Summary
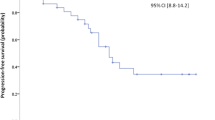
Objective: To assess the efficacy and toxicity of the combination of interferon-alpha and doxycycline in patients with metastatic renal cell carcinoma and to assess the effect of this treatment on serum vascular endothelial growth factor (VEGF) levels. Patients and Methods: Seventeen patients with Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1 and life expectancy greater than 4 months with radiologically evident advanced renal cell carcinoma were enrolled. Eight patients had prior nephrectomy and 10 patients were treated within 4 months of their diagnosis. Treatment consisted of interferon-alpha up to 9 million units subcutaneously three times per week and doxycycline 300 mg orally twice per day for weeks one and three of each four-week cycle. Toxicity was evaluated on a biweekly basis and response on a bimonthly basis. VEGF plasma levels were assessed monthly as a measure of potential antiangiogenic effect. Results: No objective responses were seen. The mean duration of study was 2.6 cycles (range: 0.8–6.0 cycles). Three patients (17%) tolerated therapy and displayed stable disease for greater than four months. Five patients withdrew from study before the first response evaluation. Ten patients experienced grade 2 gastrointestinal toxicity requiring dose reduction of doxycycline. Eight patients experienced grade 2 fatigue requiring dose reduction of interferon. VEGF plasma levels were initially suppressed in patients who demonstrated progressive disease but not in patients with stable disease. Conclusion: This regimen of doxycycline and interferon-alpha was not efficacious as treatment for renal cell carcinoma. Plasma VEGF levels were significantly decreased during the first two cycles of treatment, but this does not correlate with clinical outcome.
Similar content being viewed by others
References
Ahmedin J, Thomas A, Taylor M, Thun M: Cancer Statistics 2002. CA Cancer J Clin 52: 23–47, 2002
Motzer RJ, Bander NH, Nanus DM: Renal-Cell Carcinoma. N Engl J Med 335: 865–75, 1996
Eckhardt SG: Angiogenesis inhibitors as cancer therapy. Hospital Practice Jan 15, 1999
Slaton JW, Inoue K, Perotte P, El-Naggar AK, Swanson DA, Fidler IJ, Dinney CPN: Expression levels of genes that regulate metastases and angiogenesis correlate with advanced pathological stage of renal cell carcinoma. Am J Pathol 158: 735–743, 2001
Sasamura H, Takahashe A, Miyao N, Masumori N, Kitamura H, Itoh N, Tsukamoto T: Inhibitory expression of angiogenic factors by antiangiogenic agents in renal cell carcinoma. BJU Int 86: 768–773, 2002
Lokeshwar BL: MMP inhibition in prostate cancer. Ann NY Acad Sci 878: 271–289, 1999
Kelly K: New chemotherapy. Chest 117(4): 156S–162S, 2000
Gu Y, Lee HM, Roemer EJ, Musacchia L, Golub LM, Simon SR: Inhibition of tumor cell invasiveness by chemically modified tetracyclines. Curr Med Chem 8(3): 261–270, 2001
Fife RS, Sledge GW, Roth BJ, Proctor C: Effects of doxycycline on human prostate cancer cells in vitro. Cancer Letters 127: 37–41, 1998
Fife RS, Sledge GW: Effects of doxycycline on in vitro growth, migration, and gelatinase activity of breast carcinoma cells. J Lab Clin Med 125: 407–411, 1995
Lukkonen A, Sorsa T, Salo T, Tervahartiala T, Koivunen E, Golub L, Simon S, Stenman UH: Down-regulation of trypsinogen-2 expression by chemicallymodified tetracyclines: association with reduced cancer cell migration. Int J Cancer 86(4): 577–581, 2000
Stebbing J, Gore M: The current status of interferon-(alpha) treatment in advanced renal cancer. BJU int 87(7): 599–601, 2001
