Abstract
Background
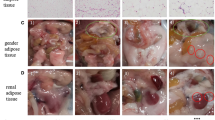
Visceral adipose tissue (VAT) is related to SAP prognosis. As a depot of VAT, mesenteric adipose tissue (MAT) resides between pancreas and gut, which might affect SAP and the secondary intestinal injury.
Aims
To investigate the changes of MAT in SAP.
Methods
24 SD rats were randomly divided into four groups. 18 rats in SAP group were euthanized in time gradients (6 h, 24 h, and 48 h after modeling) and the others in control group. Blood samples and tissues of pancreas, gut, and MAT were taken for analysis.
Results
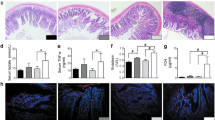
Compared to the control group, SAP rats appeared MAT inflammation, presenting higher mRNA expression of TNF-α and IL-6 and lower IL-10, and histological changes after 6 h of modeling, which became worse over time. Flow cytometry showed that B lymphocytes increased in MAT after 24 h of SAP modeling and lasted up to 48 h, earlier than the changes of T lymphocytes and macrophages. The intestinal barrier integrity was damaged after 6 h of modeling, presenting lower mRNA and protein expression of ZO-1 and occludin, higher serum levels of LPS and DAO, with pathological changes, which gradually aggravated after 24 h and 48 h. SAP rats had higher serum levels of inflammatory indicators and revealed histological inflammation of pancreas, the severity of which increased with the passage of modeling time.
Conclusion
MAT appeared inflammation in early-stage SAP, and became worse over time, with the same trend as the intestinal barrier injury and the severity of pancreatitis. B lymphocytes infiltrated early in MAT, which might promote the MAT inflammation.







Similar content being viewed by others
References
Frossard J, Steer M, Pastor C. Acute pancreatitis. Lancet. 2008;371:143–152.
Afghani E, Pandol S, Shimosegawa T et al. Acute Pancreatitis-Progress and Challenges: A Report on an International Symposium. Pancreas. 2015;44:1195–1210.
Fritz S, Hackert T, Hartwig W et al. Bacterial translocation and infected pancreatic necrosis in acute necrotizing pancreatitis derives from small bowel rather than from colon. Am J Surg. 2010;200:111–117.
Kuan L, Dennison A, Garcea G. Association of visceral adipose tissue on the incidence and severity of acute pancreatitis: a systematic review. Pancreatology. 2020;20:1056–1061.
Bukulmez A, Gokaslan C, Oflu A. Increase in visceral adipose tissue and subcutaneous adipose tissue thickness in children with acute pancreatitis. A case-control study. Arch Pediatr. 2021;28:29–32.
Mao R, Kurada S, Gordon I et al. The mesenteric fat and intestinal muscle interface: creeping fat influencing stricture formation in Crohn’s disease. Inflamm Bowel Dis. 2019;25:421–426.
Gea-Sorlí S, Laia B, Daniel C et al. Differences in the Inflammatory Response Induced by Acute Pancreatitis in Different White Adipose Tissue Sites in the Rat. Plos One 2012;7:e41933.
Li Y, Zhu W, Zuo L et al. The role of the mesentery in Crohn’s Disease: The contributions of nerves, vessels, lymphatics, and rat to the pathogenesis and disease course. Inflamm Bowel Dis. 2016;22:1483–1495.
Wu Z, Tan J, Chi Y et al. Mesenteric adipose tissue contributes to intestinal barrier integrity and protects against nonalcoholic fatty liver disease in mice. Am J Physiol Gastrointest Liver Physiol. 2018;15:G659-670.
Schmidt J, Rattner D, Lewandrowski K et al. A better model of acute pancreatitis for evaluating therapy. Ann Surg. 1992;215:44–56.
Chiu C, Scott H, Gurd F. Intestinal mucosal lesion in low-flow states. II. The protective effect of intraluminal glucose as energy substrate. Archiv Surg. 1970;101:484–488.
Besselink M, Van Santvoort H, Renooij W et al. Intestinal barrier dysfunction in a randomized trial of a specific probiotic composition in acute pancreatitis. Ann Surg. 2009;250:712–719.
