Summary
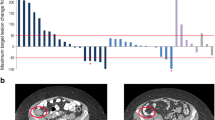
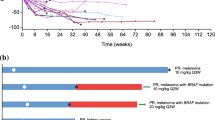
Purpose This first-in-human study evaluated the CD70-targeted antibody-drug conjugate SGN-75 in patients with relapsed or refractory CD70-positive non-Hodgkin lymphoma (NHL) or metastatic renal cell carcinoma (RCC). Methods SGN-75 was administered intravenously to 58 patients (39 RCC, 19 NHL) every 3 weeks (Q3Wk; doses escalated from 0.3 to 4.5 mg/kg) or on Days 1, 8, and 15 of 28-day cycles (weekly; doses of 0.3 or 0.6 mg/kg). Dose-limiting toxicities were evaluated during Cycle 1; treatment response was monitored every 2 cycles. Results The maximum tolerated dose of SGN-75 in RCC patients was 3 mg/kg Q3Wk. Due to toxicity concerns (idiopathic thrombocytopenic purpura in 2 NHL patients treated weekly), dose escalation in the weekly schedule was terminated; no regimen was recommended for NHL patients. The most common adverse events reported in patients treated Q3Wk (N = 47) were fatigue (40 %), dry eye (32 %), nausea (30 %), and thrombocytopenia (26 %). The nadir for thrombocytopenia typically occurred during Cycle 1. Ocular adverse events (e.g., corneal epitheliopathy, dry eye) were reported for 57 % of patients treated Q3Wk and were generally reversible. Antitumor activity in patients treated Q3Wk included 1 complete response, 2 partial responses, and 20 stable disease. SGN-75 exposures were approximately dose proportional, with a mean terminal half-life of 10 days. Substantial depletions of CD70-positive peripheral blood lymphocytes were observed after SGN-75 treatment. Conclusions Modest single-agent activity and generally manageable adverse events were observed in heavily pretreated RCC and NHL patients. Administration Q3Wk was better tolerated than weekly dosing. Targeted ablation of CD70-positive lymphocytes was demonstrated.



Similar content being viewed by others
References
Bowman MR, Crimmins MA, Yetz-Aldape J et al (1994) The cloning of CD70 and its identification as the ligand for CD27. J Immunol 152:1756–61
Bullock TN, Yagita H (2005) Induction of CD70 on dendritic cells through CD40 or TLR stimulation contributes to the development of CD8+ T cell responses in the absence of CD4+ T cells. J Immunol 174:710–7
Goodwin RG, Alderson MR, Smith CA et al (1993) Molecular and biological characterization of a ligand for CD27 defines a new family of cytokines with homology to tumor necrosis factor. Cell 73:447–56
Lens SM, Baars PA, Hooibrink B et al (1997) Antigen-presenting cell-derived signals determine expression levels of CD70 on primed T cells. Immunology 90:38–45
Lens SM, de Jong R, Hooibrink B et al (1996) Phenotype and function of human B cells expressing CD70 (CD27 ligand). Eur J Immunol 26:2964–71
Orengo AM, Cantoni C, Neglia F et al (1997) Reciprocal expression of CD70 and of its receptor, CD27, in human long term-activated T and natural killer (NK) cells: inverse regulation by cytokines and role in induction of cytotoxicity. Clin Exp Immunol 107:608–13
Peitsch MC, Tschopp J (1995) Comparative molecular modelling of the Fas-ligand and other members of the TNF family. Mol Immunol 32:761–72
Grewal IS (2008) CD70 as a therapeutic target in human malignancies. Expert Opin Ther Targets 12:341–51
Hintzen RQ, de Jong R, Lens SM et al (1994) CD27: marker and mediator of T-cell activation? Immunol Today 15:307–11
Lens SM, Tesselaar K, van Oers MH et al (1998) Control of lymphocyte function through CD27-CD70 interactions. Semin Immunol 10:491–9
Borst J, Hendriks J, Xiao Y (2005) CD27 and CD70 in T cell and B cell activation. Curr Opin Immunol 17:275–81
Yang ZZ, Novak AJ, Ziesmer SC et al (2007) CD70+ non-Hodgkin lymphoma B cells induce Foxp3 expression and regulatory function in intratumoral CD4 + CD25 T cells. Blood 110:2537–44
Diegmann J, Junker K, Gerstmayer B, Bosio A, Hindermann W, Rosenhahn J et al (2005) Identification of CD70 as a diagnostic biomarker for clear cell renal cell carcinoma by gene expression profiling, real-time RT-PCR and immunohistochemistry. Eur J Cancer 41:1794–801
Law CL, Gordon KA, Toki BE et al (2006) Lymphocyte activation antigen CD70 expressed by renal cell carcinoma is a potential therapeutic target for anti-CD70 antibody-drug conjugates. Can cer Res 66:2328–37
Lens SM, Drillenburg P, den Drijver BF et al (1999) Aberrant expression and reverse signalling of CD70 on malignant B cells. Br J Haematol 106:491–503
