundance increase. One factor that partially mitigates this predation risk is the presence of alternative prey for bass.
We documented distinct changes in smallmouth bass diets along the continuum from riverine to reservoir habitats that influenced predation losses of subyearlings. In riverine habitats, the presence of sculpin and crayfish probably reduced predation on subyearlings to some extent, particularly in the upper and middle reaches of Hells Canyon. In contrast, in the lower reach of Hells Canyon, subyearling loss was lowest during April and May when the presence of sand roller in bass diets was greatest, and subyearling loss was highest in June when bass consumed fewer sand roller. This agrees with past studies that found inverse relationships between subyearling and sand roller losses to smallmouth bass predation in this reach and in the upper portion of Lower Granite Reservoir (Erhardt and Tiffan 2018; Hemingway et al. 2019). This relationship was also apparent in lower reservoir reaches where subyearling losses increased as the consumption and abundance of sand roller decreased (Erhardt et al. 2017, 2018b). This led Hemingway et al. (2019) to speculate that sand roller provide a predation buffer for subyearlings, and this is probably true for other prey that smallmouth bass consume.
Translating subyearling predation losses into foregone adult production served to illustrate the potential population-level effect that smallmouth bass predation can have on Snake River fall Chinook salmon. Although a relatively large number of adults returned from 2013 juvenile outmigrants, the high SAR in that year (1.24%) resulted in the largest number of potential adults lost because of predation. Under a high SAR, each juvenile contributes more to adult production than under a low SAR. But as SARs stay relatively constant and adult returns decline, juvenile predation exacts a greater toll on potential adult production. This is illustrated by estimates made for 2015 and 2016 wherein SARs were similar between years, but as adult returns declined by almost one-half in 2016, the percentage of the potential adult run lost to juvenile predation almost doubled (Table 3). This relationship would become even more complicated if cumulative juvenile loss varied between years, which we assumed for illustrative purposes that it did not. This assumption is valid for hatchery-origin subyearlings whose annual release numbers typically do not vary (DART 2019), but it is less certain for natural-origin fish for which annual production estimates do not exist. This analysis also assumed no compensatory mortality occurred downstream of Lower Granite Dam had the subyearlings not been depredated, which is certainly not the case. We recognize that some subyearlings that emigrate later than our sampling period or downstream of the dam likely die from factors such as additional predation, dam passage, disease, and starvation. This mortality could not be accounted for, so we assumed no mortality for simplicity. As such, our estimates of foregone adult production represent a best-case scenario. This analysis, however, provides a context for our loss estimate, and shows that significant numbers of adults could have contributed to this ESA-listed population had predation on juveniles not existed or been reduced. To our knowledge relating juvenile salmon predation to adult returns has not been done and should provide insight for future work on this topic.
It is important to recognize the assumptions and limitations of this study. It was necessary to pool data across years to make inferences about predation effects over our entire study area. We assumed that smallmouth bass diets, abundance, consumption, and subyearling losses were representative of all years when sampling occurred even though not all reaches were sampled in all years. While we recognize that this assumption may not always be valid due to annual variability, the results can be thought of as reflecting some “average” year and do provide a relative index of the magnitude of predation losses in various reaches and the potential mechanisms that affect loss estimates along the river continuum. We also assumed that our sampling adequately represented each reach that we studied. During all study years and reaches, the large number of smallmouth bass collected (22,861 for diet analysis) makes us confident in our reach-specific results.
It is important to recognize that loss estimates are sensitive to the quality of estimates of smallmouth bass abundance and C, both of which were variable during our study years (Erhardt et al. 2014, 2017, 2018b). Because loss estimates are derived from expansions of bass abundance and C, small changes in either, or both, can greatly influence estimates. However, each reach was represented by at least two (Hells Canyon) or three (reservoir) years of sampling, which captured some of the annual variability for estimating mean values of predation metrics. There were also aspects of predation such as size-selectivity of prey and differential size-based consumption rates of smallmouth bass that we did not account for that could have affected our estimates. For example, if bass selected smaller, weaker subyearlings that were destined to die anyway, then estimates of foregone adult production would have been biased high. Finally, we recognize that both angling and electrofishing each have their own biases that could have affected our estimates of diet, consumption, abundance, and loss.
