Summary
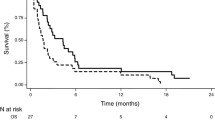
Purpose: The aim of this study was to evaluate the efficacy and tolerability of S-IROX and modified FOLFIRINOX (mFFX) after gemcitabine plus nab-paclitaxel for advanced pancreatic cancer (PC) in the real world setting. Methods: Consecutive patients receiving S-IROX or mFFX as a second-line chemotherapy for advanced PC refractory to gemcitabine plus nab-paclitaxel were retrospectively studied. Patients were treated every 2 weeks: S-1 40 mg/m2 was administered orally twice daily on days 1 to 7 in S-IROX and 5-fluorouracil 2400 mg/m2 was intravenously administered for 46 h without bolus infusion in mFFX, in addition to intravenous oxaliplatin 85 mg/m2 and irinotecan 150 mg/m2 on day 1 in both regimens. Results: Fifty-four patients with advanced PC who received S-IROX (n = 19) or mFFX (n = 35) were retrospectively studied. The disease control rate and response rate were 73.7% and 10.5% in the S-IROX group and 62.2% and 2.7% in the mFFX group, respectively. The median progression free survival (PFS) was 7.8 and 5.7 months in the S-IROX and mFFX groups (p = 0.24). The median overall survival (OS) was 14.2 and 11.5 months in the S-IROX and mFFX groups (p = 0.34). There were no significant differences in the incidences of grade 3–4 adverse effects. The subgroup analyses suggested S-IROX demonstrated favorable OS in patients with PFS ≥6 months of first-line gemcitabine plus nab-paclitaxel (p for interaction = 0.02). Conclusions: S-IROX and mFFX were similarly tolerable and effective as a second-line chemotherapy in patients with PC refractory to gemcitabine plus nab-paclitaxel.






Similar content being viewed by others
References
Sasaki T, Kanata R, Yamada I, Matsuyama M, Ozaka M, Sasahira N (2019) Improvement of treatment outcomes for metastatic pancreatic Cancer: a real-world data analysis. In Vivo 33(1):271–276. https://doi.org/10.21873/invivo.11471
Nakai Y, Isayama H, Sasaki T, Sasahira N, Tsujino T, Toda N, Kogure H, Matsubara S, Ito Y, Togawa O, Arizumi T, Hirano K, Tada M, Omata M, Koike K (2012) A multicentre randomised phase II trial of gemcitabine alone vs gemcitabine and S-1 combination therapy in advanced pancreatic cancer: GEMSAP study. Br J Cancer 106(12):1934–1939. https://doi.org/10.1038/bjc.2012.183
Ueno H, Ioka T, Ikeda M, Ohkawa S, Yanagimoto H, Boku N, Fukutomi A, Sugimori K, Baba H, Yamao K, Shimamura T, Sho M, Kitano M, Cheng AL, Mizumoto K, Chen JS, Furuse J, Funakoshi A, Hatori T, Yamaguchi T, Egawa S, Sato A, Ohashi Y, Okusaka T, Tanaka M (2013) Randomized phase III study of gemcitabine plus S-1, S-1 alone, or gemcitabine alone in patients with locally advanced and metastatic pancreatic cancer in Japan and Taiwan: GEST study. J Clin Oncol 31(13):1640–1648. https://doi.org/10.1200/JCO.2012.43.3680
Saito K, Isayama H, Nakai Y, Takahara N, Ishigaki K, Takeda T, Hakuta R, Saito T, Uchino R, Kishikawa T, Hamada T, Mizuno S, Sasaki T, Kogure H, Matsubara S, Yamamoto N, Ijichi H, Tateishi K, Tada M, Koike K (2018) A phase II trial of gemcitabine, S-1 and LV combination (GSL) therapy in patients with advanced pancreatic cancer. Investig New Drugs 37:338–344. https://doi.org/10.1007/s10637-018-0691-9
