Abstract
Background
miR-29-3p, an important tumor suppressor, with inhibitory effects in multiple cancers that have been studied. Its exact molecular function is in HCC, however, still not been explored clearly. The purpose of our study is to make certain how miR-29c-3p affects HCC through TPX2.
Materials and Methods
Expression profile data of miR-29c-3p and TPX2 were acquired and downloaded from the TCGA database, and the respective differential expression was verified by qPCR and immunohistochemistry. The StarBase and dual luciferase reporter confirmed TPX2 targeting miR-29c-3p. Their effects on the biological functions of Hep3B and HepG2 were investigated by cellular assays.
Results
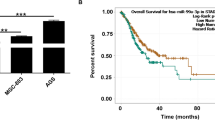
miR-29-3p was found to be significantly down-regulated in HCC, and the miR-29-3p low expression group had a poor prognosis. Overexpression of miR-29-3p was detrimental to invasion and migration ability of HCC cells and promoted their apoptosis. We identified miR-29c-3p targeting TPX2 by predictive analysis. TPX2 was significantly upregulated in HCC, and patients with high TPX2 expression had a poor prognosis. TPX2 knockdown partially counteracted the promoting effect of miR-29-3p inhibition on hepatocellular carcinoma cells, and its effect on hepatocellular carcinoma cell biology was similar to miR-29c-3p overexpression.
Conclusion
miR-29c, a key gene regulating HCC, is lowly expressed in HCC, its overexpression can remarkably inhibit the biological function of tumor cells. miR-29c can perform this function by regulating the expression of TPX2.






Similar content being viewed by others
References
McGlynn KA, Petrick JL, El-Serag HB. Epidemiology of hepatocellular carcinoma. Hepatology (Baltimore, Md.) 2021;73(1):4–13.
Chakraborty E, Sarkar D. Emerging therapies for hepatocellular carcinoma (HCC). Cancers. 2022;14:9.
Wang W, Wei C. Advances in the early diagnosis of hepatocellular carcinoma. Genes Dis. 2020;7:308–319.
Lee YS, Dutta A. MicroRNAs in cancer. Ann Rev Pathol. 2009;4:199–227.
Lujambio A, Lowe SW. The microcosmos of cancer. Nature. 2012;482:347–355.
Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136:215–233.
Lu J, Getz G, Miska EA et al. MicroRNA expression profiles classify human cancers. Nature. 2005;435:834–838.
Uddin A, Chakraborty S. Role of miRNAs in lung cancer. J Cell Physiol. 2018;9(8):773–776.
Zan Y, Wang B, Liang L et al. MicroRNA-139 inhibits hepatocellular carcinoma cell growth through down-regulating karyopherin alpha 2. J Exp Clin Cancer Res. 2019;38:182.
Fu W, Yu G, Liang J et al. miR-144-5p and miR-451a Inhibit the growth of cholangiocarcinoma cells through decreasing the expression of ST8SIA4. Front Oncol. 2020;10:563486.
Yen YT, Yang JC, Chang JB, Tsai SC. Down-regulation of miR-194-5p for predicting metastasis in breast cancer cells. Int J Mol Sci. 2021;23:15.
Chen C, Huang Z, Mo X et al. The circular RNA 001971/miR-29c-3p axis modulates colorectal cancer growth, metastasis, and angiogenesis through VEGFA. J Exp Clin Cancer Res. 2020;39:91.
Wang H, Fu L, Wei D et al. MiR-29c-3p suppresses the migration, invasion and cell cycle in esophageal carcinoma via CCNA2/p53 Axis. Front Bioeng Biotechnol. 2020;8:75.
Zou T, Gao Y, Qie M. MiR-29c-3p inhibits epithelial-mesenchymal transition to inhibit the proliferation, invasion and metastasis of cervical cancer cells by targeting SPARC. Ann Transl Med. 2021;9:125.
Hu Z, Cai M, Zhang Y, Tao L, Guo R. miR-29c-3p inhibits autophagy and cisplatin resistance in ovarian cancer by regulating FOXP1/ATG14 pathway. Cell Cycle (Georgetown, Tex.) 2020;19:193–206.
