Abstract
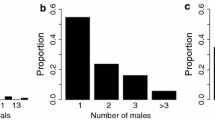
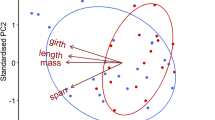
We examined the importance of sex differences in predation risk in generating sexual segregation in the guppy, Poecilia reticulata. We hypothesised that sex differences in predation risk will result in habitat segregation and ultimately social segregation of the sexes, with the more vulnerable sex (males in this case) using safer habitats. In accordance with the predation risk hypothesis we observed sexual segregation in a population associated with high but not low predation risk. Under high predation risk we observed a larger proportion of males in shallow marginal habitats resulting in habitat segregation and ultimately social segregation of the sexes. Furthermore, habitat segregation by sex was associated with habitat segregation by body length with shoals in deeper water having a larger mean body length. Shoaling fish species have been key models in investigating group living, and further research directed towards understanding sexual segregation in other fish species would be valuable.
Similar content being viewed by others
References
Abrahams, M. V. & L. M. Dill. 1989. A determination of the energetic equivalence of the risk of predation. Ecology 70: 999–1007.
Coleman, R. M. & V. Kutty. 2001. The predator of guppies on Trinidad is the pike cichlid Crenicichla frenata, not Crenicichla alta: a caution about working with cichlids. J. Aquaricult. Aquat. Sci. 9: 89–92.
Conradt, L. 1998. Measuring the degree of sexual segregation in group-living animals. J. Anim. Ecol. 67: 217–226.
Croft, D. P., B. J. Arrowsmith, J. Bielby, K. Skinner, E. White, I. D. Couzin, A. E. Magurran, I. Ramnarine & J. Krause. 2003. Mechanisms underlying shoal composition in the Trinidadian guppy (Poecilia reticulata ). Oikos 100: 429–438.
Gilliam, J. F. & D. F. Fraser. 1987. Habitat selection under predation hazard-test of a model with foraging minnows. Ecology 68: 1856–1862.
Grether, G. F., D. F. Millie, M. J. Bryant, D. N. Reznick & W. Mayea. 2001. Rain forest canopy cover, resource availability, and life history evolution in guppies. Ecology 82: 1546–1559.
Griffths, S. W. 1996. Sex differences in the trade-off between feeding and mating in the guppy. J. Fish Biol. 48: 891–898.
Haskins, C. P., E. F. Haskins, J. J. A. McLaughlin & R. E. Hewitt 1961. Polymorphism and population structure in Lebistes reticulatus, an ecological study. pp. 320–395. In: W. F. Blair (ed. ), Vertebrate Speciation, University of Texas Press, Austin.
Houde, A. E. 1997. Sex, Color, and Mate Choice in Guppies, Princeton University Press, Princeton. 207 pp.
Jakimchuk, R. D., S. H. Ferguson & L. G. Sopuck. 1987. Differential habitat use and sexual segregation in the Central Arctic caribou herd. Can. J. Zool.-Rev. Can. Zool. 65: 534–541.
Johnson, C. N. & P. G. Bayliss. 1981. Habitat selection by sex, age and reproductive class in the red kangaroo, Macropus rufus, in Western-New-South-Wales. Aust. Wildlife Res. 8: 465–474.
Liley, N. R. 1966. Ethological isolating mechanisms in four sympatric species of Poeciliid shes. Behav. Suppl. 13: 1–197.
Liley, N. R. & B. H. Seghers. 1975. Factors affecting the morphology and behaviour of the guppy (Poecilia reticulata )in Trinidad. pp. 92–118. In: G. Baerands & A. Manning (eds. ), Function and Behaviour in Evolution, Oxford University Press, Oxford.
Luyten, P. H. & N. R. Liley. 1991. Sexual selection and com-petitive mating success of males guppies (Poecilia reticulata ) from 4 Trinidad populations. Behav. Ecol. Sociobiol. 28: 329–336.
Maglio, V. J. & D. E. Rosen. 1969. Changing preferences for substrate colour by reproductively active mosquito fish, Gambusia affinis (Baird and Girard)(Poeciliidae, Atherini-formes). Am. Mus. Novitates 2379: 1–37.