There are few options for reducing smallmouth bass predation risk to subyearlings, which exists along the entire river continuum due to habitat overlap between the two species. Sport fishing bag limits on smallmouth bass have already been eliminated in the Snake River downstream of Hells Canyon Dam in Washington, Oregon, and Idaho. Subyearling emigration behavior may, however, reduce predation risk to some extent. Once subyearlings become smolts, they tend to emigrate in offshore, main-channel habitats (Connor et al. 2003), which would segregate them from shoreline-oriented bass. Early emigrants may also reduce their predation risk by emigrating under cooler temperatures when bass metabolic activity and consumption rates are low. Our results suggest that bass consumption of alternative prey reduces the incidence of predation on subyearlings; thus, it is important to understand and monitor food web changes that might ultimately affect smallmouth bass predation on salmonids. Estimating juvenile predation loss over larger landscape scales and translating that loss to adult returns are essential for contextualizing the threat that non-native piscine predation poses to the recovery of ESA-listed salmonids in large, complex river systems. In addition, understanding the potential of food web interactions on predation loss over a river continuum is critical for developing effective system-wide mitigation strategies (Naiman et al. 2012). Although this study was limited by a number of assumptions and constraints, it does provide an illustration of how predation affects juvenile and adult salmon loss over a broad, changing river landscape.
Data availability
Not applicable.
References
Anglea SM (1997) Abundance, food habits, and salmonid fish consumption of smallmouth bass and distribution of crayfish in Lower Granite Reservoir, Idaho-Washington. Master’s Thesis, University of Idaho
Carey MP, Sanderson BL, Friesen TA, Barnas KA, Olden JD (2011) Smallmouth bass in the Pacific Northwest: A threat to native species; a benefit for anglers. Rev Fish Sci 19:305–315
Carlander KD (1977) Handbook of freshwater biology, vol 2. Iowa State University Press, Ames
Carter MW, Shoup DE, Dettmers JM, Wahl DH (2010) Effects of turbidity and cover on prey selectivity of adult smallmouth bass. Trans Am Fish Soc 139:353–361
Collis K, Beaty RE, Crain BR (1995) Changes in catch rate and diet of northern squawfish associated with the release of hatchery-reared juvenile salmonids in a Columbia River reservoir. N Am J Fish Manag 15:346–357
Connor WP, Burge HL, Waitt R, Bjornn TC (2002) Juvenile life history of wild fall Chinook salmon in the Snake and Clearwater rivers. N Am J Fish Manag 22:703–712
Connor WP, Steinhorst RK, Burge HL (2003) Migrational behavior and seaward movement of wild subyearling fall Chinook salmon in the Snake River. N Am J Fish Manag 23:414–430
Connor WP, Tiffan KF, Mullins F, Arnsberg B, Cooney TD, Groves PA, Milks DJ, Rosenberger SJ, Young W (2014) Research, monitoring, and evaluation of emerging issues and measures to recover the Snake River fall Chinook salmon ESU. 2013 Annual Report to the Bonneville Power Administration, Project 199102900, Portland, Oregon. Available: https://www.cbfish.org/Document.mvc/Viewer/P139946. Accessed Feb 2020
Connor WP, Mullins FL, Tiffan KF, Perry RW, Erhardt JM, St. John SJ, Bickford BK, Rhodes TN (2015) Research, monitoring, and evaluation of emerging issues and measures to recover the Snake River fall Chinook salmon ESU. 2014 Annual Report to the Bonneville Power Administration, Project 199102900, Portland, Oregon. Available: https://www.cbfish.org/Document.mvc/Viewer/P143033. Accessed Feb 2020
DART (Data Access in Real Time) (2019) Columbia River DART. Columbia Basin Research, University of Washington, Seattle. Available: www.cbr.washington.edu/dart/dart.html. Accessed Dec 2019
Erhardt JM, St. John SJ, Bickford BK, Rhodes TN, Tiffan KF (2014) Smallmouth bass predation on juvenile fall Chinook salmon in Lower Granite Reservoir on the Snake River. In: Tiffan KF, Connor WP, Bellgraph BJ, Chittaro PM (ed) pp 27–60. Snake River fall Chinook salmon life history investigations. 2012 Annual Report to the Bonneville Power Administration, Project 2002-032-00, Portland, Oregon. Available: https://www.cbfish.org/Document.mvc/Viewer/P139225. Accessed Feb 2020
