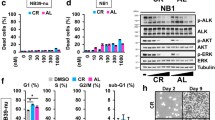
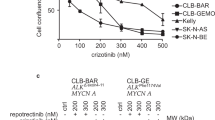
Summary
Anaplastic lymphoma kinase (ALK), which belongs to the insulin receptor tyrosine kinase superfamily, plays an important role in nervous system development. Due to chromosomal translocations, point mutations, and gene amplification, constitutively activated ALK has been implicated in a variety of human cancers, including anaplastic large-cell lymphoma (ALCL), non-small cell lung cancer, and neuroblastoma. We evaluated the anti-cancer activity of the ALK inhibitor KRCA-0008 using ALCL cell lines that express NPM (nucleophosmin)-ALK. KRCA-0008 strongly suppressed the proliferation and survival of NPM-ALK-positive ALCL cells. Additionally, it induced G0/G1 cell cycle arrest and apoptosis by blocking downstream signals including STAT3, Akt, and ERK1/2. Tumor growth was strongly suppressed in mice inoculated with Karpas-299 tumor xenografts and orally treated with KRCA-0008 (50 mg/kg, BID) for 2 weeks. Our results suggest that KRCA-0008 will be useful in further investigations of ALK signaling, and may provide therapeutic opportunities for NPM-ALK-positive ALCL patients.







Similar content being viewed by others
References
Webb TR, Slavish J, George RE, Look AT, Xue L, Jiang Q, Cui X, Rentrop WB, Morris SW (2009) Anaplastic lymphoma kinase: role in cancer pathogenesis and small-molecule inhibitor development for therapy. Expert Rev Anticancer Ther 9:331–356
Iwahara T, Fujimoto J, Wen D, Cupples R, Bucay N, Arakawa T, Mori S, Ratzkin B, Yamamoto T (1997) Molecular characterization of ALK, a receptor tyrosine kinase expressed specifically in the nervous system. Oncogene 14:439–449
Bilsland JG, Wheeldon A, Mead A, Znamenskiy P, Almond S, Waters KA, Thakur M, Beaumont V, Bonnert TP, Heavens R, Whiting P, McAllister G, Munoz-Sanjuan I (2008) Behavioral and neurochemical alterations in mice deficient in anaplastic lymphoma kinase suggest therapeutic potential for psychiatric indications. Neuropsychopharmacology 33:685–700
Roskoski R Jr (2013) Anaplastic lymphoma kinase (ALK): structure, oncogenic activation, and pharmacological inhibition. Pharmacol Res 68:68–94
Stein H, Mason DY, Gerdes J, O'Connor N, Wainscoat J, Pallesen G, Gatter K, Falini B, Delsol G, Lemke H, Schwarting R, Lennert K (1985) The expression of the Hodgkin's disease associated antigen Ki-1 in reactive and neoplastic lymphoid tissue: evidence that reed-Sternberg cells and histiocytic malignancies are derived from activated lymphoid cells. Blood 66:848–858
Gascoyne RD, Aoun P, Wu D, Chhanabhai M, Skinnider BF, Greiner TC, Morris SW, Connors JM, Vose JM, Viswanatha DS, Coldman A, Weisenburger DD (1999) Prognostic significance of anaplastic lymphoma kinase (ALK) protein expression in adults with anaplastic large cell lymphoma. Blood 93:3913–3921
Morris SW, Kirstein MN, Valentine MB, Dittmer K, Shapiro DN, Look AT, Saltman DL (1995) Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin's lymphoma. Science 267:316–317
Falini B, Pulford K, Pucciarini A, Carbone A, De Wolf-Peeters C, Cordell J, Fizzotti M, Santucci A, Pelicci PG, Pileri S, Campo E, Ott G, Delsol G, Mason DY (1999) Lymphomas expressing ALK fusion protein(s) other than NPM-ALK. Blood 94:3509–3515
Pulford K, Morris SW, Turturro F (2004) Anaplastic lymphoma kinase proteins in growth control and cancer. J Cell Physiol 199:330–358
Duyster J, Bai RY, Morris SW (2001) Translocations involving anaplastic lymphoma kinase (ALK). Oncogene 20:5623–5637
Drexler HG, Gignac SM, von Wasielewski R, Werner M, Dirks WG (2000) Pathobiology of NPM-ALK and variant fusion genes in anaplastic large cell lymphoma and other lymphomas. Leukemia 14:1533–1559
Bischof D, Pulford K, Mason DY, Morris SW (1997) Role of the nucleophosmin (NPM) portion of the non-Hodgkin's lymphoma-associated NPM-anaplastic lymphoma kinase fusion protein in oncogenesis. Mol Cell Biol 17:2312–2325
