Summary
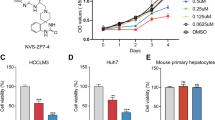
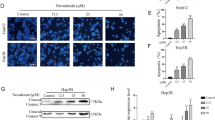
Background Hepatocellular carcinoma (HCC) is one of the most common cancers with a high mortality rate due to metastasis and relapse. Purpose Here, we reported a small-molecule pyridazinone compound, designated as IMB5036. Its antitumor activity against HCC and underlying mechanism were studied. Methods In vitro cytotoxicity, apoptosis, DNA breaks, and cell motility assays were performed. Protein expression was analyzed by Western blot and microarray analysis. A xenograft tumor model in athymic mice was used to evaluate the antitumor activity. Results IMB5036 displayed potent cytotoxicity against various HCC cell lines. It caused double DNA breakages and induced cell death via apoptosis. It also significantly inhibited the motility of HCC cells. Western blot showed that IMB5036 induced the up-regulation of E-cadherin, while down-regulation of N-cadherin. The gene expression profile analysis and Western blot assay revealed that IMB5036 down-regulated the expression of Tau protein. Analysis of the TCGA dataset revealed that high expression of Tau decreased the survival rate of HCC patients. In vivo experiments proved that IMB5036 significantly inhibited the growth of HCC xenografts in athymic mice. Conclusions These results collectively demonstrate IMB5036 can be a promising therapeutic candidate for patients with HCC.






Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer j clinicians 68(6):394–424. https://doi.org/10.3322/caac.21492
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA: a cancer j clinicians 70(1):7–30. https://doi.org/10.3322/caac.21590
Kim JJ, McFarlane T, Tully S, Wong WWL (2020) Lenvatinib Versus Sorafenib as First-Line Treatment of Unresectable Hepatocellular Carcinoma: A Cost-Utility Analysis. Oncologist 25(3):e512–e519. https://doi.org/10.1634/theoncologist.2019-0501
Kudo M, Finn RS, Qin S, Han KH, Ikeda K, Piscaglia F, Baron A, Park JW, Han G, Jassem J, Blanc JF, Vogel A, Komov D, Evans TRJ, Lopez C, Dutcus C, Guo M, Saito K, Kraljevic S, Tamai T, Ren M, Cheng AL (2018) Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet (London, England) 391(10126):1163–1173. https://doi.org/10.1016/s0140-6736(18)30207-1
Toh TB, Lim JJ, Hooi L, Rashid M, Chow EK (2020) Targeting Jak/Stat pathway as a therapeutic strategy against SP/CD44+ tumorigenic cells in Akt/β-catenin-driven hepatocellular carcinoma. J Hepatol 72(1):104–118. https://doi.org/10.1016/j.jhep.2019.08.035
Fu ZG, Wang L, Cui HY, Peng JL, Wang SJ, Geng JJ, Liu JD, Feng F, Song F, Li L, Zhu P, Jiang JL, Chen ZN (2016) A novel small-molecule compound targeting CD147 inhibits the motility and invasion of hepatocellular carcinoma cells. Oncotarget 7(8):9429–9447. https://doi.org/10.18632/oncotarget.6990
Lv J, Zhu B, Zhang L, Xie Q, Zhuo W (2015) Detection and screening of small molecule agents for overcoming Sorafenib resistance of hepatocellular carcinoma: a bioinformatics study. Int J Clin Exp Med 8(2):2317–2325
Jung KH, Yoo W, Stevenson HL, Deshpande D, Shen H, Gagea M, Yoo SY, Wang J, Eckols TK, Bharadwaj U, Tweardy DJ, Beretta L (2017) Multifunctional Effects of a Small-Molecule STAT3 Inhibitor on NASH and Hepatocellular Carcinoma in Mice. Clinical cancer research : an official journal of the American Association for Cancer Research 23(18):5537–5546. https://doi.org/10.1158/1078-0432.Ccr-16-2253
Giannelli G, Koudelkova P, Dituri F, Mikulits W (2016) Role of epithelial to mesenchymal transition in hepatocellular carcinoma. J Hepatol 65(4):798–808. https://doi.org/10.1016/j.jhep.2016.05.007
Xu J, Lamouille S, Derynck R (2009) TGF-beta-induced epithelial to mesenchymal transition. Cell Res 19(2):156–172. https://doi.org/10.1038/cr.2009.5
Cao ZQ, Wang Z, Leng P (2019) Aberrant N-cadherin expression in cancer. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie 118:109320. https://doi.org/10.1016/j.biopha.2019.109320
Li G, Satyamoorthy K, Herlyn M (2001) N-cadherin-mediated intercellular interactions promote survival and migration of melanoma cells. Can Res 61(9):3819–3825
Nieman MT, Prudoff RS, Johnson KR, Wheelock MJ (1999) N-cadherin promotes motility in human breast cancer cells regardless of their E-cadherin expression. J Cell Biol 147(3):631–644. https://doi.org/10.1083/jcb.147.3.631
Gong J, Zheng Y, Wang Y, Sheng W, Li Y, Liu X, Si S, Shao R, Zhen Y (2018) A new compound of thiophenylated pyridazinone IMB5043 showing potent antitumor efficacy through ATM-Chk2 pathway. PLoS ONE 13(2):e0191984. https://doi.org/10.1371/journal.pone.0191984
