Abstract
Background
Differentiated embryonic chondrocyte expressed genes (DECs) are critical regulators of cellular proliferation and differentiation. However, DEC1 and DEC2 as family member have opposite or identical roles in tumor, acting as an “accelerator” or a “brake” in progression.
Aims
The possible crosstalk between DEC1 and DEC2 in the gastric cancer (GC).
Methods
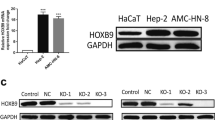
The association of DEC1 and DEC2 expression with prognosis was investigated by immunohistochemistry. The expression pattern of DECs in GC cells was examined using the CCLE database. DECs knockdown or overexpression was conducted via lentiviral transfection. The proliferation of GC cells was evaluated by CCK8, EdU, and Colony forming. ChIP and luciferase reporter assays were used to verify interaction between DEC1 and the DEC2 promoter. The combination downstream with DEC1 and DEC2 was predicted by bioinformation, with Western blot providing further verification.
Results
We found that reciprocal expression of DEC1 and DEC2 works together to sustain the progression of GC by promoting cell growth. We confirmed this observation in vivo, showing that inhibition DEC1expression could increase DEC2 expression. DEC1 suppresses DEC2 expression by directly binding to the E-box of the DEC2 promoter in GC cells. Furthermore, this regulation of DEC1 on DEC2 enables the further indirect or cooperative activation of additional downstream target genes, MAPK, and STAT3.
Conclusion
Our data demonstrate that DEC1 and DEC2 interact physically and functionally and identify a novel mode of cross-regulatory interaction between DECs that abrogates their functional activity.





Similar content being viewed by others
References
Boudjelal M, Taneja R, Matsubara S, Bouillet P, Dolle P, Chambon P. Overexpression of Stra13, a novel retinoic acid-inducible gene of the basic helix-loop-helix family, inhibits mesodermal and promotes neuronal differentiation of P19 cells. Genes Dev 1997;11:2052–2065. https://doi.org/10.1101/gad.11.16.2052.
Fujimoto K, Shen M, Noshiro M, Matsubara K, Shingu S, Honda K et al. Molecular cloning and characterization of DEC2, a new member of basic helix-loop-helix proteins. Biochem Biophys Res Commun 2001;280:164–171. https://doi.org/10.1006/bbrc.2000.4133.
Rossner MJ, Dorr J, Gass P, Schwab MH, Nave KA. SHARPs: mammalian enhancer-of-split- and hairy-related proteins coupled to neuronal stimulation. Mol Cell Neurosci 1997;10:460–475. https://doi.org/10.1006/mcne.1997.0640.
Shen M, Kawamoto T, Yan W, Nakamasu K, Tamagami M, Koyano Y et al. Molecular characterization of the novel basic helix-loop-helix protein DEC1 expressed in differentiated human embryo chondrocytes. Biochem Biophys Res Commun 1997;236:294–298. https://doi.org/10.1006/bbrc.1997.6960.
Liu Q, Wu Y, Seino H, Haga T, Yoshizawa T, Morohashi S et al. Correlation between DEC1/DEC2 and epithelialmesenchymal transition in human prostate cancer PC3 cells. Mol Med Rep 2018;18:3859–3865. https://doi.org/10.3892/mmr.2018.9367.
Gallo C, Fragliasso V, Donati B, Torricelli F, Tameni A, Piana S et al. The bHLH transcription factor DEC1 promotes thyroid cancer aggressiveness by the interplay with NOTCH1. Cell Death Dis 2018;9:871. https://doi.org/10.1038/s41419-018-0933-y.
Wu Y, Sato F, Bhawal UK, Kawamoto T, Fujimoto K, Noshiro M et al. Basic helix-loop-helix transcription factors DEC1 and DEC2 regulate the paclitaxel-induced apoptotic pathway of MCF-7 human breast cancer cells. Int J Mol Med 2011;27:491–495. https://doi.org/10.3892/ijmm.2011.617.
Seino H, Wu Y, Morohashi S, Kawamoto T, Fujimoto K, Kato Y et al. Basic helix-loop-helix transcription factor DEC1 regulates the cisplatin-induced apoptotic pathway of human esophageal cancer cells. Biomed Res 2015;36:89–96. https://doi.org/10.2220/biomedres.36.89.
Wu Y, Sato F, Bhawal UK, Kawamoto T, Fujimoto K, Noshiro M et al. BHLH transcription factor DEC2 regulates pro-apoptotic factor Bim in human oral cancer HSC-3 cells. Biomed Res 2012;33:75–82. https://doi.org/10.2220/biomedres.33.75.
Jia Y, Hu R, Li P, Zheng Y, Wang Y, Ma X. DEC1 is required for anti-apoptotic activity of gastric cancer cells under hypoxia by promoting Survivin expression. Gastric Cancer 2018;21:632–642. https://doi.org/10.1007/s10120-017-0780-z.
Jia YF, Xiao DJ, Ma XL, Song YY, Hu R, Kong Y et al. Differentiated embryonic chondrocyte-expressed gene 1 is associated with hypoxia-inducible factor 1alpha and Ki67 in human gastric cancer. Diagn Pathol 2013;8:37. https://doi.org/10.1186/1746-1596-8-37.