Bai RY, Dieter P, Peschel C, Morris SW, Duyster J (1998) Nucleophosmin-anaplastic lymphoma kinase of large-cell anaplastic lymphoma is a constitutively active tyrosine kinase that utilizes phospholipase C-gamma to mediate its mitogenicity. Mol Cell Biol 18:6951–6961
Bai RY, Ouyang T, Miething C, Morris SW, Peschel C, Duyster J (2000) Nucleophosmin-anaplastic lymphoma kinase associated with anaplastic large-cell lymphoma activates the phosphatidylinositol 3-kinase/Akt antiapoptotic signaling pathway. Blood 96:4319–4327
Kuefer MU, Look AT, Pulford K, Behm FG, Pattengale PK, Mason DY, Morris SW (1997) Retrovirus-mediated gene transfer of NPM-ALK causes lymphoid malignancy in mice. Blood 90:2901–2910
Katayama R, Lovly CM, Shaw AT (2015) Therapeutic targeting of anaplastic lymphoma kinase in lung cancer: a paradigm for precision cancer medicine. Clin Cancer Res 21:2227–2235
Kwak EL, Bang YJ, Camidge DR, Shaw AT, Solomon B, Maki RG, Ou SH, Dezube BJ, Jänne PA, Costa DB, Varella-Garcia M, Kim WH, Lynch TJ, Fidias P, Stubbs H, Engelman JA, Sequist LV, Tan W, Gandhi L, Mino-Kenudson M, Wei GC, Shreeve SM, Ratain MJ, Settleman J, Christensen JG, Haber DA, Wilner K, Salgia R, Shapiro GI, Clark JW, Iafrate AJ (2010) Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med 363:1693–1703
Katayama R, Shaw AT, Khan TM, Mino-Kenudson M, Solomon BJ, Halmos B, Jessop NA, Wain JC, Yeo AT, Benes C, Drew L, Saeh JC, Crosby K, Sequist LV, Iafrate AJ, Engelman JA (2012) Mechanisms of acquired crizotinib resistance in ALK-rearranged lung cancers. Sci Transl Med 4:120ra17
Lovly CM, Pao W (2012) Escaping ALK inhibition: mechanisms of and strategies to overcome resistance. Sci Transl Med 4:120ps2
Peters S, Camidge DR, Shaw AT, Gadgeel S, Ahn JS, Kim DW, Ou SI, Pérol M, Dziadziuszko R, Rosell R, Zeaiter A, Mitry E, Golding S, Balas B, Noe J, Morcos PN, Mok T, ALEX Trial Investigators (2017) Alectinib versus Crizotinib in untreated ALK-positive non-small-cell lung Cancer. N Engl J Med 377:829–838
Shaw AT, Kim TM, Crinò L, Gridelli C, Kiura K, Liu G, Novello S, Bearz A, Gautschi O, Mok T, Nishio M, Scagliotti G, Spigel DR, Deudon S, Zheng C, Pantano S, Urban P, Massacesi C, Viraswami-Appanna K, Felip E (2017) Ceritinib versus chemotherapy in patients with ALK-rearranged non-small-cell lung cancer previously given chemotherapy and crizotinib (ASCEND-5): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol 18:874–886
Gettinger SN, Bazhenova LA, Langer CJ, Salgia R, Gold KA, Rosell R, Shaw AT, Weiss GJ, Tugnait M, Narasimhan NI, Dorer DJ, Kerstein D, Rivera VM, Clackson T, Haluska FG, Camidge DR (2016) Activity and safety of brigatinib in ALK-rearranged non-small-cell lung cancer and other malignancies: a single-arm, open-label, phase 1/2 trial. Lancet Oncol 17:1683–1696
Shaw AT, Friboulet L, Leshchiner I, Gainor JF, Bergqvist S, Brooun A, Burke BJ, Deng YL, Liu W, Dardaei L, Frias RL, Schultz KR, Logan J, James LP, Smeal T, Timofeevski S, Katayama R, Iafrate AJ, Le L, McTigue M, Getz G, Johnson TW, Engelman JA (2016) Resensitization to Crizotinib by the Lorlatinib ALK resistance mutation L1198F. N Engl J Med 374:54–61
Galkin AV, Melnick JS, Kim S, Hood TL, Li N, Li L, Xia G, Steensma R, Chopiuk G, Jiang J, Wan Y, Ding P, Liu Y, Sun F, Schultz PG, Gray NS, Warmuth M (2007) Identification of NVP-TAE684, a potent, selective, and efficacious inhibitor of NPM-ALK. Proc Natl Acad Sci U S A 104:270–275
Wan W, Albom MS, Lu L, Quail MR, Becknell NC, Weinberg LR, Reddy DR, Holskin BP, Angeles TS, Underiner TL, Meyer SL, Hudkins RL, Dorsey BD, Ator MA, Ruggeri BA, Cheng M (2006) Anaplastic lymphoma kinase activity is essential for the proliferation and survival of anaplastic large-cell lymphoma cells. Blood 107:1617–1623
Piva R, Chiarle R, Manazza AD, Taulli R, Simmons W, Ambrogio C, D'Escamard V, Pellegrino E, Ponzetto C, Palestro G, Inghirami G (2006) Ablation of oncogenic ALK is a viable therapeutic approach for anaplastic large-cell lymphomas. Blood 107:689–697
Gambacorti-Passerini C, Messa C, Pogliani EM (2011) Crizotinib in anaplastic large-cell lymphoma. N Engl J Med 364:775–776
