Summary
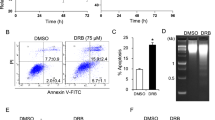
The fungal drug cordycepin (3-deoxyadenosine) is known to exert anti-tumor activities, preferentially by interfering with RNA synthesis. We have investigated the effect of cordycepin on human breast epithelial cell lines, ranging from non-malignant MCF10A cells to highly de-differentiated MDA-MB-435 cancer cells. Treatment of human breast cancer cells with cordycepin caused either apoptosis or persistent cell cycle arrest that was associated with reduced clonal growth of cordycepin-treated breast cancer cells. Highly de-differentiated breast cancer cell lines, such as MDA-MB-231 and MDA-MB-435, reacted more sensitive to cordycepin than less aggressive breast cancer cell lines (MCF7, T47D) or non-malignant breast epithelial cells (MCF10A), which poorly reacted to cordycepin. In cordycepin-sensitive breast cancer cells, a marked induction of the DNA damage response (DDR), including the phosphorylation of ATM, ATR, and histone γH2AX could be observed. These data indicate that cordycepin, which was believed to cause cancer cell death by inhibition of RNA synthesis, induces DNA double strand breaks in breast cancer cells. The genotoxic effect of cordycepin on breast cancer cells indicates a new mechanism of cordycepin-induced cancer cell death, and its activity against highly undifferentiated breast cancer cells provides a new perspective of how cordycepin may be used in the treatment of advanced breast cancer.







Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
Levi F, Bosetti C, Lucchini F, Negri E, La Vecchia C (2005) Monitoring the decrease in breast cancer mortality in Europe. Eur J Cancer Prev 14:497–502
Dawood S (2010) Triple-negative breast cancer: epidemiology and management options. Drugs 70:2247–58
Paterson RR (2008) Cordyceps: a traditional Chinese medicine and another fungal therapeutic biofactory? Phytochemistry 69:1469–1495
Kodama EN, McCaffrey RP, Yusa K, Mitsuya H (2000) Antileukemic activity and mechanism of action of cordycepin against terminal deoxynucleotidyl transferase-positive (TdT+) leukemic cells. Biochem Pharmacol 59:273–281
Yoshikawa N, Nakamura K, Yamaguchi Y, Kagota S, Shinozuka K, Kunitomo M (2004) Antitumour activity of cordycepin in mice. Clin Exp Pharmacol Physiol 31:S51–3
Nakamura K, Konoha K, Yoshikawa N, Yamaguchi Y, Kagota S, Shinozuka K, Kunitomo M (2005) Effect of cordycepin (3'-deoxyadenosine) on hematogenic lung metastatic model mice. In Vivo 19:137–141
Chen LS, Stellrecht CM, Gandhi V (2008) RNA-directed agent, cordycepin, induces cell death in multiple myeloma cells. Br J Haematol 140:682–391
Lee SJ, Moon GS, Jung KH, Kim WJ, Moon SK (2010) c-Jun N-terminal kinase 1 is required for cordycepin-mediated induction of G2/M cell-cycle arrest via p21WAF1 expression in human colon cancer cells. Food Chem Toxicol 48:2772–2783
Noh EM, Youn HJ, Jung SH, Han JH, Jeong YJ, Chung EY, Jung JY, Kim BS, Lee SH, Lee YR, Kim JS (2010) Cordycepin inhibits TPA-induced matrix metalloproteinase-9 expression by suppressing the MAPK/AP-1 pathway in MCF-7 human breast cancer cells. Int J Mol Med 25:255–260
Jeong JW, Jin CY, Park C, Hong SH, Kim GY, Jeong YK, Lee JD, Yoo YH, Choi YH (2011) Induction of apoptosis by cordycepin via reactive oxygen species generation in human leukemia cells. Toxicol In Vitro 25:817–824
Thomadaki H, Tsiapalis CM, Scorilas A (2005) Polyadenylate polymerase modulations in human epithelioid cervix and breast cancer cell lines, treated with etoposide or cordycepin, follow cell cycle rather than apoptosis induction. Biol Chem 386(5):471–80
