Summary
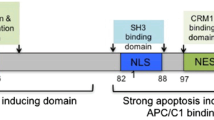
Apoptin is a nonstructural protein encoded by one of the three open reading frames of the chicken anemia virus genome. It has attracted a great deal of interest due to its ability to induce apoptosis in multiple transformed and malignant mammalian cell lines without affecting primary and non-transformed cells. However, the use of Apoptin as an anticancer drug is restricted by its strong tendency to aggregate. A number of methods to overcome this problem have been proposed, including transduction techniques to deliver the Apoptin gene into tumor cells, but all such methods have certain drawbacks. Here we describe that a truncated variant of Apoptin, lacking residues 1 to 43, is a soluble, non-aggregating protein that maintains most of the biological properties of wild-type Apoptin when transfected into cells. We show that the cytotoxic effect of this variant is also present when it is added exogenously to cancer cells, but not to normal cells. In addition to the interest this protein has attracted as a promising therapeutic strategy, it is also an excellent model to study the structural properties of Apoptin and how they relate to its mechanism of action.




Similar content being viewed by others
References
Prasetyo AA, Kamahora T, Kuroishi A et al (2009) Replication of chicken anemia virus (CAV) requires apoptin and is complemented by VP3 of human torque Teno virus (TTV). Virology 385:85–92. doi:10.1016/j.virol.2008.10.043
Maddika S, Mendoza FJ, Hauff K et al (2006) Cancer-selective therapy of the future: apoptin and its mechanism of action. Cancer Biol Ther 5:10–19. doi:10.4161/cbt.5.1.2400
Poon IKH, Oro C, Dias MM et al (2005) A tumor cell-specific nuclear targeting signal within chicken anemia virus VP3/apoptin. J Virol 79:1339–1341. doi:10.1128/JVI.79.2.1339-1341.2005
Leliveld SR, Zhang Y-H, Rohn JL et al (2003) Apoptin induces tumor-specific apoptosis as a globular multimer. J Biol Chem 278:9042–9051. doi:10.1074/jbc.M210803200
Leliveld SR, Dame RT, Mommaas MA et al (2003) Apoptin protein multimers form distinct higher-order nucleoprotein complexes with DNA. Nucleic Acids Res 31:4805–4813
Danen-Van Oorschot AAAM, Zhang Y-HH, Leliveld SR et al (2003) Importance of nuclear localization of apoptin for tumor-specific induction of apoptosis. J Biol Chem 278:27729–27736. doi:10.1074/jbc.M303114200
Rollano Peñaloza OM, Lewandowska M, Stetefeld J et al (2014) Apoptins: selective anticancer agents. Trends Mol Med 20:519–528. doi:10.1016/j.molmed.2014.07.003
Rohn JL (2002) A tumor-specific kinase activity regulates the viral death protein apoptin. J Biol Chem 277:50820–50827. doi:10.1074/jbc.M208557200
Poon IKH, Oro C, Dias MM et al (2005) Apoptin nuclear accumulation is modulated by a CRM1-recognized nuclear export signal that is active in normal but not in tumor cells. Cancer Res 65:7059–7064. doi:10.1158/0008-5472.CAN-05-1370
Zhou S, Zhang M, Zhang J et al (2012) Mechanisms of apoptin-induced cell death. Med Oncol 29:2985–2991. doi:10.1007/s12032-011-0119-2
Leliveld SR, Dame RT, Rohn JL et al (2004) Apoptin’s functional N- and C-termini independently bind DNA. FEBS Lett 557:155–158
Guelen L, Paterson H, Gäken J et al (2004) TAT-apoptin is efficiently delivered and induces apoptosis in cancer cells. Oncogene 23:1153–1165. doi:10.1038/sj.onc.1207224
Zhang M, Guller S, Huang Y (2007) Method to enhance transfection efficiency of cell lines and placental fibroblasts. Placenta 28:779–782. doi:10.1016/j.placenta.2007.01.012