Erhardt JM, Bickford BK, Hemingway RJ, Rhodes TN, Tiffan KF (2017) Snake River fall Chinook salmon life history investigations. 2016 Annual Report to the Bonneville Power Administration, Project 200203200, Portland, Oregon. Available: https://www.cbfish.org/Document.mvc/Viewer/P154611 Accessed Feb 2020
Erhardt JM, Tiffan KF (2018) Post-release predation mortality of age-0 hatchery Chinook salmon from non-native smallmouth bass in the Snake River. Fish Manag Ecol 25:474–487
Erhardt JM, Tiffan KF, Connor WP (2018a) Juvenile Chinook salmon mortality in a Snake River reservoir: smallmouth bass predation revisited. Trans Am Fish Soc 147:316–328
Erhardt JM, Hemingway RJ, Tiffan KF, Chittaro P, Kennedy B, Hegg J (2018b) Snake River fall Chinook salmon life history investigations. 2017 Annual Report to the Bonneville Power Administrations, Project 200203200, Portland, Oregon. Available: https://www.cbfish.org/Document.mvc/Viewer/P159940 . Accessed Feb 2020
Evans AF, Hostetter NJ, Roby DD, Collis K, Lyons DE, Sandford BP, Ledgerwood RD, Sebring S (2012) Systemwide evaluation of avian predation on juvenile salmonids from the Columbia River based on recoveries of passive integrated transponder tags. Trans Am Fish Soc 141:975–989
Fritts AL, Pearsons TN (2004) Smallmouth bass predation on hatchery and wild salmonids in the Yakima River, Washington. Trans Am Fish Soc 133:880–895
Furey NB, Hinch SG, Mesa MG, Beauchamp DA (2016) Piscivorous fish exhibit temperature-influenced binge feeding during an annual prey pulse. J Animal Ecol 85:1307–1317
Gregory RS, Levings CD (1998) Turbidity reduce predation migrating juvenile Pacific salmon. Trans Am Fish Soc 127:275–285
Grossman GD (2016) Predation on fishes in the Sacramento–San Joaquin Delta: current knowledge and future directions. San Francisco Estuary Watershed Sci 14(2):8 Available: https://doi.org/10.15447/sfews.2016v14iss2art8. Accessed Dec 2019
Hemingway RJ, Tiffan KF, Erhardt JM, Rhodes TN, Bickford BK (2019) Fall Chinook salmon (Oncorhynchus tshawytscha), sand roller (Percopsis transmontana), and smallmouth bass (Micropterus dolomieu) interactions in a Snake River reservoir: a tale of three species. Northwestern Nat 100:26–36
Miller S, Glanzman E, Doran S, Parkinson K, Buffington J, Milligan J (2002) Geomorphology of the Hells Canyon reach of the Snake River. Technical report E. 1–2. Idaho Power Company, Boise, ID. 182 pp
Naiman RJ, Alldredge JR, Beauchamp DA, Bisson PA, Congleton J, Henny CJ, Huntly N, Lamberson R, Levings C, Merrill EN, Pearcy WG, Rieman BE, Ruggerone GT, Scarnecchia D, Smouse PE, Wood CC (2012) Developing a broader scientific foundation for river restoration: Columbia River food webs. Proc Natl Acad Sci USA 109:21201–21207
Naughton GP, Bennett DH, Newman KB (2004) Predation on juvenile salmonids by smallmouth bass in the Lower Granite Reservoir system, Snake River. N Am J Fish Manag 24:534–544
Nelle RD (1999) Smallmouth bass predation on juvenile fall Chinook salmon in the Hells Canyon reach of the Snake River, Idaho. Master’s Thesis, University of Idaho, Moscow
Parrish JK, Haapa-aho K, Walker W, Stratton M, Walsh J, Ziel H (2006) Small-bodied and juvenile fishes of the mid-Columbia region including keys to diagnostic otoliths and cranial bones. University of Washington, Seattle
Poe TP, Hansel HC, Vigg S, Palmer DE, Predergast LA (1991) Feeding of predaceous fishes on out-migrating juvenile salmonids in John Day Reservoir, Columbia River. Trans Am Fish Soc 120:405–420
Rieman BE, Beamsderfer RC, Vigg S, Poe TP (1991) Estimated loss of juvenile salmonids to predation by Northern Squawfish, Walleyes, and Smallmouth Bass in John Day Reservoir, Columbia River. Trans Am Fish Soc 120:448–458
Rogers JB, Burley CC (1991) A sigmoid model to predict gastric evacuation rates of smallmouth bass (Micropterus dolomieu) fed juvenile salmon. Can J Fish Aquat Sci 48:933–937