Kim H, Naura AS, Errami Y, Ju J, Boulares AH (2011) Cordycepin blocks lung injury-associated inflammation and promotes BRCA1-deficient breast cancer cell killing by effectively inhibiting PARP. Mol Med. doi:10.2119/molmed.2011.00032
Jackson SP, Bartek J (2009) The DNA-damage response in human biology and disease. Nature 461:1071–1078
Miwa M, Masutani M (2007) PolyADP-ribosylation and cancer. Cancer Sci 98:1528–1535
Weil MK, Chen AP (2011) PARP inhibitor treatment in ovarian and breast cancer. Curr Probl Cancer 35:7–50
Plesca D, Mazumder S, Almasan A (2008) DNA damage response and apoptosis. Methods Enzymol 446:107–122
North TW, Cohen SS (1978) Erythro-9-(2-hydroxy-3-nonyl)adenine as a specific inhibitor of herpes simplex virus replication in the presence and absence of adenosine analogues. Proc Natl Acad Sci USA 75:4684–4688
Gonzalez-Angulo AM, Morales-Vasquez F, Hortobagyi GN (2007) Overview of resistance to systemic therapy in patients with breast cancer. Adv Exp Med Biol 608:1–22
Amar S, Roy V, Perez EA (2009) Treatment of metastatic breast cancer: looking towards the future. Breast Cancer Res Treat 114:413–422
Swain SM (2011) Chemotherapy: updates and new perspectives. Oncologist 16(Suppl 1):30–39
Pal SK, Childs BH, Pegram M (2011) Triple negative breast cancer: unmet medical needs. Breast Cancer Res Treat 125:627–636
Cimprich KA, Cortez D (2008) ATR: an essential regulator of genome integrity. Nat Rev Mol Cell Biol 9:616–627
Riches LC, Lynch AM, Gooderham NJ (2008) Early events in the mammalian response to DNA double-strand breaks. Mutagenesis 23:331–339
López-Contreras AJ, Fernandez-Capetillo O (2010) The ATR barrier to replication-born DNA damage. DNA Repair (Amst) 9:1249–1255
Roos WP, Kaina B (2006) DNA damage-induced cell death by apoptosis. Trends Mol Med 12:440–450
Carden CP, Yap TA, Kaye SB (2010) PARP inhibition: targeting the Achilles’ heel of DNA repair to treat germline and sporadic ovarian cancers. Current Opinion in Oncology 22:473–480
Turner N, Tutt A, Ashworth A (2011) Hallmarks of ‘BRCAness’ in sporadic cancers. Nat Rev Cancer 4:814–819
Rastelli F, Biancanelli S, Falzetta A, Martignetti A, Casi C, Bascioni R, Giustini L, Crispino S (2010) Triple-negative breast cancer: current state of the art. Tumori 96(6):875–88
Plunkett W, Huang P, Xu YZ, Heinemann V, Grunewald R, Gandhi V (1995) Gemcitabine: metabolism, mechanisms of action, and self-potentiation. Semin Oncol 22(suppl 11):3–10
Klenow H (1963) Formation of the mono-, di-, and triphosphate of cordycepin in Ehrlich ascites-tumor cells in vitro. Biochim Biophys Acta 76:347–353
Cory JG, Suhadolnik RJ, Resnick B, Rich MA (1965) Incorporation of cordycepin (3'-deoxyadenosine) into ribonucleic acid and deoxyribonucleic acid of human tumor cells. Biochim Biophys Acta 103:646–653
Wong YY, Moon A, Duffin R, Barthet-Barateig A, Meijer HA, Clemens MJ, de Moor CH (2010) Cordycepin inhibits protein synthesis and cell adhesion through effects on signal transduction. J Biol Chem 285:2610–2621
Choi S, Lim MH, Kim KM, Jeon BH, Song WO, Kim TW (2011) Cordycepin-induced apoptosis and autophagy in breast cancer cells are independent of the estrogen receptor. Toxicol Appl Pharmacol 257:165–173
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, H.J., Burger, P., Vogel, M. et al. The nucleoside antagonist cordycepin causes DNA double strand breaks in breast cancer cells. Invest New Drugs 30, 1917–1925 (2012). https://doi.org/10.1007/s10637-012-9859-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-012-9859-x