Summary
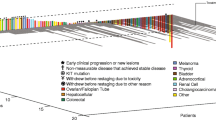
Background IMC-18F1 (icrucumab), a human monoclonal antibody against vascular endothelial growth factor receptor-1 (VEGFR-1), potently inhibits ligand-dependent phosphorylation of VEGFR-1 and downstream signaling, making icrucumab an attractive candidate for antitumor activity. Objectives The primary objective was to determine the safety profile and maximum tolerated dose of icrucumab in patients with advanced solid tumors that were previously unresponsive to standard therapy or for which no standard therapy was available. Methods In this open-label, dose-escalation, Phase 1 study, patients received icrucumab intravenously weekly at 2, 3, 6, and 12 mg/kg (Cohorts 1–4), every other week (q2w) at 15 mg/kg (Cohort 5), or every third week at 20 mg/kg (Cohort 6). Patients received icrucumab until evidence of progressive disease or other withdrawal criteria were met. Results Twenty-six patients received icrucumab. The most common adverse events were fatigue, nausea, peripheral edema, anemia, dyspnea, and vomiting. No dose-limiting toxicities (DLTs) were observed in Cohorts 1–5. Two DLTs were observed in Cohort 6 (anemia and hyponatremia), and enrollment was stopped. No patient demonstrated an immunogenic response. Overall, icrucumab exhibited nonlinear pharmacokinetics at doses >6 mg/kg. Six patients (23.1 %) achieved stable disease with median duration of 11.1 weeks (range = 10.3–18.7 weeks); tumor types were thyroid, melanoma, colorectal (3 patients), and small-cell lung cancers. Conclusions Icrucumab was safely administered weekly at doses of 2–12 mg/kg and q2w at a dose of 15 mg/kg with no DLTs. Based on achievement of stable disease, icrucumab has potential for antitumor activity against advanced solid tumors.
Similar content being viewed by others
References
Sawano A, Iwai S, Sakurai Y, Ito M, Shitara K, Nakahata T, Shibuya M (2001) Flt-1, vascular endothelial growth factor receptor 1, is a novel cell surface marker for the lineage of monocyte-macrophages in humans. Blood 97(3):785–791
Sato K, Sasaki R, Ogura Y, Shimoda N, Togashi H, Terada K, Sugiyama T, Kakinuma H, Ogawa O, Kato T (1998) Expression of vascular endothelial growth factor gene and its receptor (flt-1) gene in urinary bladder cancer. Tohoku J Exp Med 185(3):173–184
Decaussin M, Sartelet H, Robert C, Moro D, Claraz C, Brambilla C, Brambilla E (1999) Expression of vascular endothelial growth factor (VEGF) and its two receptors (VEGF-R1-Flt1 and VEGF-R2-Flk1/KDR) in non-small cell lung carcinomas (NSCLCs): correlation with angiogenesis and survival. J Pathol 188(4):369–377
Wang ES, Teruya-Feldstein J, Wu Y, Zhu Z, Hicklin DJ, Moore MA (2004) Targeting autocrine and paracrine VEGF receptor pathways inhibits human lymphoma xenografts in vivo. Blood 104:2893–2902
Fischer C, Mazzone M, Jonckx B, Carmeliet P (2008) FLT1 and its ligands VEGFB and PlGF: drug targets for anti-angiogenic therapy? Nat Rev Cancer 8:942–956
Schwartz JD, Rowinsky EK, Youssoufian H, Pytowski B, Wu Y (2010) Vascular endothelial growth factor receptor-1 in human cancer: concise review and rationale for development of IMC-18F1 (Human antibody targeting vascular endothelial growth factor receptor-1). Cancer 116(4 suppl):1027–1032. doi:10.1002/cncr.24789
Vincent L, Jin DK, Karajannis MA, Shido K, Hooper AT, Rashbaum WK, Pytowski B, Wu Y, Hicklin DJ, Zhu Z, Bohlen P, Niesvizky R, Rafii S (2005) Fetal stromal-dependent paracrine and intracrine vascular endothelial growth factor-a/vascular endothelial growth factor receptor-1 signaling promotes proliferation and motility of human primary myeloma cells. Cancer Res 65(8):3185–3192
Kaplan RN, Rafii S, Lyden D (2006) Preparing the “soil”: the premetastatic niche. Cancer Res 66:11089–11093
Lyden D, Hattori K, Dias S, Costa C, Blaikie P, Butros L, Chadburn A, Heissig B, Marks W, Witte L, Wu Y, Hicklin D, Zhu Z, Hackett NR, Crystal RG, Moore MA, Hajjar KA, Manova K, Benezra R, Rafii S (2001) Impaired recruitment of bone-marrow-derived endothelial and hematopoietic precursor cells blocks tumor angiogenesis and growth. Nat Med 7(11):1194–1201
Pollard JW (2004) Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer 4:71–78
Hiratsuka S, Nakamura K, Iwai S, Murakami M, Itoh T, Kijima H, Shipley JM, Senior RM, Shibuya M (2002) MMP9 induction by vascular endothelial growth factor receptor-1 is involved in lung-specific metastasis. Cancer Cell 2(4):289–300