Moore MJ, Goldstein D, Hamm J, Figer A, Hecht JR, Gallinger S, Au HJ, Murawa P, Walde D, Wolff RA, Campos D, Lim R, Ding K, Clark G, Voskoglou-Nomikos T, Ptasynski M, Parulekar W, National Cancer Institute of Canada Clinical Trials G (2007) Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada clinical trials group. J Clin Oncol 25(15):1960–1966. https://doi.org/10.1200/JCO.2006.07.9525
Conroy T, Desseigne F, Ychou M, Bouche O, Guimbaud R, Becouarn Y, Adenis A, Raoul JL, Gourgou-Bourgade S, de la Fouchardiere C, Bennouna J, Bachet JB, Khemissa-Akouz F, Pere-Verge D, Delbaldo C, Assenat E, Chauffert B, Michel P, Montoto-Grillot C, Ducreux M (2011) FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 364(19):1817–1825. https://doi.org/10.1056/NEJMoa1011923
Von Hoff DD, Ervin T, Arena FP, Chiorean EG, Infante J, Moore M, Seay T, Tjulandin SA, Ma WW, Saleh MN, Harris M, Reni M, Dowden S, Laheru D, Bahary N, Ramanathan RK, Tabernero J, Hidalgo M, Goldstein D, Van Cutsem E, Wei X, Iglesias J, Renschler MF (2013) Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med 369(18):1691–1703. https://doi.org/10.1056/NEJMoa1304369
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology. Pancreatic Adenocarcinoma. Ver- sion 2. 2016 ed 2016. Available from http://www.nccnorg/professionals/physician_gls/pdf/pancreatic.pdf Accessed January 14, 2016 -
Oettle H, Riess H, Stieler JM, Heil G, Schwaner I, Seraphin J, Gorner M, Molle M, Greten TF, Lakner V, Bischoff S, Sinn M, Dorken B, Pelzer U (2014) Second-line oxaliplatin, folinic acid, and fluorouracil versus folinic acid and fluorouracil alone for gemcitabine-refractory pancreatic cancer: outcomes from the CONKO-003 trial. J Clin Oncol 32(23):2423–2429. https://doi.org/10.1200/JCO.2013.53.6995
Wang-Gillam A, Li CP, Bodoky G, Dean A, Shan YS, Jameson G, Macarulla T, Lee KH, Cunningham D, Blanc JF, Hubner RA, Chiu CF, Schwartsmann G, Siveke JT, Braiteh F, Moyo V, Belanger B, Dhindsa N, Bayever E, Von Hoff DD, Chen LT (2016) Nanoliposomal irinotecan with fluorouracil and folinic acid in metastatic pancreatic cancer after previous gemcitabine-based therapy (NAPOLI-1): a global, randomised, open-label, phase 3 trial. Lancet 387(10018):545–557. https://doi.org/10.1016/s0140-6736(15)00986-1
Sawada M, Kasuga A, Mie T, Furukawa T, Taniguchi T, Fukuda K, Yamada Y, Takeda T, Kanata R, Matsuyama M, Sasaki T, Ozaka M, Sasahira N (2020) Modified FOLFIRINOX as a second-line therapy following gemcitabine plus nab-paclitaxel therapy in metastatic pancreatic cancer. BMC Cancer 20(1):449. https://doi.org/10.1186/s12885-020-06945-8
Nakai Y, Isayama H, Sasaki T, Sasahira N, Kogure H, Hirano K, Tsujino T, Ijichi H, Tateishi K, Tada M, Omata M, Koike K (2010) Impact of S-1 in patients with gemcitabine-refractory pancreatic cancer in Japan. Jpn J Clin Oncol 40(8):774–780. https://doi.org/10.1093/jjco/hyq059
Hamada C, Okusaka T, Ikari T, Isayama H, Furuse J, Ishii H, Nakai Y, Imai S, Okamura S (2017) Efficacy and safety of gemcitabine plus S-1 in pancreatic cancer: a pooled analysis of individual patient data. Br J Cancer 116(6):1544–1550. https://doi.org/10.1038/bjc.2017.128
