Abstract
Background
An immature intestine is a high-risk factor for necrotizing enterocolitis (NEC), which is a serious intestinal disease in newborns. The regulation of developmentally regulated GTP-binding protein 1 (DRG1) during organ development suggests a potential role of DRG1 in the maturation process of the intestine.
Aim
To illustrate the function of DRG1 during the pathogenesis of NEC.
Methods
DRG1 expression in the intestine was measured using immunohistochemistry and q-PCR. Immunoprecipitation coupled with mass spectrometry was used to identify the interacting proteins of DRG1. The biological functions of the potential interactors were annotated with the Database for Annotation, Visualization and Integrated Discovery. Caco2 and FHs74Int cells with stable DRG1 silencing or overexpression were used to investigate the influence of DRG1 on cell junctions and intestinal barrier permeability and to elucidate the downstream mechanism.
Results
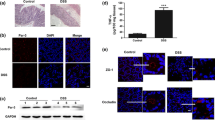
DRG1 was constitutively expressed during the intestinal maturation process but significantly decreased in the ileum in the context of NEC. Protein interaction analysis revealed that DRG1 was closely correlated with cell junctions. DRG1 deficiency destabilized the E-cadherin and occludin proteins near the cell membrane and increased the permeability of the epithelial cell monolayer, while DRG1 overexpression prevented lipopolysaccharide-induced disruption of E-cadherin and occludin expression and cell monolayer integrity. Further investigation suggested that DRG1 maintained cell junctions, especially adherens junctions, by regulating RAC1 activity, and RAC1 inhibition with NSC23766 attenuated intestinal injury and led to improved barrier integrity in experimental NEC.
Conclusions
Our findings illustrate the mechanism underlying the effect of DRG1 deficiency on epithelial cell permeability regulation and provide evidence supporting the application of RAC1 inhibitors for protection against NEC.







Similar content being viewed by others
References
Neu J, Walker WA. Necrotizing enterocolitis. N Engl J Med. 2011;364:255–264. https://doi.org/10.1056/NEJMra1005408.
Mathews S, Xu M, Wang H, Bertola A, Gao B. Animals models of gastrointestinal and liver diseases. Animal models of alcohol-induced liver disease: pathophysiology, translational relevance, and challenges. Am J Physiol Gastrointest Liver Physiol. 2014;306:G819–G823. https://doi.org/10.1152/ajpgi.00041.2014.
Tanner SM, Berryhill TF, Ellenburg JL, et al. Pathogenesis of necrotizing enterocolitis: modeling the innate immune response. Am J Pathol. 2015;185:4–16. https://doi.org/10.1016/j.ajpath.2014.08.028.
Liu Z, Li N, Neu J. Tight junctions, leaky intestines, and pediatric diseases. Acta Paediatr. 2005;94:386–393. https://doi.org/10.1111/j.1651-2227.2005.tb01904.x.
Guo S, Nighot M, Al-Sadi R, Alhmoud T, Nighot P, Ma TY. Lipopolysaccharide regulation of intestinal tight junction permeability is mediated by TLR4 signal transduction pathway activation of FAK and MyD88. J Immunol. 2015;195:4999–5010. https://doi.org/10.4049/jimmunol.1402598.
Li B, Lee C, Cadete M, Miyake H, Lee D, Pierro A. Neonatal intestinal organoids as an ex vivo approach to study early intestinal epithelial disorders. Pediatr Surg Int. 2019;35:3–7. https://doi.org/10.1007/s00383-018-4369-3.
Xiao S, Li Q, Hu K, et al. Vitamin A and retinoic acid exhibit protective effects on necrotizing enterocolitis by regulating intestinal flora and enhancing the intestinal epithelial barrier. Arch Med Res. 2018;49:1–9. https://doi.org/10.1016/j.arcmed.2018.04.003.
Grothaus JS, Ares G, Yuan C, Wood DR, Hunter CJ. Rho kinase inhibition maintains intestinal and vascular barrier function by upregulation of occludin in experimental necrotizing enterocolitis. Am J Physiol Gastrointest Liver Physiol. 2018;315:G514–G528. https://doi.org/10.1152/ajpgi.00357.2017.
Blackwood BP, Yuan CY, Wood DR, Nicolas JD, Grothaus JS, Hunter CJ. Probiotic Lactobacillus species strengthen intestinal barrier function and tight junction integrity in experimental necrotizing enterocolitis. J Probiotics Health. 2017;5:159. https://doi.org/10.4172/2329-8901.1000159.
Buonpane C, Yuan C, Wood D, Ares G, Klonoski SC, Hunter CJ. ROCK1 inhibitor stabilizes E-cadherin and improves barrier function in experimental necrotizing enterocolitis. Am J Physiol Gastrointest Liver Physiol. 2020;318:G781–G792. https://doi.org/10.1152/ajpgi.00195.2019.
Sazuka T, Tomooka Y, Ikawa Y, Noda M, Kumar S. DRG: a novel developmentally regulated GTP-binding protein. Biochem Biophys Res Commun. 1992;189:363–370.
Li B, Trueb B. DRG represents a family of two closely related GTP-binding proteins. Biochem Biophys Acta. 2000;1491:196–204.
Lu L, Lv Y, Dong J, Hu S, Peng R. DRG1 is a potential oncogene in lung adenocarcinoma and promotes tumor progression via spindle checkpoint signaling regulation. Oncotarget. 2016;7:72795–72806. https://doi.org/10.18632/oncotarget.11973.
Schellhaus AK, Moreno-Andres D, Chugh M, et al. Developmentally regulated GTP binding protein 1 (DRG1) controls microtubule dynamics. Sci Rep. 2017;7:9996. https://doi.org/10.1038/s41598-017-10088-5.
