Abstract
Background and Aims
Hepatitis B virus (HBV) reactivation has been reported in patients co-infected with hepatitis C virus (HCV) during direct acting antiviral (DAA) therapy, leading the United States Food and Drug Administration (U.S. FDA) to issue a black box warning on all DAA drug labels recommending monitoring for HBV reactivation. We conducted a comprehensive evaluation to assess the rate of HBV reactivation among patients with chronic hepatitis C (CHC) during DAA therapy.
Methods
Patients with CHC and recovered HBV infection (hepatitis B surface antigen negative (HBsAg)/anti-hepatitis B core positive), treated with DAAs were included if stored sera were available. Samples were tested for HBV DNA, HBsAg, and ALT. HBV reactivation was considered if (1) HBV DNA was undetectable pre-DAA therapy and became detectable post-therapy, or (2) HBV DNA was detectable pre-treatment, but not quantifiable (< 20 IU/mL) and became quantifiable post-treatment.
Result
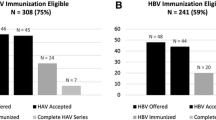
79 patients with median age of 62 years were included. 68% were male and Caucasian. Various DAA regimens were administered for 12–24 weeks. Reactivation occurred in 8/79 (10%) of patients and occurred more frequently in men compared to women: 6 during treatment and 2 after treatment. Neither an ALT flare nor HBsAg seroreversion were observed. Detectable HBV DNA was transient in 5/8 and could not be determined in 3/8 but ALT flares were not observed in follow-up of these patients.
Conclusion
The risk of HBV reactivation was low in CHC patients with resolved HBV during DAA therapy. Our data support testing for HBV DNA only in selected patients with ALT flares or failure of ALT normalization during DAA treatment.
Similar content being viewed by others

Abbreviations
- HBV:
-
Hepatitis B virus
- HCV:
-
Hepatitis C virus
- HIV:
-
Human immunodeficiency virus
- DAA:
-
Direct acting antiviral
- U.S. FDA:
-
United States Food and Drug Administration
- CHC:
-
Chronic hepatitis C
- CD20:
-
Cluster of differentiate 20
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- ALKP:
-
Alkaline phosphatase
- DNA:
-
Deoxyribonucleic acid
- RNA:
-
Ribonucleic acid
- ULN:
-
Upper limit of normal
- pegIFN:
-
Pegylated interferon
- HBsAg:
-
Hepatitis B surface antigen
- Anti-HBc:
-
Hepatitis B core antibody
- ETV:
-
Entecavir
- LDB:
-
Liver diseases branch
- NIDDK:
-
National Institute of Diabetes and Digestive and Kidney Diseases
- NIH:
-
National Institutes of Health
References
Dill
Loomba R, Liang TJ. Hepatitis B Reactivation Associated With Immune Suppressive and Biological Modifier Therapies: Current Concepts, Management Strategies, and Future Directions. Gastroenterology 2017;152:1297–1309.
https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-warns-about-risk-hepatitis-b-reactivating-some-patients-treated. Last accessed November 1st, FDA Drug Safety Communication: FDA warns about the risk of hepatitis B reactivating in some patients treated with direct -acting antivirals for hepatitis C. 2016. 2016.
Bersoff-Matcha SJ et al. Hepatitis B Virus Reactivation Associated With Direct-Acting Antiviral Therapy for Chronic Hepatitis C Virus. Ann Intern Med 2017;167:760.
Liu CJ et al. Efficacy of Ledipasvir and Sofosbuvir Treatment of HCV Infection in Patients Coinfected With HBV. Gastroenterology 2018;154:989–997.
Mucke MM et al. Hepatitis B virus reactivation during direct-acting antiviral therapy for hepatitis C: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol 2018;3:172–180.
Jiang XW et al. Hepatitis B reactivation in patients receiving direct-acting antiviral therapy or interferon-based therapy for hepatitis C: A systematic review and meta-analysis. World J Gastroenterol 2018;24:3181–3191.
El Kassas M et al. Risk of hepatitis B virus reactivation with direct-acting antivirals against hepatitis C virus: A cohort study from Egypt and meta-analysis of published data. Liver Int 2018;38:2159–2169.
Chen G et al. Hepatitis B reactivation in hepatitis B and C coinfected patients treated with antiviral agents: A systematic review and meta-analysis. Hepatology 2017;66:13–26.
Murai K et al. Hepatitis C virus infection suppresses hepatitis B virus replication via the RIG-I-like helicase pathway. Sci Rep 2020;10:941.
Cheng X et al. Diminished hepatic IFN response following HCV clearance triggers HBV reactivation in coinfection. J Clin Invest 2020;130:3205–3220.
Cheng PN et al. Entecavir Prevents HBV Reactivation During Direct Acting Antivirals for HCV/HBV Dual Infection: A Randomized Trial. Clin Gastroenterol Hepatol 2022;20:2800–2808.
Belperio PS et al. Evaluation of hepatitis B reactivation among 62,920 veterans treated with oral hepatitis C antivirals. Hepatology 2017;66:27–36.
Wang C et al. Hepatitis due to Reactivation of Hepatitis B Virus in Endemic Areas Among Patients With Hepatitis C Treated With Direct-acting Antiviral Agents. Clin Gastroenterol Hepatol 2017;15:132–136.
Mucke MM et al. Absence of HBV Reactivation in Patients With Resolved HBV Infection Following DAA Therapy for Hepatitis C: A 1-Year Follow-up Study. Open Forum Infect Dis 2019;6:ofy340.
Funding
The study was supported by the Intramural Research Program, National Institute of Diabetes and Digestive and Kidney Diseases.
Author information
Authors and Affiliations
Contributions
NAM: analysis and interpretation and data, statistical analysis, drafting of the manuscript, critical revision for intellectual content. ASA, KSL, JJF: study concept and design, acquisition of data, analysis and interpretation and data, statistical analysis, critical revision for intellectual content. VK: study concept and design, acquisition of data, analysis and interpretation and data, critical revision for intellectual content. HA, HLAJ: study concept and design, acquisition of data, statistical analysis, critical revision for intellectual content. MGG: study concept and design, acquisition of data, analysis and interpretation and data, statistical analysis, drafting of the manuscript, critical revision for intellectual content.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Guarantor
Marc G. Ghany accepts full responsibility for the conduct of the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Majeed, N.A., Alawad, A.S., Liem, K.S. et al. Low Rate of Hepatitis B Reactivation Among Patients with Chronic Hepatitis C During Direct Acting Antiviral Therapy. Dig Dis Sci 68, 3193–3198 (2023). https://doi.org/10.1007/s10620-023-07916-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-023-07916-2