Summary
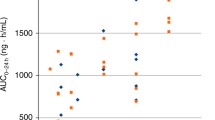
Approved doses of a number of drugs in Japan are known to be different from those in the United States (US) and the European Union (EU), and doses are often set lower for the Japanese than for the western people. Similarly, some oncology drugs also have lower dose approved for the Japanese than for the western people. A total of 40 oncology drugs were approved as new molecular entities in Japan between 2001 and 2013. Of the 40 drugs, 21 were molecularly targeted drugs and 13 were cytotoxic drugs. Five (12.5 %) of the 40 drugs had different approved dose from that in the US and the EU. Of the 13 cytotoxic drugs, four drugs (30.8 %) differed in approved dose, while all the molecularly targeted drugs (21 of 21 drugs) had the same approved dose. We compared the maximum tolerated dose (MTD) of the 21 molecularly targeted drugs in the Japanese with that in the western people and found that the MTD was determined lower in the Japanese than that in the western people (two drugs), was not different (10 drugs), and MTD was not determined in the Japanese and incommensurable because of the different dose range tested in Japan (nine drugs). All the molecularly targeted drugs are the same in approved doses and few molecularly targeted drugs differ in MTD between Japan and the Western countries.


Similar content being viewed by others
References
Malinowski HJ, Westelinck A, Sato J, Ong T (2008) Same drug, different dosing: differences in dosing for drugs approved in the United States, Europe, and Japan. J Clin Pharmacol 48:900–908
Fukunaga S, Kusama M, Arnold FL, Ono S (2011) Ethnic differences in pharmacokinetics in new drug applications and approved doses in Japan. J Clin Pharmacol 51:1237–1240
Arnold FL, Kusama M, Ono S (2010) Exploring differences in drug doses between Japan and Western countries. Clin Pharmacol Ther 87(6):714–720
Nakashima K, Narukawa M, Takeuchi M (2011) Approaches to Japanese dose evaluation in global drug development: factors that generate different dosage between Japan and the United States. Clin Pharmacol Ther 90(6):836–843
Hyodo I, Shirao K, Doi T, Hatake K, Arai Y, Yamaguchi K et al (2006) A phase II study of the global dose and schedule of capecitabine in Japanese patients with metastatic colorectal cancer. Jpn J Clin Oncol 36(7):410–417
Sai K, Sawada J, Minami H (2008) Irinotecan pharmacogenetics in Japanese cancer patients: role of UGT1A1 6 and 28. Yakugaku Zasshi 128(4):575–584
Kudoh S, Takeda K, Nakagawa K, Takada M et al (2006) Phase III study of docetaxel compared with vinorelbine in elderly patients with advanced non-small-cell lung cancer: results of the west Japan thoracic oncology group trial (WJTOG 9904). J Clin Oncol 24:3657–3663
Murakami H, Shimizu K, Sawamura M, Suzuki K et al (2009) Phase II study and pharmacokinetic study of thalidomide in Japanese patients with relapsed/refractory multiple myeloma. Int J Hematol 89(5):636–641
Singhal S, Mehta J, Desikan R, Ayers D et al (1999) Antitumor activity of thalidomide in refractory multiple myeloma. N Engl J Med 341:1565–1571
Usami M, Akaza H, Arai Y, Hirano Y, Kagawa S et al (2007) Bicalutamide 80mg combined with a luteinizing hormone relieasing hormone agonist (LHRH-A) versus LHRH-A monotherapy in advanced prostate cancer findings from a phase III randomized, double-blind, multicenter trial in Japanese patients. Prostate Cancer Prostatic Dis 10(2):194–201
Akaza H, Yamaguchi A, Matsuda T, Igawa M et al (2004) Superior anti-tumor efficacy of bicalutamide 80mg in combination with luteinizing hormone (LHRH) agonist versus LHRH agonist monotherapy as first-line treatment for advanced prostate cancer: interim results of a randomized study in Japanese patients. Jpn J Clin Oncol 34(1):20–28
McLeod DG, Iversen P, See WA, Morris T et al (2006) Bicalutamide 150 mg plus standard care alone for early prostate cancer. BJU Int 97(2):247–254
Ichimaru K, Toyoshima S, Uyama T (2010) Effective global development strategy for obtaining regulatory approval in the context of ethnicity-related drug response factors. Clin Pharmacol Ther 87(3):362–366
Merenda C (2012) Racial/ethnic composition of study participants in FDA-approved oncology new molecular entities, 2006–2008. J Natl Med Assoc 104(9–10):430–435
Salzberg M (2012) First-in-human phase 1 studies in oncology: the new challenge for investigative sites. Rambam Maimoinides Med J 3(2):e0007
Sinha G (2010) Japan works to shorten “Drug Lag”, boost trials of new drugs. J Natl Cancer Inst 102(3):148–151
Evaluation and Licensing Division, Pharmaceutical and Food Safety Bureau, Ministry of Health, Labour and Welfare., Basic Principles of Global Clinical Trials. Notification no. 0928010, 2007; http://www.pmda.go.jp/operations/notice/2007/file/0928010-e.pdf. Accessed Feb 2014
Asano K, Tanaka A, Sato T, Uyama T (2013) Regulatory challenges in the review of data from global clinical trials: the PMDA prerspective. Clin Pharmacol Ther 94(2):195–198
Molzon JA, Giaquinto A, Lindstrom L, Tominaga T et al (2011) The value and benefit of the international conference on harmonization to drug regulatory authorities: advancing harmonization for better public health. Clin Pharmacol Ther 89(4):503–512
Glickman SW, McHutchison JG, Peterson ED, Cairns CB et al (2009) Ethical and scientific implications of the globalization of clinical research. NEJM 360(8):816–823
Pharmaceuticals and Medical Devices Agency Review report of docetaxel 75 mg/m2 (in Japanese), http://www.info.pmda.go.jp/shinyaku/P201000172/780069000_22100AMX01369_A100_1.pdf. Accessed Feb 2014
Forster MD, Saijo N, Seymour L, Calvert H (2010) Performing phase I clinical trials of anticancer agents: perspectives from within the European Union and Japan. Clin Cancer Res 16(6):1737–1744
Acknowledgments
Authors thank to Kentaro Takeda for his statistical advice and Minako Takizawa for her assistance in writing the manuscript.
Funding sources
No specific funding was disclosed.
Conflict of interest disclosures
Hideki Maeda is an employee of Astellas Pharma Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maeda, H., Kurokawa, T. Differences in maximum tolerated doses and approval doses of molecularly targeted oncology drug between Japan and Western countries. Invest New Drugs 32, 661–669 (2014). https://doi.org/10.1007/s10637-014-0080-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-014-0080-y