Abstract
Background and Aims
Advanced F3–4 fibrosis predicts liver-related mortality in nonalcoholic fatty liver disease (NAFLD). Noninvasive tests, designed to rule in/out advanced fibrosis, are limited by indeterminates, necessitating biopsy. We aimed to determine whether stepwise combinations of noninvasive serum-based tests and elastography (VCTE) could predict F3–4, reduce indeterminates, and decrease liver biopsies.
Methods and Results
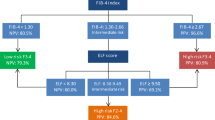
Five hundred forty-one biopsy-proven NAFLD cases were identified between 2010 and 2018 from two Canadian centers. Characteristics of training (n = 407)/validation (n = 134) cohorts included: males 54%/59%; mean age 48.5/52.5 years; mean body mass index 32.3/33.6 kg/m2; diabetes mellitus 30%/34%; and F3–4 48%/43%. For training/validation cohorts, area under the receiver operating curve (AUROC) for FIB-4, AST-platelet ratio index (APRI), NAFLD fibrosis score (NFS), BARD score, and AST/ALT ratio ranged from 0.70 to 0.83/0.68 to 0.81, with indeterminates 25–39%/34–45%, for F3–4. In the training cohort, parallel FIB-4 + NFS had good accuracy (AUROC = 0.81) but was limited by 38% indeterminates and 16% misclassified. Sequential FIB-4 → NFS reduced indeterminates to 10%, and FIB-4 → VCTE to 0%, misclassified 20–22%, while maintaining high specificity (0.88–0.92) and accuracy (AUROC 0.75–0.78) for combined cohorts. Liver biopsy could have been avoided in 27–29% of patients using sequential algorithms.
Conclusions
Sequential FIB-4 ➔ NFS/VCTE predicts F3–4 with high specificity and good accuracy, while reducing indeterminates and need for biopsy. Parallel algorithms are limited by high indeterminates. Sequential FIB-4 ➔ NFS had similar accuracy to VCTE-containing algorithms. Validation in low-prevalence cohorts may allow for potential use in community or resource-limited areas for risk stratification.


Similar content being viewed by others
References
Younossi ZM, Blissett D, Blissett R, et al. The economic and clinical burden of nonalcoholic fatty liver disease in the United States and Europe. Hepatology.. 2016;64:1577–1586.
Younossi ZM, Koenig AB, Abdelatif D, et al. Global epidemiology of nonalcoholic fatty liver disease—meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology.. 2016;64:73–84.
Araujo AR, Rosso N, Bedogni G, et al. Global epidemiology of non-alcoholic fatty liver disease/non-alcoholic steatohepatitis: what we need in the future. Liver Int.. 2018;38:47–51.
Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology.. 2018;67:328–357.
Baffy G, Brunt EM, Caldwell SH. Hepatocellular carcinoma in non-alcoholic fatty liver disease: an emerging menace. J Hepatol.. 2012;56:1384–1391.
Wong RJ, Aguilar M, Cheung R, et al. Nonalcoholic steatohepatitis is the second leading etiology of liver disease among adults awaiting liver transplantation in the United States. Gastroenterology.. 2015;148:547–555.
Byrne CD, Targher G. EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease: is universal screening appropriate? Diabetologia.. 2016;59:1141–1144.
Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology.. 2005;41:1313–1321.
Musso G, Cassader M, Gambino R. Non-alcoholic steatohepatitis: emerging molecular targets and therapeutic strategies. Nat Rev Drug Discov.. 2016;15:249–274.
Angulo P, Kleiner DE, Dam-Larsen S, et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology.. 2015;149:389–397.
Hagstrom H, Nasr P, Ekstedt M, et al. Fibrosis stage but not NASH predicts mortality and time to development of severe liver disease in biopsy-proven NAFLD. J Hepatol.. 2017;67:1265–1273.
Dulai PS, Singh S, Patel J, et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: systematic review and meta-analysis. Hepatology.. 2017;65:1557–1565.
Castera L, Friedrich-Rust M, Loomba R. Noninvasive assessment of liver disease in patients with nonalcoholic fatty liver disease. Gastroenterology.. 2019;156:1264–1281.
Vuppalanchi R, Siddiqui MS, Van Natta ML, et al. Performance characteristics of vibration-controlled transient elastography for evaluation of nonalcoholic fatty liver disease. Hepatology.. 2018;67:134–144.
Bedossa P, Patel K. Biopsy and noninvasive methods to assess progression of nonalcoholic fatty liver disease. Gastroenterology.. 2016;150:1811–1822.
Musso G, Gambino R, Cassader M, et al. Meta-analysis: natural history of non-alcoholic fatty liver disease (NAFLD) and diagnostic accuracy of non-invasive tests for liver disease severity. Ann Med.. 2011;43:617–649.