Zheng YB, Gong JH, Liu XJ, Li Y, Zhen YS (2017) A CD13-targeting peptide integrated protein inhibits human liver cancer growth by killing cancer stem cells and suppressing angiogenesis. Mol Carcinog 56(5):1395–1404. https://doi.org/10.1002/mc.22600
Kinner A, Wu W, Staudt C, Iliakis G (2008) Gamma-H2AX in recognition and signaling of DNA double-strand breaks in the context of chromatin. Nucleic Acids Res 36(17):5678–5694. https://doi.org/10.1093/nar/gkn550
Mendonsa AM, Na TY, Gumbiner BM (2018) E-cadherin in contact inhibition and cancer. Oncogene 37(35):4769–4780. https://doi.org/10.1038/s41388-018-0304-2
Wells A, Yates C, Shepard CR (2008) E-cadherin as an indicator of mesenchymal to epithelial reverting transitions during the metastatic seeding of disseminated carcinomas. Clin Exp Metas 25(6):621–628. https://doi.org/10.1007/s10585-008-9167-1
Loh CY, Chai JY, Tang TF, Wong WF, Sethi G, Shanmugam MK, Chong PP, Looi CY (2019) The E-Cadherin and N-Cadherin Switch in Epithelial-to-Mesenchymal Transition: Signaling, Therapeutic Implications, and Challenges. Cells 8(10). https://doi.org/10.3390/cells8101118
Roos WP, Kaina B (2006) DNA damage-induced cell death by apoptosis. Trends Mol Med 12(9):440–450. https://doi.org/10.1016/j.molmed.2006.07.007
Smith J, Tho LM, Xu N, Gillespie DA (2010) The ATM-Chk2 and ATR-Chk1 pathways in DNA damage signaling and cancer. Adv Cancer Res 108:73–112. https://doi.org/10.1016/b978-0-12-380888-2.00003-0
White RR, Vijg J (2016) Do DNA Double-Strand Breaks Drive Aging? Mol Cell 63(5):729–738. https://doi.org/10.1016/j.molcel.2016.08.004
Amir Mishan M, Rezaei Kanavi M, Shahpasand K, Ahmadieh H (2019) Pathogenic Tau Protein Species: Promising Therapeutic Targets for Ocular Neurodegenerative Diseases. J Ophthalmic Vis Res 14(4):491–505. https://doi.org/10.18502/jovr.v14i4.5459
Gu Y, Oyama F, Ihara Y (1996) Tau is widely expressed in rat tissues. J Neurochem 67(3):1235–1244. https://doi.org/10.1046/j.1471-4159.1996.67031235.x
Papin S, Paganetti P (2020) Emerging Evidences for an Implication of the Neurodegeneration-Associated Protein TAU in Cancer. Brain sciences 10(11). https://doi.org/10.3390/brainsci10110862
Baquero MT, Lostritto K, Gustavson MD, Bassi KA, Appia F, Camp RL, Molinaro AM, Harris LN, Rimm DL (2011) Evaluation of prognostic and predictive value of microtubule associated protein tau in two independent cohorts. Breast cancer research : BCR 13(5):R85. https://doi.org/10.1186/bcr2937
Smoter M, Bodnar L, Grala B, Stec R, Zieniuk K, Kozlowski W, Szczylik C (2013) Tau protein as a potential predictive marker in epithelial ovarian cancer patients treated with paclitaxel/platinum first-line chemotherapy. Journal of experimental & clinical cancer research : CR 32(1):25. https://doi.org/10.1186/1756-9966-32-25
Talbot SR, Biernot S, Bleich A, van Dijk RM, Ernst L, Häger C, Helgers SOA, Koegel B, Koska I, Kuhla A, Miljanovic N, Müller-Graff FT, Schwabe K, Tolba R, Vollmar B, Weegh N, Wölk T, Wolf F, Wree A, Zieglowski L, Potschka H, Zechner D (2020) Defining body-weight reduction as a humane endpoint: a critical appraisal. Lab Anim 54(1):99–110. https://doi.org/10.1177/0023677219883319
Acknowledgements
Not applicable.
Funding
This work was supported by the CAMS Innovation Fund for Medical Sciences (No. 2021-I2M-1-030) and National Natural Science Foundation of China (No. 81201665 and 81972325).
Author information
Authors and Affiliations
Contributions
X.L., Q.Z. and Y.Q.D. performed the experiments, and analyzed the data. J.H.G. designed and performed the experiments, analyzed the data, and revised the manuscript. L.J.Y. revised the manuscript. Y.B.Z. designed the experiments, analyzed the data, and wrote the manuscript. T.Y. performed the analysis and revised the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Animal experiments were approved by the ethics committee for animal experiments of the Institute of Medicinal Biotechnology, Chinese Academy of Medical Sciences (IMBF20161101D7).
Informed consent
Not applicable.
Consent for publication
Not applicable.
Research involving Human Participants and/or Animals
Animal experiments were conducted in accordance with the ethical standards of the ethics committee for animal experiments of the Institute of Medicinal Biotechnology, Chinese Academy of Medical Sciences (IMBF20161101D7).
Conflict of interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lv, X., Zhao, Q., Dong, Y. et al. IMB5036, a novel pyridazinone compound, inhibits hepatocellular carcinoma growth and metastasis. Invest New Drugs 40, 487–496 (2022). https://doi.org/10.1007/s10637-021-01210-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-021-01210-6