Ohkawa S, Okusaka T, Isayama H, Fukutomi A, Yamaguchi K, Ikeda M, Funakoshi A, Nagase M, Hamamoto Y, Nakamori S, Tsuchiya Y, Baba H, Ishii H, Omuro Y, Sho M, Matsumoto S, Yamada N, Yanagimoto H, Unno M, Ichikawa Y, Takahashi S, Watanabe G, Wakabayashi G, Egawa N, Tsuda M, Hosotani R, Hamada C, Hyodo I (2015) Randomised phase II trial of S-1 plus oxaliplatin vs S-1 in patients with gemcitabine-refractory pancreatic cancer. Br J Cancer 112(9):1428–1434. https://doi.org/10.1038/bjc.2015.103
Takahara N, Isayama H, Nakai Y, Ishigami H, Satoi S, Mizuno S, Kogure H, Matsubara S, Yamamoto N, Yamaguchi H, Tada M, Kitayama J, Watanabe T, Koike K (2016) Intravenous and intraperitoneal paclitaxel with S-1 for treatment of refractory pancreatic cancer with malignant ascites. Investig New Drugs 34(5):636–642. https://doi.org/10.1007/s10637-016-0369-0
Ioka T, Ueno M, Ueno H, Park JO, Chang HM, Sasahira N, Kanai M, Chung IJ, Ikeda M, Nakamori S, Mizuno N, Omuro Y, Yamaguchi T, Hara H, Sugimori K, Furuse J, Maguchi H, Furukawa M, Fukuzawa K, Kim JS, Yukisawa S, Takeuchi M, Okusaka T, Boku N, Hyodo I (2019) TAS-118 (S-1 plus leucovorin) versus S-1 in patients with gemcitabine-refractory advanced pancreatic cancer: a randomised, open-label, phase 3 study (GRAPE trial). Eur J Cancer 106:78–88. https://doi.org/10.1016/j.ejca.2018.10.004
Kimura G, Takahashi H, Umemoto K, Watanabe K, Sasaki M, Hashimoto Y, Imaoka H, Ohno I, Mitsunaga S, Ikeda M (2017) Efficacy of S-1 compared to modified FOLFIRINOX as second-line chemotherapy regimens after gemcitabine plus nab-paclitaxel for patients with metastatic pancreatic cancer. J Clin Oncol 35(4_suppl):449–449. https://doi.org/10.1200/JCO.2017.35.4_suppl.449
Yanagimoto H, Ishii H, Nakai Y, Ozaka M, Ikari T, Koike K, Ueno H, Ioka T, Satoi S, Sho M, Okusaka T, Tanaka M, Shimokawa T, Kwon AH, Isayama H (2014) Improved survival with combined gemcitabine and S-1 for locally advanced pancreatic cancer: pooled analysis of three randomized studies. J Hepatobiliary Pancreat Sci 21(10):761–766. https://doi.org/10.1002/jhbp.130
Sasaki M, Ueno H, Shiba S, Hosoi H, Ohba A, Sakamoto Y, Kondo S, Morizane C, Okusaka T (2015) Phase I study of S-1, irinotecan plus oxaliplatin combination therapy for advanced pancreatic cancer. Ann Oncol 26
Akahori T, Sho M, Yanagimoto H, Satoi S, Nagai M, Nishiwada S, Nakagawa K, Nakamura K, Yamamoto T, Hirooka S, Yamaki S, Ikeda N (2019) Phase II study of the triple combination chemotherapy of SOXIRI (S-1/Oxaliplatin/Irinotecan) in patients with Unresectable pancreatic ductal adenocarcinoma. Oncologist 24(6):749–e224. https://doi.org/10.1634/theoncologist.2018-0900
Ozaka M, Ishii H, Sato T, Ueno M, Ikeda M, Uesugi K, Sata N, Miyashita K, Mizuno N, Tsuji K, Okusaka T, Furuse J (2018) A phase II study of modified FOLFIRINOX for chemotherapy-naive patients with metastatic pancreatic cancer. Cancer Chemother Pharmacol 81(6):1017–1023. https://doi.org/10.1007/s00280-018-3577-9
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45(2):228–247. https://doi.org/10.1016/j.ejca.2008.10.026
National Cancer Institute USDOHAHS. Common Terminology Criteria for Adverse Events (CTCAE), Version 4.0. (2009). https://www.evsncinihgov/ftp1/CTCAE/CTCAE_403_20100614_QuickReference_5x7.pdf