Magurran, A. E. & M. A. Nowak. 1991. Another battle of the sexes-the consequences of sexual asymmetry in mating costs and predation risk in the guppy, Poecilia reticulate. Proc. R. Soc. London, Ser. B-Biol. Sci. 246: 31–38.
Magurran, A. E. & B. H. Seghers. 1994a. A cost of sexual harassment in the guppy, Poecilia reticulate. Proc. R. Soc. London, Ser. B-Biol. Sci. 258: 89–92.
Magurran, A. E. & B. H. Seghers. 1994b. Sexual conflict as a consequence of ecology-evidence from guppy, Poecilia reticulata, populations in Trinidad. Proc. R. Soc. London, Ser. B-Biol. Sci. 255: 31–36.
Magurran, A. E., B. H. Seghers, G. R. Carvalho & P. W. Shaw. 1992. Behavioural consequences of an artificial introduction of guppies (Poecilia reticulata )in N. Trinidad: evidence for the evolution of antipredator behaviour in the wild. Proc. R. Soc. London, Ser. B-Biol. Sci. 248: 117–122.
Main, M. B. & B. E. Coblentz. 1990. Sexual segregation among ungulates-a critique. Wildlife Soc. Bull. 18: 204–210.
Main, M. B. & B. E. Coblentz. 1996. Sexual segregation in rocky mountain mule deer. J. Wildlife Manage. 60: 497–507.
Main, M. B., F. W. Weckerly & F. C. Bleich. 1996. Sexual segregation in ungulates: new directions for research. J. Mammal. 77: 449–461.
Mattingly, H. T. & J. Butler. 1994. Laboratory predation on the Trinidadian guppy-implications for the size-selective predation hypothesis and guppy life-history evolution. Oikos 69: 54–64.
Mysterud, A. 2000. The relationship between ecological segregation and sexual body size dimorphism in large herbivores. Oecologia 124: 40–54.
Ranta, E., N. Peuhkuri & A. Laurila. 1994. A theoretical exploration of antipredatory and foraging factors promoting phenotype-assorted fish schools. Ecoscience 1: 99–106.
Reznick, D., M. J. Butler & H. Rodd. 2001. Life-history evolution in guppies. VII. The comparative ecology of high-and low-predation environments. Am. Nat. 157: 12–26.
Rodd, F. H. & D. N. Reznick. 1997. Variation in the demography of guppy populations: the importance of predation and life histories. Ecology 78: 405–418.
Ruckstuhl, K. E. 1998. Foraging behaviour and sexual segregation in bighorn sheep. Anim. Behav. 56: 99–106.
Ruckstuhl, K. E. & P. Neuhaus. 2000. Sexual segregation in ungulates: a new approach. Behaviour 137: 361–377.
Ruckstuhl, K. E. & P. Neuhaus. 2002. Sexual segregation in ungulates: a comparative test of three hypotheses. Biol. Rev. 77: 77–96.
Seghers, B. H. 1973. An analysis of geographic variation in the antipredator adaptations of the guppy, Poecilia reticulate. Ph. D. Thesis, The University of British Columbia.
Shaw, P. W., G. R. Carvalho, A. E. Magurran & B. H. Seghers. 1991. Population differentiation in Trinidadian guppies (Poecilia reticulata )-patterns and problems. J. Fish Biol. 39: 203–209.
Sims, D. W., J. P. Nash & D. Morritt. 2001. Movements and activity of male and female dogfish in a tidal sea lough: alternative behavioural strategies and apparent sexual seg-regation. Mar. Biol. 139: 1165–1175.
Smultea, M. A. 1994. Segregation by humpback whale (Megaptera novaeangliae )cows with a calf in coastal habitat near the Island of Hawaii. Can. J. Zool. 72: 805–811.
Weeden, R. 1964. Spatial segregation of sexes in rock and willow ptarmigan in winter. Auk 81: 534–541.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Croft, D., Krause, M. Is sexual segregation in the guppy, Poecilia reticulata, consistentwith the predation risk hypothesis?. Environmental Biology of Fishes 71, 127–133 (2004). https://doi.org/10.1007/s10641-004-0092-5
Issue Date:
DOI: https://doi.org/10.1007/s10641-004-0092-5