Summary
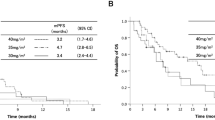
Purpose: We conducted a multicenter phase II study of amrubicin, a novel 9-aminoanthracycline, to evaluate its efficacy and safety in patients with non-small-cell lung cancer (NSCLC). Patients and methods: Entry requirements included cytologically or histologically proven measurable NSCLC, stage III or IV, no prior therapy, an Eastern Cooperative Oncology Group (ECOG) performance status of 0 to 2, and adequate organ function. Amrubicin was given by daily intravenous injection at 45 mg/m2/day for three consecutive days, repeated at 3 week intervals. Each patient received at least three treatment cycles. Results: Sixty-two patients were enrolled in this study. Of the 62 registered patients, 60 were eligible and assessable for efficacy, and 59 for toxicity. Overall response rate was 18.3% (95% confidence interval [CI], 9.5 to 30.4%) and median survival time was 8.2 months (95% CI, 6.7 to 10.4 months). Major toxicity was myelosuppression, with incidences of grade 3 or 4 toxicity of 78.0% for neutropenia, 54.2% for leukopenia, 30.5% for anemia, and 28.8% for thrombocytopenia. Non-hematological toxicities with a greater than 50% incidence were anorexia (69.5%), nausea/vomiting (55.9%), and alopecia (75.9%), but were relatively mild, with grade 3 toxicities observed in only one patient each (1.7%). Conclusion: Amrubicin was an active, well-tolerated agent in the treatment of NSCLC.


Similar content being viewed by others
References
Ishizumi K, Ohashi N, Tanno N (1987) Stereospecific total synthesis of 9-aminoanthracyclines: (+)-9-amino-9-deoxydaunomycin and related compounds. J Org Chem 52:4477–4485
Tani N, Yabuki M, Komuro S, Kanamaru H (2005) Characterization of the enzymes involved in the in vitro metabolism of amrubicin hydrochloride. Xenobiotica 35:1121–1133
Yamaoka T, Hanada M, Ichii S, Morisada S, Noguchi T, Yanagi Y (1998) Cytotoxicity of amrubicin, a novel 9-aminoanthracycline, and its active metabolite amrubicinol on human tumor cells. Jpn J Cancer Res 89:1067–1073
Morisada S, Yanagi Y, Noguchi T, Kashiwazaki Y, Fukui M (1989) Antitumor activities of a novel 9-aminoanthracycline (SM-5887) against mouse experimental tumors and human tumor xenografts. Jpn J Cancer Res 80:69–76
Noguchi T, Ichii S, Morisada S, Yamaoka T, Yanagi Y (1998) In vivo efficacy and tumor-selective metabolism of amrubicin to its active metabolite. Jpn J Cancer Res 89:1055–1060
Morisada S, Yanagi Y, Kashiwazaki Y, Fukui M (1989) Toxicological aspects of a novel 9-aminoanthracyline, SM-5887. Jpn J Cancer Res 80:77–82
Suzuki T, Minamide S, Iwasaki T, Yamamoto H, Kanda H (1997) Cardiotoxicity of a new anthracycline derivative (SM-5887) following intravenous administration to rabbits: Comparative study with doxorubicin. Invest New Drugs 15:219–225
Noda T, Watanabe T, Kohda A, Hosokawa S, Suzuki T (1998) Chronic effects of a novel synthetic anthracycline derivative (SM-5887) on normal heart and doxorubicin-induced cardiomyopathy in dogs. Invest New Drugs 16:121–128
Hanada M, Mizuno S, Fukushima A, Saito Y, Noguchi T, Yamaoka T (1998) A new antitumor agent amrubicin induces cell growth inhibition by stabilizing topoisomerase II-DNA complex. Jpn J Cancer Res 89:1229–1238
Hiraki S, Shinkai T, Furuse K, Fukuoka M, Ohnoshi T, Kimura I (1993) A phase II study of SM-5887, a novel 9-aminoanthracycline, for non-small-cell lung cancer. Proceedings of the 18th international congress of chemotherapy 866–867
Sugiura T, Ariyoshi Y, Negoro S, Nakamura N, Ikegami H, Takada M, Yana T, Fukuoka M (2005) Phase I/II study of amrubicin, a novel 9-aminoanthracycline, in patients with advanced non-small-cell lung cancer. Invest New Drugs 23:331–337
Noguchi T, Ichii S, Morisada S, Yamaoka T, Yanagi Y (1999) Evaluation of amrubicin with a 5 day administration schedule in a mouse model. Jpn J Cancer Chemother 26:1305–1312
Cortes EP, Takita H, Holland JF (1974) Adriamycin in advanced bronchogenic carcinoma. Cancer 34:518–525
Sawa T, Yana T, Takada M, Sugiura T, Kudoh S, Kamei T, Isobe T, Yamamoto H, Yokota S, Katakami N, Tohda Y, Kawakami A, Nakanishi Y, Ariyoshi Y (2006) Multicenter phase II study of amrubicin, 9-amino-anthracycline, in patients with advanced non-small-cell lung cancer (Study 1): West Japan Thoracic Oncology Group (WJTOG) trial. Invest New Drugs 24:151–158
Japan Society for Cancer Therapy (1993) Criteria for the evaluation of the clinical effects of solid cancer chemotherapy. J Jpn Soc Cancer Ther 28:101–130
World Health Organization (1979) Handbook for Reporting Results of Cancer Treatment (WHO Offst Publication No. 48). World Health Organization, Geneva, Switzerland
Kaplan WH, Meier P (1952) Nonparametric estimation from incomplete observations. J Am Stat Assoc 53:583–612
Grilli R, Oxman AD, Julian JA (1993) Chemotherapy for advanced non-small cell lung cancer: how much benefit is enough? J Clin Oncol 11:1866–1872
Souquet PJ, Chauvin F, Boissel JP, Cellerino R, Cormier Y, Ganz PA, Kaasa S, Pater JL, Quoix E, Rapp E, Tumarello D, Williams J, Woods BL, Bernard JP (1993) Polychemotherapy in advanced non-small cell lung cancer: a meta-analysis. Lancet 342:19–21
Non-small Cell Lung Cancer Collaborative Group (1995) Chemotherapy in non-small cell lung cancer: a meta-analysis using updated data on individual patients from 52 randomised clinical trials. BMJ 311:899–909
Ginsberg RJ, Vokes EE, Raben A (2001) Non-small cell lung cancer. In: DeVita VTJ, Hellman S, Rosenberg SA (eds) Cancer: principles and practice of oncology, 6th edn. Lippincott Williams and Wilkins, Philadelphia, pp 925–983
Bunn PA, Kelly K (1998) New chemotherapeutic agents prolong survival and improve quality of life in non-small cell lung cancer: a review of the literature and future directions. Clin Cancer Res 5:1087–1100
Ohe Y, Negoro S, Matsui K, Nakagawa K, Sugiura T, Takada Y, Nishiwaki Y, Yokota S, Kawahara M, Saijo N, Fukuoka M, Aritoshi Y (2005) Phase I–II study of amrubicin and cisplatin in previously untreated patients with extensive-stage small-cell lung cancer. Ann Oncol 16:430–436
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takeda, K., Takifuji, N., Negoro, S. et al. Phase II study of amrubicin, 9-amino-anthracycline, in patients with advanced non-small-cell lung cancer: a West Japan Thoracic Oncology Group (WJTOG) study. Invest New Drugs 25, 377–383 (2007). https://doi.org/10.1007/s10637-007-9039-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-007-9039-6