Sabal M, Hayes S, Merz J, Setka J (2016) Habitat alterations and a nonnative predator, the striped bass, increase native Chinook Salmon mortality in the Central Valley, California. N Am J Fish Manag 36:309–320
Sanderson BL, Barnas KA, Rub AMW (2009) Nonindigenous species of the Pacific Northwest: An overlooked risk to endangered salmon? Bioscience 59:245–256
Scheaffer RL, Mendenhall W III, Ott RL (1996) Elementary survey sampling. 5th edn. Duxbury Press, Belmont
Schwarz CJ, Arnason AN (1996) A general methodology for the analysis of open model capture recapture experiments. Biometrics 52:860–873
Seaburg KG (1957) A stomach sampler for live fish. Progress Fish Cult 19:137–139
Seber GAF (1982) The estimation of animal abundance and related parameters. Edward Arnold, London
Shields PA, Carlson SR (1996) Effects of formalin and alcohol preservation on lengths and weights of juvenile Sockeye Salmon. Alsk Fish Res Bull 3:81–93
Shively RS, Poe TP, Sauter ST (1996) Feeding response by northern squawfish to a hatchery release of juvenile salmonids in the Clearwater River, Idaho. Trans Am Fish Soc 125:230–236
Tabor RA, Shively RS, Poe TP (1993) Predation on juvenile salmonids by smallmouth bass and northern squawfish in the Columbia River near Richland, Washington. N Am J Fish Manag 13:831–838
Tiffan KF, Kock TJ, Haskell CA, Connor WP, Steinhorst RK (2009) Water velocity, turbulence, and migration rate of subyearling fall Chinook salmon in the free-flowing and impounded Snake River. Trans Am Fish Soc 138:373–384
Tiffan KF, Hatten JR, Trachtenbarg DA (2016) Assessing juvenile salmon rearing habitat and associated predation risk in a lower Snake River reservoir. River Res Appl 32:1030–1038
Tiffan KF, Erhardt JM, Rhodes TN, Hemingway RJ (2017a) Ecology of the Sand Roller (Percopsis transmontana) in a lower Snake River reservoir, Washington. Northwestern Nat 98:203–214
Tiffan KF, Erhardt JM, Bickford BK (2017b) Ecology of the opossum shrimp (Neomysis mercedis) in a lower Snake River reservoir, Washington. Northwest Sci 91:124–139
Tiffan KF, Chittaro P, Kennedy B (2019) Snake River fall Chinook salmon life history investigations. 2018 Annual Report to the Bonneville Power Administrations, Project 200203200, Portland, Oregon. Available: https://www.cbfish.org/Document.mvc/Viewer/P166058. Accessed Feb 2020
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE (1980) The river continuum concept. Can J Fish Aquat Sci 37:130–137
Vigg S, Poe TP, Prendergast LA, Hansel HC (1991) Rates of consumption of juvenile salmonids and alternate prey fish by northern squawfish, walleyes, smallmouth bass, and channel catfish in John Day Reservoir, Columbia River. Trans Am Fish Soc 120:421–438
Ward DL, Petersen JH, Loch JJ (1995) Index of predation on juvenile salmonids by northern squawfish in the lower and middle Columbia River and in the lower Snake River. Trans Am Fish Soc 124:321–334
Whitledge GW, Hayward RS, Rabeni CG (2002) Effects of temperature on specific daily metabolic demand and growth scope of sub-adult and adult smallmouth bass. J Freshw Ecol 17:353–361
Zimmerman MP, Parker RM (1995) Relative density and distribution of Smallmouth Bass, Channel Catfish and Walleye in the lower Columbia and Snake rivers. Northwest Sci 69:19–28
Acknowledgements
We thank our former U.S. Geological Survey colleagues, in particular, Scott St. John, Larry Durham, Dustin Lyons, and Margaret Zee whose efforts contributed to this study. This study was funded by the Bonneville Power Administration, project 200203200, and administered by D. Docherty. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Funding
This study was funded by the Bonneville Power Administration.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Not applicable.
Ethics approval
All animals were collected and handled according to guidelines specified in state fish collection and federal Endangered Species Act permits.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tiffan, K.F., Erhardt, J.M., Hemingway, R.J. et al. Impact of smallmouth bass predation on subyearling fall Chinook salmon over a broad river continuum. Environ Biol Fish 103, 1231–1246 (2020). https://doi.org/10.1007/s10641-020-01016-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-020-01016-0