Hozaka Y, Seki N, Tanaka T et al. Molecular pathogenesis and regulation of the miR-29–3p-family: involvement of ITGA6 and ITGB1 in intra-hepatic cholangiocarcinoma. Cancers 2021;13:25.
Che J, Su Z, Yang Wet al. Tumor-suppressor p53 specifically binds to miR-29c-3p and reduces ADAM12 expression in hepatocellular carcinoma. Dig Liver Dis. 2022.
Wadsworth P. TPX. Curr Biol. 2015;25:1156–1158.
Neumayer G, Belzil C, Gruss OJ, Nguyen MD. TPX2: of spindle assembly DNA damage response, and cancer. Cell Mol Life Sci. 2014;71:3027–3047.
Aguirre-Portolés C, Bird AW, Hyman A, Cañamero M, Pérez de Castro I, Malumbres M. Tpx2 controls spindle integrity, genome stability, and tumor development. Cancer Res. 2012;72:1518–1528.
Wei P, Zhang N, Xu Y et al. TPX2 is a novel prognostic marker for the growth and metastasis of colon cancer. J Transl Med. 2013;11:313.
Warner SL, Stephens BJ, Nwokenkwo S et al. Validation of TPX2 as a potential therapeutic target in pancreatic cancer cells. Clin Cancer Res. 2009;15:6519–6528.
Zou Z, Zheng B, Li J et al. TPX2 level correlates with cholangiocarcinoma cell proliferation, apoptosis, and EMT. Biomed Pharmacother. 2018;107:1286–1293.
Ma Y, Lin D, Sun W et al. Expression of targeting protein for xklp2 associated with both malignant transformation of respiratory epithelium and progression of squamous cell lung cancer. Clin. Cancer Res. 2006;12:1121–1127.
Huang Y, Guo W, Kan H. TPX2 is a prognostic marker and contributes to growth and metastasis of human hepatocellular carcinoma. Int. J. Mol. Sci. 2014;15:18148–18161.
Paijens ST, Vledder A, de Bruyn M, Nijman HW. Tumor-infiltrating lymphocytes in the immunotherapy era. Cell. Mol. Immunol. 2021;18:842–859.
Lu C, Rong D, Zhang B et al. Current perspectives on the immunosuppressive tumor microenvironment in hepatocellular carcinoma: challenges and opportunities. Mol Cancer. 2019;18:130.
Fridman WH, Zitvogel L, Sautès-Fridman C, Kroemer G. The immune contexture in cancer prognosis and treatment. Nat Rev Clin Oncol. 2017;14:717–734.
Chen Z, Xie H, Hu M et al. Recent progress in treatment of hepatocellular carcinoma. Am J Cancer Res. 2020;10:2993–3036.
Liu Q, Tu K, Zhang H, Zheng X, Yao Y, Liu Q. TPX2 as a novel prognostic biomarker for hepatocellular carcinoma. Hepatol Res. 2015;45:906–918.
Huang DH, Jian J, Li S, Zhang Y, Liu LZ. TPX2 silencing exerts anti-tumor effects on hepatocellular carcinoma by regulating the PI3K/AKT signaling pathway. Int J Mol Med. 2019;44:2113–2122.
Zhang Y, Zhang Z. The history and advances in cancer immunotherapy: understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell Mol Immunol. 2020;17:807–821.
Konishi H, Sato H, Takahashi K, Fujiya M. Tumor-progressive mechanisms mediating miRNA-protein interaction. Int J Mol Sci. 2021;22:26.
Farazi TA, Spitzer JI, Morozov P, Tuschl T. miRNAs in human cancer. J Pathol. 2011;223:102–115.
Funding
Funding were provided by Anhui Key Research and Development Program Project (Grant No.: 202104j07020005) and Hefei Key Common Technology R&D and Engineering of Major Scientific and Technological Achievements Project (Grant No.: 2021YL001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, H., Chen, W., Qi, Y. et al. miR-29c Suppresses the Malignant Phenotype of Hepatocellular Carcinoma Cells In Vitro by Mediating TPX2 Associated with Immune Infiltration. Dig Dis Sci 68, 1923–1935 (2023). https://doi.org/10.1007/s10620-022-07810-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-022-07810-3