Negrier S, Escudier B, Lasset C, Douillard JY, Savary J, Chevreau C, Ravaud A, Mercatello A, Peny J, Mousseau M, Philip T. Tursz T: Recombinant human interleukin-2, recombinant human interferon alfa-2a, or both in metastatic renal-cell carcinoma. N Engl J of Med 338(18): 1272–1278, 1998
Stadler WM, Kuzel T, Shapiro C Sosman J, Clark J, Vogelzang NJ: Multi-institutional study of the angiogenesis inhibitor TNP-470 in metastatic renal cell carcinoma. J Clin Oncol 17(8): 2541–2545, 1999
Motzer RJ, Berg W, Ginsberg M, Russo P, Vuky J, Yu R, Bacik J, Mozumdar M: Phase II trial of thalidomide for patients with advanced renal cell carcinoma. J Clin Oncol 20(1): 302–306, 2002
Batist G, Patenaude F, Champagne P, Croteau D, Levinton C, Hariton C, Escudier B, Dupont E: Neovastat (AE-941) in refractory renal cell carcinoma patients: report of a phase II trial with two dose levels. Ann Oncol. 13(8): 1259–1263, 2002
Kuenen BC, Tabernero J, Baselga J, Cavalli F, Pfanner E, Conte PF, Seeber S, Madhusudan S, Deplanque G, Huisman H, Scigalla P, Hoekman K, Harris AL: Efficacy and toxicity of the angiogenesis inhibitor SU5416 as a single agent in patients with advanced renal cell carcinoma, melanoma, and soft tissue sarcoma. Clin Cancer Res 9(5): 1648–1655, 2003
Lara PN Jr, Quinn DI, Margolin K, Meyers FJ, Longmate J, Frankel P, Mack PC, Turrell C, Valk P, Rao J, Buckley P, Wun T, Gosselin R, Galvin I, Gumerlock PH, Lenz HJ, Doroshow JH, Gandara DR: SU5416 plus interferon alpha in advanced renal cell carcinoma: A phase II California Cancer Consortium Study with biological and imaging correlates of angiogenesis inhibition. Clin Cancer Res 9(13): 4772–4781, 2003
Hernberg M, Virkkunen P, Bono P, Ahtinen H, Maenpaa H, Joensuu H: Interferon alfa-2b three times daily and thalidomide in the treatment of metastatic renal cell carcinoma. J Clin Oncol 21(20): 3770–3776, 2003
Kuenen BC, Levi M, Meijers JC, Kakkar AK, van Hinsbergh VW. Kostense PJ, Pinedo HM, Hoekman K: Analysis of coagulation cascade and endothelial cell activation during inhibition of vascular endothelial growth factor/vascular endothelial growth factor receptor pathway in cancer patients. Arteriosclerosis. Thrombosis & Vascular Biology 22(9): 1500–1505, 2002
Motzer RJ, Rini BI, Michaelson M, Redman BG, Hudes GR, Wilding G, Bukowski DJ, George ST, Kim C, Baum M: Phase 2 trials of SU11248 show antitumor activity in second-line therapy for patients with metastatic renal cell carcinoma (RCC). ASCO Proc. Abstract 4508, 2005
Rini B, Rixi R, Bukowski MD, Michaelson G, Wilding G, Hudes O, Bolte H, Steinfeldt S, Reich D, Motzer R: AG-013736, a multi-target tyrosine kinase receptor inhibitor, demonstrates anti-tumor activity in a Phase 2 study of cytokine-refractory, metastatic renal cell cancer (RCC). ASCO Proc. Abstract 4509, 2005
Escudier B, Szczylik C, Eisen T, Stadler WM, Schwartz B, Shan M, Bukowski RM: Randomized Phase III trial of the Raf kinase and VEGFR inhibitor sorafenib (BAY 43-9006) in patients with advanced renal cell carcinoma (RCC). ASCO Proc. Abstract LB4510, 2005
Gordon MS, Battiato LA, Jones D, Roth BJ, Harrison-Mann B, Fife R, Collins M, Sledge GW: A phase I trial of doxycycline in patients with cancer. ASCO Proc Abstract 794, 1997
Liang KY, Zeger SL: Longitudinal data analysis using generalized linear models. Biometrika: 13–22, 1986
Motzer RJ, Mazumdar M, Bacik J, Berg W, Amsterdam A, Ferrara J: Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol 17(8): 2530–2540, 1999
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Huie, M., Oettel, K., Van Ummersen, L. et al. Phase II study of interferon-alpha and doxycycline for advanced renal cell carcinoma. Invest New Drugs 24, 255–260 (2006). https://doi.org/10.1007/s10637-005-3903-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-005-3903-z