Castro J, Ribó M, Puig T et al (2012) A cytotoxic ribonuclease reduces the expression level of P-glycoprotein in multidrug-resistant cell lines. Invest New Drugs 30:880–888. doi:10.1007/s10637-011-9636-2
Castro J, Ribó M, Navarro S et al (2011) A human ribonuclease induces apoptosis associated with p21WAF1/CIP1 induction and JNK inactivation. BMC Cancer 11:9. doi:10.1186/1471-2407-11-9
Gras SL, Waddington LJ, Goldie KN (2011) Transmission electron microscopy of amyloid fibrils. Methods Mol Biol 752:197–214. doi:10.1007/978-1-60327-223-0_13
Noteborn MH, Todd D, Verschueren CA et al (1994) A single chicken anemia virus protein induces apoptosis. J Virol 68:346–351
Zhang YH, Leliveld SR, Kooistra K et al (2003) Recombinant apoptin multimers kill tumor cells but are nontoxic and epitope-shielded in a normal-cell-specific fashion. Exp Cell Res 289:36–46. doi:10.1016/S0014-4827(03)00188-5
Ruiz-Martínez S, Pantoja-Uceda D, Castro J et al (2017) Insights into the mechanism of Apoptin’s exquisitely selective anti-tumor action from atomic level characterization of its conformation and dynamics. Arch Biochem Biophys 614:53–64. doi:10.1016/j.abb.2016.12.010
Gualfetti PJ, Iwakura M, Lee JC et al (1999) Apparent radii of the native, stable intermediates and unfolded conformers of the alpha-subunit of tryptophan synthase from E. coli, a TIM barrel protein. Biochemistry 38:13367–13378
Rohn JL, Zhang Y-H, Leliveld SR et al (2005) Relevance of apoptin’s integrity for its functional behavior. J Virol 79:1337–1338. doi:10.1128/JVI.79.2.1337-1338.2005
Pavlakis N, Vogelzang NJ (2006) Ranpirnase-an antitumour ribonuclease: its potential role in malignant mesothelioma. Expert Opin Biol Ther 6:391–399. doi:10.1517/14712598.6.4.391
Zhao J, Gao P, Xiao W et al (2011) A novel human derived cell-penetrating peptide in drug delivery. Mol Biol Rep 38:2649–2656. doi:10.1007/s11033-010-0406-6
Sun J, Yan Y, Wang X-T et al (2009) PTD4-apoptin protein therapy inhibits tumor growth in vivo. Int J Cancer 124:2973–2981. doi:10.1002/ijc.24279
Zhao M, Hu B, Gu Z et al (2013) Degradable polymeric nanocapsule for efficient intracellular delivery of a high molecular weight tumor-selective protein complex. Nano Today 8:11–20. doi:10.1016/j.nantod.2012.12.003
Ring J, Seifert J, Jesch F, Brendel W (1977) Anaphylactoid reactions due to non-immune complex serum protein aggregates. Monogr Allergy 12:27–35
Benito A, Vilanova M, Ribó M (2008) Intracellular routing of cytotoxic pancreatic-type ribonucleases. Curr Pharm Biotechnol 9:169–179
Acknowledgements
We are very grateful to Dr. Malvash Tavassoli (King’s College, UK) for providing us with the Apoptin gene, to Dr. Bruno Beaumelle (CPBS, France) for providing us with K-562 cell line and to Dr. J. L. Corchero (Autonomous University of Barcelona, Spain) for help with the DLS experiment. S. R-M gratefully acknowledges his FPU fellowship from the MEC, Spain.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
The work was supported by grants BFU2009-06935, BIO2013-43517, SAF-2013-49179-C2-2-R, QTC2014-52633-P and UNGI 10-4E-417 from MINECO (Spain) and MPCU2016/18 and SING12/0 from UdG.
Ethical approval
This article does not contain any studies performed by any of the authors involving human or animal participants.
Informed consent
For this type of study, formal consent is not required.
Electronic supplementary material
ESM 1
(DOCX 2694 kb)
Rights and permissions
About this article
Cite this article
Ruiz-Martínez, S., Castro, J., Vilanova, M. et al. A truncated apoptin protein variant selectively kills cancer cells. Invest New Drugs 35, 260–268 (2017). https://doi.org/10.1007/s10637-017-0431-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-017-0431-6