Shen Q, Xu G, Shen M. Effect of early enteral nutrition on endotoxin in serum and intestinal permeability in patients with severe acute pancreatitis. Eur Rev Med Pharmacol Sci. 2017;21:2764–2768.
O’Leary D, O’Neill D, Mclaughlin P et al. Effects of Abdominal Fat Distribution Parameters on Severity of Acute Pancreatitis. World Journal of Surgery. 2012;36:1679–1685.
Caesar R, Manieri M, Kelder T et al. A combined transcriptomics and lipidomics analysis of subcutaneous, epididymal and mesenteric adipose tissue reveals marked functional differences. PLoS ONE. 2010;5:e11525.
Cohen C, Shea A, Heffron C et al. Intraabdominal fat depots represent distinct immunomodulatory microenvironments: a murine model. PLoS ONE. 2013;8:e66477.
Wueest S, Yang X, Liu J et al. Inverse regulation of basal lipolysis in perigonadal and mesenteric fat depots in mice. Am J Physiol Endocrinol Metab. 2012;302:E153–E160.
Ko G, Liu K, So W et al. Cutoff values for central obesity in Chinese based on mesenteric fat thickness. Clin Nutr. 2009;28:679–683.
Catalano K, Stefanovski D, Bergman R. Critical role of the mesenteric depot versus other intra-abdominal adipose depots in the development of insulin resistance in young rats. Diabetes. 2010;59:1416–1423.
Navina S, Acharya C, Delany J et al. Lipotoxicity causes multisystem organ failure and exacerbates acute pancreatitis in obesity. Sci Transl Med 2011;3:107–110.
Durgampudi C, Noel P, Patel K et al. Acute Lipotoxicity Regulates Severity of Biliary Acute Pancreatitis without Affecting Its Initiation. Am J Pathol. 2014;184:1773–1784.
Lin T, Zhang Y, Wang Y et al. NAFLD aggravates acute pancreatitis through bacterial translocation and cholesterol metabolic dysregulation in the liver and pancreas in mice. Hepatobiliary Pancreat Dis Int 2022;S1499–3872(22):00180–00181.
Brestoff J, Artis D. Immune regulation of metabolic homeostasis in health and disease. Cell. 2015;161:146–160.
Beek L, Klinken J, Pronk A et al. The limited storage capacity of gonadal adipose tissue directs the development of metabolic disorders in male C57Bl/6J mice. Diabetologia. 2015;58:1601–1609.
Winer D, Winer S, Shen L et al. B lymphocytes promote insulin resistance through modulation of T cells and production of pathogenic IgG antibodies. Nat Med. 2011;17:610–617.
Ying W, Wollam J, Ofrecio J et al. Adipose tissue B2 cells promote insulin resistance through leukotriene LTB4/LTB4R1 signaling. J Clin Invest. 2017;127:1019–1030.
Defuria J, Belkina A, Jaqannathan-Bogdan M et al. B cells promote inflammation in obesity and type 2 diabetes through regulation of T-cell function and an inflammatory cytokine profile. Proc Natl Acad Sci USA 2013;110:5133–5138.
Wu Z, Xu J, Tan J et al. Mesenteric adipose tissue B lymphocytes promote local and hepatic inflammation in non-alcoholic fatty liver disease mice. J Cell Mol Med. 2019;23:3375–3385.
Acknowledgments
We thank Zhe Wu for the assistance of research design and analysis. We thank Shizhong Yang for the clinical support.
Funding
This study was supported by the Youth Fund of Beijing Tsinghua Changgung Hospital (12022C1004) and Tsinghua Precision Medicine Foundation (10001020127).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
All authors declare no competing interests.
Ethical approval
All protocols were approved by the Beijing Tsinghua Changgung Hospital Ethics Committee (22271-4-01).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, Q., Liu, J., Zhou, Z. et al. Inflammation of Mesenteric Adipose Tissue Correlates with Intestinal Injury and Disease Severity in Rats with Severe Acute Pancreatitis. Dig Dis Sci 68, 2474–2481 (2023). https://doi.org/10.1007/s10620-023-07846-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-023-07846-z