ClinicalTrials.gov (2016) Ceritinib With Brentuximab Vedotin in Treating Patients with ALK-Positive Anaplastic Large Cell Lymphoma. https://clinicaltrials.gov/ct2/show/NCT02729961. Accessed 14 June 2019
Nagai H, Fukano R, Sekimizu M, Kada A, M Saito A, Asada R, Mori T (2017) Phase II trial of CH5424802 (alectinib hydrochloride) for recurrent or refractory ALK-positive anaplastic large cell lymphoma: study protocol for a non-randomized non-controlled trial. Nagoya J Med Sci 79:407–413
ClinicalTrials.gov (2018) A Study of Oral Lorlatinib in Patients With Relapsed ALK Positive Lymphoma. https://clinicaltrials.gov/ct2/show/NCT03505554. Accessed 14 June 2019
ClinicalTrials.gov (2018) Brigatinib in Relapsed or Refractory ALK-Positive Anaplastic Large Cell Lymphoma. https://clinicaltrials.gov/ct2/show/NCT03719898. Accessed 14 June 2019
Park CH, Choe H, Jang IY, Kwon SY, Latif M, Lee HK, Lee HJ, Yang EH, Yun JI, Chae CH, Cho SY, Choi SU, Ha JD, Jung H, Kim HR, Kim P, Lee CO, Yun CS, Lee K (2013) Novel bis-ortho-alkoxy-Para-piperazinesubstituted-2,4-dianilinopyrimidines (KRCA-0008) as potent and selective ALK inhibitors for anticancer treatment. Bioorg Med Chem Lett 23:6192–6196
Lee HJ, Latif M, Choe H, Ali I, Lee HK, Yang EH, Yun JI, Chae CH, Jung JK, Kim HR, Lee CO, Park CH, Lee K (2014) ALK inhibitors of bis-ortho-alkoxy-Para-piperazinesubstituted-pyrimidines and -triazines for cancer treatment. Arch Pharm Res 37:1130–1138
Cui JJ, Tran-Dubé M, Shen H, Nambu M, Kung PP, Pairish M, Jia L, Meng J, Funk L, Botrous I, McTigue M, Grodsky N, Ryan K, Padrique E, Alton G, Timofeevski S, Yamazaki S, Li Q, Zou H, Christensen J, Mroczkowski B, Bender S, Kania RS, Edwards MP (2011) Structure based drug design of crizotinib (PF-02341066), a potent and selective dual inhibitor of mesenchymal-epithelial transition factor (c-MET) kinase and anaplastic lymphoma kinase (ALK). J Med Chem 54:6342–6363
Ferreri AJ, Govi S, Pileri SA, Savage KJ (2012) Anaplastic large cell lymphoma, ALK-positive. Crit Rev Oncol Hematol 83:293–302
Sitzia J, North C, Stanley J, Winterberg N (1997) Side effects of CHOP in the treatment of non-hodgkin's lymphoma. Cancer Nurs 20:430–439
Damm-Welk C, Mussolin L, Zimmermann M, Pillon M, Klapper W, Oschlies I, d'Amore ES, Reiter A, Woessmann W, Rosolen A (2014) Early assessment of minimal residual disease identifies patients at very high relapse risk in NPM-ALK-positive anaplastic large-cell lymphoma. Blood 123:334–337
Zamo A, Chiarle R, Piva R, Howes J, Fan Y, Chilosi M, Levy DE, Inghirami G (2002) Anaplastic lymphoma kinase (ALK) activates Stat3 and protects hematopoietic cells from cell death. Oncogene 21:1038–1047
Amin HM, McDonnell TJ, Ma Y, Lin Q, Fujio Y, Kunisada K, Leventaki V, Das P, Rassidakis GZ, Cutler C, Medeiros LJ, Lai R (2004) Selective inhibition of STAT3 induces apoptosis and G(1) cell cycle arrest in ALK-positive anaplastic large cell lymphoma. Oncogene 23:5426–5434
Taylor RC, Cullen SP, Martin SJ (2008) Apoptosis: controlled demolition at the cellular level. Nat Rev Mol Cell Biol 9:231–241
Acknowledgements
This work was taken in part from the master’s thesis of J. Hwang and the doctoral thesis of I. Song. We thank the Central Laboratory of Kangwon National University for providing us with technical assistance on the microscopic and cytometry experiments.
Funding
This work was financially supported by National Research Foundation of Korea (NRF) grants (NRF-2012M3A9A9054971 and NRF-2018R1A2B6001314) funded by the Korean government (MSIT).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not describe any studies that include human participants performed by any of the authors.
Informed consent
Formal consent is not required for this type of study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hwang, J., Song, I., Lee, K. et al. KRCA-0008 suppresses ALK-positive anaplastic large-cell lymphoma growth. Invest New Drugs 38, 1282–1291 (2020). https://doi.org/10.1007/s10637-020-00896-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-020-00896-4