Li H, Ma X, Xiao D, Jia Y, Wang Y. Expression of DEC2 enhances chemosensitivity by inhibiting STAT5A in gastric cancer. J Cell Biochem. 2018. https://doi.org/10.1002/jcb.28131.
Li P, Jia YF, Ma XL, Zheng Y, Kong Y, Zhang Y et al. DEC2 suppresses tumor proliferation and metastasis by regulating ERK/NF-kappaB pathway in gastric cancer. Am J Cancer Res 2016;6:1741–1757
Zheng Y, Jia Y, Wang Y, Wang M, Li B, Shi X et al. The hypoxia-regulated transcription factor DEC1 (Stra13, SHARP-2) and its expression in gastric cancer. Omics J Integr Biol 2009;13:301–306. https://doi.org/10.1089/omi.2009.0014.
Jia Y, Gu D, Wan J, Yu B, Zhang X, Chiorean EG et al. The role of GLI-SOX2 signaling axis for gemcitabine resistance in pancreatic cancer. Oncogene 2019;38:1764–1777. https://doi.org/10.1038/s41388-018-0553-0.
Li XM, Lin W, Wang J, Zhang W, Yin AA, Huang Y et al. Dec1 expression predicts prognosis and the response to temozolomide chemotherapy in patients with glioma. Mol Med Rep 2016;14:5626–5636. https://doi.org/10.3892/mmr.2016.5921.
Wang W, Reiser-Erkan C, Michalski CW, Raggi MC, Quan L, Yupei Z et al. Hypoxia inducible BHLHB2 is a novel and independent prognostic marker in pancreatic ductal adenocarcinoma. Biochem Biophys Res Commun 2010;401:422–428. https://doi.org/10.1016/j.bbrc.2010.09.070.
Montagner M, Enzo E, Forcato M, Zanconato F, Parenti A, Rampazzo E et al. SHARP1 suppresses breast cancer metastasis by promoting degradation of hypoxia-inducible factors. Nature 2012;487:380–384. https://doi.org/10.1038/nature11207.
Sato F, Otsuka T, Kohsaka A, Le HT, Bhawal UK, Muragaki Y. Smad3 suppresses epithelial cell migration and proliferation via the clock gene Dec1, which negatively regulates the expression of clock genes Dec2 and Per1. Am J Pathol 2019;189:773–783. https://doi.org/10.1016/j.ajpath.2019.01.006.
Liu Y, Sato F, Kawamoto T, Fujimoto K, Morohashi S, Akasaka H et al. Anti-apoptotic effect of the basic helix-loop-helix (bHLH) transcription factor DEC2 in human breast cancer cells. Genes Cells 2010;15:315–325. https://doi.org/10.1111/j.1365-2443.2010.01381.x.
Li Y, Xie M, Song X, Gragen S, Sachdeva K, Wan Y et al. DEC1 negatively regulates the expression of DEC2 through binding to the E-box in the proximal promoter. J Biol Chem 2003;278:16899–16907. https://doi.org/10.1074/jbc.M300596200.
Khan SA, Amnekar R, Khade B, Barreto SG, Ramadwar M, Shrikhande SV et al. p38-MAPK/MSK1-mediated overexpression of histone H3 serine 10 phosphorylation defines distance-dependent prognostic value of negative resection margin in gastric cancer. Clin Epigenet 2016;8:88. https://doi.org/10.1186/s13148-016-0255-9.
Ma W, Xu Z, Wang Y, Li W, Wei Z, Chen T et al. A positive feedback loop of SLP2 activates MAPK signaling pathway to promote gastric cancer progression. Theranostics 2018;8:5744–5757. https://doi.org/10.7150/thno.28898.
Chen Z, Huang W, Tian T, Zang W, Wang J, Liu Z et al. Characterization and validation of potential therapeutic targets based on the molecular signature of patient-derived xenografts in gastric cancer. J Hematol Oncol 2018;11:20. https://doi.org/10.1186/s13045-018-0563-y.
Soutto M, Chen Z, Bhat AA, Wang L, Zhu S, Gomaa A et al. Activation of STAT3 signaling is mediated by TFF1 silencing in gastric neoplasia. Nat Commun 2019;10:3039. https://doi.org/10.1038/s41467-019-11011-4.
Acknowledgments
This study was financially supported by the National Natural Science Foundation of China (Nos. 31671468 and 31970728), the Academic promotion programme of Shandong First Medical University (No. 2019QL024), the Shandong Provincial Natural Science Foundation of China (Nos. ZR2016HM15and ZR2018MH021)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10620_2021_6921_MOESM2_ESM.tif
Real-time PCR analysis of DEC1 and DEC2 expression in DEC1 silenced or overexpression and DEC2 overespression GC cells. EV, empty vector; over, overexpression. Data are represented as the means ± SD. *P < 0.05 (TIF 309 KB)
Rights and permissions
About this article
Cite this article
Li, B., Chu, Y., Yan, B. et al. Reciprocal Expression of Differentiated Embryonic Chondrocyte Expressed Genes Result in Functional Antagonism in Gastric Cancer. Dig Dis Sci 67, 904–914 (2022). https://doi.org/10.1007/s10620-021-06921-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-021-06921-7