Kang J, Hwang I, Yoo C, Kim KP, Jeong JH, Chang HM, Lee SS, Park DH, Song TJ, Seo DW, Lee SK, Kim MH, Hong SM, Shin SH, Hwang DW, Song KB, Lee JH, Kim SC, Ryoo BY (2018) Nab-paclitaxel plus gemcitabine versus FOLFIRINOX as the first-line chemotherapy for patients with metastatic pancreatic cancer: retrospective analysis. Investig New Drugs 36:732–741. https://doi.org/10.1007/s10637-018-0598-5
Tahara J, Shimizu K, Otsuka N, Akao J, Takayama Y, Tokushige K (2018) Gemcitabine plus nab-paclitaxel vs. FOLFIRINOX for patients with advanced pancreatic cancer. Cancer Chemother Pharmacol 82:245–250. https://doi.org/10.1007/s00280-018-3611-y
Hamada T, Nakai Y, Yasunaga H, Isayama H, Matsui H, Takahara N, Sasaki T, Takagi K, Watanabe T, Yagioka H, Kogure H, Arizumi T, Yamamoto N, Ito Y, Hirano K, Tsujino T, Tada M, Koike K (2014) Prognostic nomogram for nonresectable pancreatic cancer treated with gemcitabine-based chemotherapy. Br J Cancer 110(8):1943–1949. https://doi.org/10.1038/bjc.2014.131
Inoue D, Ozaka M, Matsuyama M, Yamada I, Takano K, Saiura A, Ishii H (2015) Prognostic value of neutrophil-lymphocyte ratio and level of C-reactive protein in a large cohort of pancreatic cancer patients: a retrospective study in a single institute in Japan. Jpn J Clin Oncol 45(1):61–66. https://doi.org/10.1093/jjco/hyu159
Chiorean EG, Von Hoff DD, Tabernero J, El-Maraghi R, Ma WW, Reni M, Harris M, Whorf R, Liu H, Li JS, Manax V, Romano A, Lu B, Goldstein D (2016) Second-line therapy after nab-paclitaxel plus gemcitabine or after gemcitabine for patients with metastatic pancreatic cancer. Br J Cancer 115(2):188–194. https://doi.org/10.1038/bjc.2016.185
Ioka T, Komatsu Y, Mizuno N, Tsuji A, Ohkawa S, Tanaka M, Iguchi H, Ishiguro A, Kitano M, Satoh T, Yamaguchi T, Takeda K, Kida M, Eguchi K, Ito T, Munakata M, Itoi T, Furuse J, Hamada C, Sakata Y (2017) Randomised phase II trial of irinotecan plus S-1 in patients with gemcitabine-refractory pancreatic cancer. Br J Cancer 116(4):464–471. https://doi.org/10.1038/bjc.2016.436
Reni M, Berardi R, Mambrini A, Pasetto L, Cereda S, Ferrari VD, Cascinu S, Cantore M, Mazza E, Grisanti S (2008) A multi-Centre retrospective review of second-line therapy in advanced pancreatic adenocarcinoma. Cancer Chemother Pharmacol 62(4):673–678. https://doi.org/10.1007/s00280-007-0653-y
Macarulla T, Blanc JF, Wang-Gillam A, Chen LT, Siveke JT, Mirakhur B, Chen J, de Jong FA (2019) Liposomal irinotecan and 5-fluorouracil/leucovorin in older patients with metastatic pancreatic cancer - a subgroup analysis of the pivotal NAPOLI-1 trial. J Geriatr Oncol 10(3):427–435. https://doi.org/10.1016/j.jgo.2019.02.011
Funding
This study has never been funded by any foundation, company, industry, or other external source.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Yousuke Nakai received research funding from Taiho Pharmaceutical Co and Yakult Honsha Co. All remaining authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the Ethical Committee of the University of Tokyo Hospital.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saito, K., Nakai, Y., Takahara, N. et al. A retrospective comparative study of S-IROX and modified FOLFIRINOX for patients with advanced pancreatic cancer refractory to gemcitabine plus nab-paclitaxel. Invest New Drugs 39, 605–613 (2021). https://doi.org/10.1007/s10637-020-01022-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-020-01022-0