Claus C, Riether C, Schürch C et al (2012) CD27 signaling increases the frequency of regulatory T cells and promotes tumor growth. Cancer Res 72:3664–76
Oflazoglu E, Stone IJ, Gordon K et al (2008) Potent anticarcinoma activity of the humanized anti-CD70 antibody h1F6 conjugated to the tubulin inhibitor auristatin via an uncleavable linker. Clin Cancer Res 14:6171–80
McEarchern JA, Oflazoglu E, Francisco L et al (2007) Engineered anti-CD70 antibody with multiple effector functions exhibits in vitro and in vivo antitumor activities. Blood 109:1185–92
McEarchern JA, Smith LM, McDonagh CF et al (2008) Preclinical characterization of SGN-70, a humanized antibody directed against CD70. Clin Cancer Res 14:7763–72
Younes A, Bartlett NL, Leonard JP et al (2010) Brentuximab vedotin (SGN-35) for relapsed CD30-positive lymphomas. N Engl J Med 363:1812–21
Ryan MC, Kostner H, Gordon KA et al (2010) Targeting pancreatic and ovarian carcinomas using the auristatin-based anti-CD70 antibody–drug conjugate SGN-75. Br J Cancer 103:676–84
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–47
Cheson BD, Pfistner B, Juweid ME et al (2007) Revised response criteria for malignant lymphoma. J Clin Oncol 25:579–86
Hauswirth AW, Skrabs C, Schützinger C et al (2008) Autoimmune thrombocytopenia in non-Hodgkin’s lymphomas. Haematologica 93:447–50
Younes A, Kim S, Romaguera J et al (2012) Phase I multidose-escalation study of the anti-CD19 maytansinoid immunoconjugate SAR3419 administered by intravenous infusion every 3 weeks to patients with relapsed/refractory B-cell lymphoma. J Clin Oncol 30:2776–82
Pro B, Advani R, Brice P et al (2012) Brentuximab vedotin (SGN-35) in patients with relapsed or refractory systemic anaplastic large cell lymphoma: results of a phase II study. J Clin Oncol 30:2190–6
Younes A, Gopal AK, Smith SE et al (2012) Results of a pivotal phase II study of brentuximab vedotin for patients with relapsed or refractory Hodgkin’s lymphoma. J Clin Oncol 30:2183–9
Alley SC, Zhang X, Okeley NM et al (2009) The pharmacologic basis for antibody-auristatin conjugate activity. J Pharmacol Exp Ther 330:932–8
Coquet JM, Middendorp S, van der Horst G et al (2013) The CD27 and CD70 costimulatory pathway inhibits effector function of T helper 17 cells and attenuates associated autoimmunity. Immunity 38:53–65
Ho AW, Hatjiharissi E, Ciccarelli BT et al (2008) CD27-CD70 interactions in the pathogenesis of Waldenstrom macroglobulinemia. Blood 112:4683–9
Jeffrey SC, Burke PJ, Lyon RP et al (2013) A potent anti-CD70 antibody-drug conjugate combining a dimeric pyrrolobenzodiazepine drug with site-specific conjugation technology. Bioconjug Chem 24:1256–63
Acknowledgments
The authors thank Elisabeth Heath, MD, FACP at the Karmanos Cancer Institute for her contributions to the study, Edmund Ng for statistical guidance and Roberta Connelly for medical writing assistance (both sponsored by Seattle Genetics).
Ethical standards
This study complied with the current laws of the country (United States) in which it was conducted.
Financial Support
Funding for this study was provided by Seattle Genetics, Inc.
Potential Conflicts of Interest
The institutions of NM Tannir, A Forero-Torres, R Ramchandren, SK Pal, SM Ansell, JR Infante, S de Vos, PA Hamlin, SK Kim, and JA Thompson received research funding from Seattle Genetics, Inc. S de Vos, PA Hamlin, and SK Kim serve as consultants to Seattle Genetics, and A Forero-Torres and R Ramchandren have received honoraria from the Seattle Genetics Speakers’ Bureau. NC Whiting, EM Gartner, and B Zhao are employees of and hold equity interest in Seattle Genetics.
Author information
Authors and Affiliations
Corresponding author
Additional information
Note
Data were presented in part as a poster at the American Society of Clinical Oncology, Genitourinary Cancers Symposium, February 2013, Orlando, FL.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 233 kb)
Rights and permissions
About this article
Cite this article
Tannir, N.M., Forero-Torres, A., Ramchandren, R. et al. Phase I dose-escalation study of SGN-75 in patients with CD70-positive relapsed/refractory non-Hodgkin lymphoma or metastatic renal cell carcinoma. Invest New Drugs 32, 1246–1257 (2014). https://doi.org/10.1007/s10637-014-0151-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-014-0151-0