Chen X, Kumamoto CA. A conserved G protein (Drg1p) plays a role in regulation of invasive filamentation in Candida albicans. Microbiology. 2006;152:3691–3700. https://doi.org/10.1099/mic.0.29246-0.
Mahajan MA, Park ST, Sun XH. Association of a novel GTP binding protein, DRG, with TAL oncogenic proteins. Oncogene. 1996;12:2343–2350.
Sazuka T, Kinoshita M, Tomooka Y, Ikawa Y, Noda M, Kumar S. Expression of DRG during murine embryonic development. Biochem Biophys Res Commun. 1992;189:371–377.
Jilling T, Simon D, Lu J, et al. The roles of bacteria and TLR4 in rat and murine models of necrotizing enterocolitis. J Immunol. 2006;177:3273–3282.
Caplan MS, Hedlund E, Adler L, Hsueh W. Role of asphyxia and feeding in a neonatal rat model of necrotizing enterocolitis. Pediatr Pathol. 1994;14:1017–1028.
Lu L, Hu S, Wei R, et al. The HECT type ubiquitin ligase NEDL2 is degraded by anaphase-promoting complex/cyclosome (APC/C)-Cdh1, and its tight regulation maintains the metaphase to anaphase transition. J Biol Chem. 2013;288:35637–35650. https://doi.org/10.1074/jbc.M113.472076.
Ma TY, Hoa NT, Tran DD, et al. Cytochalasin B modulation of Caco-2 tight junction barrier: role of myosin light chain kinase. Am J Physiol Gastrointest Liver Physiol. 2000;279:G875–885. https://doi.org/10.1152/ajpgi.2000.279.5.G875.
Bein A, Lubetzky R, Mandel D, Schwartz B. TIMP-1 inhibition of occludin degradation in Caco-2 intestinal cells: a potential protective role in necrotizing enterocolitis. Pediatr Res. 2015;77:649–655. https://doi.org/10.1038/pr.2015.26.
Ling X, Linglong P, Weixia D, Hong W. Protective effects of bifidobacterium on intestinal barrier function in LPS-induced enterocyte barrier injury of Caco-2 monolayers and in a rat NEC model. PLoS One. 2016;11:e0161635. https://doi.org/10.1371/journal.pone.0161635.
Cevik M, Karadag CA, Sakiz DE, Tander B, Embleton DD. The role of nitric oxide in an experimental necrotising enterocolitis model. Afr J Paediatr Surg. 2013;10:24–28. https://doi.org/10.4103/0189-6725.109381.
Pietzcker J, Kluthea C, Klinghammer K, Buhrer C, Rustow B, Guthmann F. Developmental delay in hypoxia-induced HO-1 expression predisposes to gut injury. J Perinat Med. 2012;40:191–197. https://doi.org/10.1515/jpm.2011.117.
Ferretti E, Tremblay E, Thibault MP, et al. The nitric oxide synthase 2 pathway is targeted by both pro- and anti-inflammatory treatments in the immature human intestine. Nitric Oxide. 2017;66:53–61. https://doi.org/10.1016/j.niox.2017.03.003.
Chen G, Li Y, Su Y, et al. Identification of candidate genes for necrotizing enterocolitis based on microarray data. Gene. 2018;661:152–159. https://doi.org/10.1016/j.gene.2018.03.088.
Miyazono Y, Hirono A, Miyamoto Y, Miwa S. Erythrocyte enzyme activities in cord blood of extremely low-birth-weight infants. Am J Hematol. 1999;62:88–92.
Iwanami K, Matsumoto I, Tanaka-Watanabe Y, et al. Crucial role of the interleukin-6/interleukin-17 cytokine axis in the induction of arthritis by glucose-6-phosphate isomerase. Arthritis Rheum. 2008;58:754–763. https://doi.org/10.1002/art.23222.
Kim HR, Lee A, Choi EJ, et al. Attenuation of experimental colitis in glutathione peroxidase 1 and catalase double knockout mice through enhancing regulatory T cell function. PLoS ONE. 2014;9:e95332. https://doi.org/10.1371/journal.pone.0095332.
Colliou N, Ge Y, Sahay B, et al. Commensal Propionibacterium strain UF1 mitigates intestinal inflammation via Th17 cell regulation. J Clin Investig. 2017;127:3970–3986. https://doi.org/10.1172/JCI95376.
Ko MS, Kim HJ, Kim HK, et al. Developmentally regulated GTP-binding protein 2 ameliorates EAE by suppressing the development of TH17 cells. Clin Immunol. 2014;150:225–235. https://doi.org/10.1016/j.clim.2013.12.004.
Funding
This work was supported by the Shanghai Pu Jiang Program (19PJ1409000), a grant from Shanghai Jiao Tong University (YG2015ZD13), the Natural Science Foundation of Shanghai (18ZR1431300), and the National Natural Science Foundation of China (81503089).
Author information
Authors and Affiliations
Contributions
All authors participated in the acquisition of data and the revision of the manuscript. All authors read and approved the final manuscript. LL conducted all the studies, analyzed the data, and drafted the manuscript. ZL was responsible for the conception and design of the study and revised the manuscript critically for important intellectual content. WX and JL supplied the clinical samples and information. LC and SH helped to construct the NEC mouse model and performed the HE and IHC assays. QS and MZ participated in the data collection and analysis.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lu, L., Xu, W., Liu, J. et al. DRG1 Maintains Intestinal Epithelial Cell Junctions and Barrier Function by Regulating RAC1 Activity in Necrotizing Enterocolitis. Dig Dis Sci 66, 4237–4250 (2021). https://doi.org/10.1007/s10620-020-06812-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-020-06812-3