Wu Y, Zhong Z, Huber J, Bassi R, Finnerty B, Corcoran E, Li H, Navarro E, Balderes P, Jimenez X, Koo H, Mangalampalli VR, Ludwig DL, Tonra JR, Hicklin DJ (2006) Anti-vascular endothelial growth factor receptor-1 antagonist antibody as a therapeutic agent for cancer. Clin Cancer Res 12(21):6573–6584
Waldner MJ, Neurath MF (2012) Targeting the VEGF signaling pathway in cancer therapy. Expert Opin Ther Targets 16(1):5–13. doi:10.1517/14728222.2011.641951
Brekken RA, Overholser JP, Stastny VA, Waltenberger J, Minna JD, Thorpe PE (2000) Selective inhibition of vascular endothelial growth factor (VEGF) receptor 2 (KDR/Flk-1) activity by a monoclonal anti-VEGF antibody blocks tumor growth in mice. Cancer Res 60(18):5117–5124
Shibuya M, Claesson-Welsh L (2006) Signal transduction by VEGF receptors in regulation of angiogenesis and lymphangiogenesis. Exp Cell Res 312(5):549–560
Cao Y (2009) Positive and negative modulation of angiogenesis by VEGFR1 ligands. Sci Signal 2(59):re1. doi:10.1126/scisignal.259re1
Hicklin DJ, Ellis LM (2005) Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol 23:1011–1027
Roskoski R Jr (2007) Vascular endothelial growth factor (VEGF) signaling in tumor progression. Crit Rev Oncol Hematol 62(3):179–213
Brekken RA, Huang X, King SW, Thorpe PE (1998) Vascular endothelial growth factor as a marker of tumor endothelium. Cancer Res 58(9):1952–1959
Grothey A, Galanis E (2009) Targeting angiogenesis: progress with anti-VEGF treatment with large molecules. Nat Rev Clin Oncol 6(9):507–518. doi:10.1038/nrclinonc.2009.110
Sunshine SB, Dallabrida SM, Durand E, Ismail NS, Bazinet L, Birsner AE, Sohn R, Ikeda S, Pu WT, Kulke MH, Javaherian K, Zurakowski D, Folkman JM, Rupnick M (2012) Endostatin lowers blood pressure via nitric oxide and prevents hypertension associated with VEGF inhibition. Proc Natl Acad Sci U S A 109(28):11306–11311. doi:10.1073/pnas.1203275109
Dienstmann R, Braña I, Rodon J, Tabernero J (2011) Toxicity as a biomarker of efficacy of molecular targeted therapies: focus on EGFR and VEGF inhibiting anticancer drugs. Oncologist 16(12):1729–1740. doi:10.1634/theoncologist.2011-0163
Robinson ES, Khankin EV, Karumanchi SA, Humphreys BD (2010) Hypertension induced by vascular endothelial growth factor signaling pathway inhibition: mechanisms and potential use as a biomarker. Semin Nephrol 30(6):591–601. doi:10.1016/j.semnephrol 2010.09.007
Ferroni P, Della-Morte D, Palmirotta R, Rundek T, Guadagni F, Roselli M (2012) Angiogenesis and hypertension: the dual role of anti-hypertensive and anti-angiogenic therapies. Curr Vasc Pharmacol 10(4):479–493
Thanigaimani S, Kichenadasse G, Mangoni AA (2011) The emerging role of vascular endothelial growth factor (VEGF) in vascular homeostasis: lessons from recent trials with anti-VEGF drugs. Curr Vasc Pharmacol 9(3):358–380
Acknowledgments
This study was sponsored by Eli Lilly and Company. The authors would like to thank Joseph Giaconia of INC Research (Raleigh, NC) for his writing assistance.
Conflicts of interest
Patricia LoRusso, Scot Remick, and Smitha Krishnamurthi declare that they have no conflict of interest. Floyd Fox and Aruna Dontabhaktuni are employees of ImClone Systems, a wholly-owned subsidiary of Eli Lilly and Company. Hagop Youssoufian, Nancy Hall, and Dmitri Grebennik were employees of ImClone Systems during the conduct of the trial and/or development of this manuscript.
Ethical standards
The protocol was approved before the start of the study by the relevant independent ethics committees in the participating institutions. The study was performed in accordance with the Declaration of Helsinki, good clinical practice guidelines, and applicable regulatory requirements. Written informed consent was obtained from all patients at initial visit.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
LoRusso, P.M., Krishnamurthi, S., Youssoufian, H. et al. Icrucumab, a fully human monoclonal antibody against the vascular endothelial growth factor receptor-1, in the treatment of patients with advanced solid malignancies: a Phase 1 study. Invest New Drugs 32, 303–311 (2014). https://doi.org/10.1007/s10637-013-9998-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-013-9998-8