Abstract
Background
While the role of anti-drug antibodies in mediating loss of response to infliximab (IFX) is known, it is possible that there are different clinical implications for high compared to low titers of antibodies to infliximab (ATI). The impact of ATI titer on efficacy of subsequent anti-TNF treatment has not been established previously.
Methods
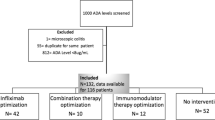
This is a multicenter retrospective cohort study of individuals with IBD who discontinued infliximab due to development of ATI and were subsequently switched to adalimumab therapy. IFX anti-drug antibody titer level was measured using Mayo or Esoterix assays. We examined clinical and endoscopic response to adalimumab at 3 months, 12 months, and 2 years.
Results
Our study included 90 patients (69 CD, 17 UC, 4 CD of the ileoanal pouch) with IBD. The median antibody titer levels for the Esoterix (208 u/mL) or Mayo clinic laboratory (236 u/mL) were similar (p > 0. 50). Patients with high ATI titers (median 824 u/ml, IQR 405–1250 u/ml) were as likely to respond to adalimumab as those with low titers (median 76u/ml, IQR 41–129 u/ml). At 3 months and 12 months, the rates of clinical response/remission to adalimumab therapy were 78% and 77%, respectively, among those with high ATI titers and 81% and 84% among those with low ATI titers (p = 0.81 and 0.62, respectively). In patients who initiated a different therapeutic mechanism after adalimumab, the response rates were similar to that observed with adalimumab.
Conclusions
The presence of high titers of anti-drug antibodies to IFX is not predictive of treatment failure with adalimumab.

Similar content being viewed by others
References
Vermeire S, Dreesen E, Papamichael K, Dubinsky MC. How, when, and for whom should we perform therapeutic drug monitoring? Clin Gastroenterol Hepatol. 2020;18:1291–1299.
Sazonovs A, Kennedy NA, Moutsianas L, Heap GA, Rice DL, Reppell M et al. HLA-DQA1*05 carriage associated with development of anti-drug antibodies to infliximab and adalimumab in patients with crohn’s disease. Gastroenterology. 2020;158:189–199.
Wong U, Cross RK. Primary and secondary nonresponse to infliximab: mechanisms and countermeasures. Expert Opin Drug Metab Toxicol. 2017;13:1039–1046.
Bor R, Farkas K, Fabian A, Balint A, Milassin A, Rutka M et al. Clinical role, optimal timing and frequency of serum infliximab and anti-infliximab antibody level measurements in patients with inflammatory bowel disease. PLoS ONE. 2017;12:e0172916.
Dreesen E, Van Stappen T, Ballet V, Peeters M, Compernolle G, Tops S et al. Anti-infliximab antibody concentrations can guide treatment intensification in patients with Crohn’s disease who lose clinical response. Aliment Pharmacol Ther. 2018;47:346–355.
Steenholdt C, Bendtzen K, Brynskov J, Thomsen OO, Ainsworth MA. Cut-off levels and diagnostic accuracy of infliximab trough levels and anti-infliximab antibodies in Crohn’s disease. Scand J Gastroenterol. 2011;46:310–318.
Vande Casteele N, Khanna R, Levesque BG, Stitt L, Zou GY, Singh S et al. The relationship between infliximab concentrations, antibodies to infliximab and disease activity in Crohn’s disease. Gut. 2015;64:1539–1545.
Frederiksen MT, Ainsworth MA, Brynskov J, Thomsen OO, Bendtzen K, Steenholdt C. Antibodies against infliximab are associated with de novo development of antibodies to adalimumab and therapeutic failure in infliximab-to-adalimumab switchers with IBD. Inflamm Bowel Dis. 2014;20:1714–1721.
Strik AS, van den Brink GR, Ponsioen C, Mathot R, Lowenberg M, D’Haens GR. Suppression of anti-drug antibodies to infliximab or adalimumab with the addition of an immunomodulator in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2017;45:1128–1134.
Ben-Horin S, Waterman M, Kopylov U, Yavzori M, Picard O, Fudim E et al. Addition of an immunomodulator to infliximab therapy eliminates antidrug antibodies in serum and restores clinical response of patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. 2013;11:444–447.
Colman RJ, Portocarrero-Castillo A, Chona D, Hellmann J, Minar P, Rosen MJ. Favorable outcomes and anti-TNF durability after addition of an immunomodulator for anti-drug antibodies in pediatric IBD Patients. Inflamm Bowel Dis. 2021;27:507–515.
Yanai H, Lichtenstein L, Assa A, Mazor Y, Weiss B, Levine A et al. Levels of drug and antidrug antibodies are associated with outcome of interventions after loss of response to infliximab or adalimumab. Clin Gastroenterol Hepatol. 2015;13:522-30e2.
van der Bijl AE, Breedveld FC, Antoni CE, Kalden JR, Kary S, Burmester GR et al. An open-label pilot study of the effectiveness of adalimumab in patients with rheumatoid arthritis and previous infliximab treatment: relationship to reasons for failure and anti-infliximab antibody status. Clin Rheumatol. 2008;27:1021–1028.
Singh S, George J, Boland BS, Vande Casteele N, Sandborn WJ. Primary non-response to tumor necrosis factor antagonists is associated with inferior response to second-line biologics in patients with inflammatory bowel diseases: a systematic review and meta-analysis. J Crohns Colitis. 2018;12:635–643.
Author information
Authors and Affiliations
Contributions
Ananthakrishnan and Sasson helped in study concept and design and editing; Ananthakrishnan contributed to statistical analysis and final approval of the manuscript; Sasson drafted the manuscript;
Corresponding author
Ethics declarations
Conflicts of interest
Sasson has no conflicts of interest to declare. Ananthakrishnan has served on the Scientific Advisory Boards of Gilead, Ikena Therapeutics, and Sun Pharma.
Ethics declarations
This retrospective chart review study involving human participants was in accordance with the ethical standards of the institutional and national research committee. The Partners Healthcare Institutional Review Board approved this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sasson, A.N., Ananthakrishnan, A.N. High Anti-Infliximab Antibody Titers Do Not Impact Response to Subsequent Adalimumab Treatment in Inflammatory Bowel Diseases. Dig Dis Sci 67, 3124–3128 (2022). https://doi.org/10.1007/s10620-021-07088-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-021-07088-x