Sumida Y, Yoneda M, Hyogo H, et al. Validation of the FIB4 index in a Japanese nonalcoholic fatty liver disease population. BMC Gastroenterol.. 2012;12:2.
Loomba R, Adams LA. Advances in non-invasive assessment of hepatic fibrosis. Gut. 2020.
Patel K, Sebastiani G. Limitations of non-invasive tests for assessment of liver fibrosis. JHEP Rep.. 2020;2:100067.
Sebastiani G, Vario A, Guido M, et al. Stepwise combination algorithms of non-invasive markers to diagnose significant fibrosis in chronic hepatitis C. J Hepatol.. 2006;44:686–693.
Castera L, Sebastiani G, Le Bail B, et al. Prospective comparison of two algorithms combining non-invasive methods for staging liver fibrosis in chronic hepatitis C. J Hepatol.. 2010;52:191–198.
Srivastava A, Gailer R, Tanwar S, et al. Prospective evaluation of a primary care referral pathway for patients with non-alcoholic fatty liver disease. J Hepatol.. 2019;71:371–378.
Davyduke T, Tandon P, Al-Karaghouli M, et al. Impact of implementing a “FIB-4 First” strategy on a pathway for patients with NAFLD referred from primary care. Hepatol Commun.. 2019;3:1322–1333.
Petta S, Wong VW, Camma C, et al. Serial combination of non-invasive tools improves the diagnostic accuracy of severe liver fibrosis in patients with NAFLD. Aliment Pharmacol Ther.. 2017;46:617–627.
Boursier J, Guillaume M, Leroy V, et al. New sequential combinations of non-invasive fibrosis tests provide an accurate diagnosis of advanced fibrosis in NAFLD. J Hepatol.. 2019;71:389–396.
EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J Hepatol. 2016;64:1388–402.
Angulo P, Hui JM, Marchesini G, et al. The NAFLD fibrosis score: a noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology.. 2007;45:846–854.
Sterling RK, Lissen E, Clumeck N, et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology.. 2006;43:1317–1325.
Harrison SA, Oliver D, Arnold HL, et al. Development and validation of a simple NAFLD clinical scoring system for identifying patients without advanced disease. Gut.. 2008;57:1441–1447.
Wai CT, Greenson JK, Fontana RJ, et al. A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology.. 2003;38:518–526.
Shah AG, Lydecker A, Murray K, et al. Comparison of noninvasive markers of fibrosis in patients with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol.. 2009;7:1104–1112.
Angulo P, Bugianesi E, Bjornsson ES, et al. Simple noninvasive systems predict long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology.. 2013;145:782–789.
DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics.. 1988;44:837–845.
Obuchowski NA. An ROC-type measure of diagnostic accuracy when the gold standard is continuous-scale. Stat Med.. 2006;25:481–493.
Sun W, Cui H, Li N, et al. Comparison of FIB-4 index, NAFLD fibrosis score and BARD score for prediction of advanced fibrosis in adult patients with non-alcoholic fatty liver disease: a meta-analysis study. Hepatol Res.. 2016;46:862–870.
Anstee QM, Lawitz EJ, Alkhouri N, et al. Noninvasive tests accurately identify advanced fibrosis due to NASH: Baseline data from the STELLAR trials. Hepatology. 2019.
Chan WK, Treeprasertsuk S, Goh GB, et al. Optimizing use of nonalcoholic fatty liver disease fibrosis score, fibrosis-4 score, and liver stiffness measurement to identify patients with advanced fibrosis. Clin Gastroenterol Hepatol.. 2019;17:2570–2580.
Staufer K, Halilbasic E, Spindelboeck W, et al. Evaluation and comparison of six noninvasive tests for prediction of significant or advanced fibrosis in nonalcoholic fatty liver disease. United European Gastroenterol J.. 2019;7:1113–1123.
Onnerhag K, Hartman H, Nilsson PM, et al. Non-invasive fibrosis scoring systems can predict future metabolic complications and overall mortality in non-alcoholic fatty liver disease (NAFLD). Scand J Gastroenterol.. 2019;54:328–334.
Lambert J, Halfon P, Penaranda G, et al. How to measure the diagnostic accuracy of noninvasive liver fibrosis indices: the area under the ROC curve revisited. Clin Chem.. 2008;54:1372–1378.
Boursier J, Vergniol J, Guillet A, et al. Diagnostic accuracy and prognostic significance of blood fibrosis tests and liver stiffness measurement by FibroScan in non-alcoholic fatty liver disease. J Hepatol.. 2016;65:570–578.
McPherson S, Hardy T, Dufour JF, et al. Age as a confounding factor for the accurate non-invasive diagnosis of advanced NAFLD fibrosis. Am J Gastroenterol.. 2017;112:740–751.
De Silva S, Li W, Kemos P, et al. Non-invasive markers of liver fibrosis in fatty liver disease are unreliable in people of South Asian descent. Frontline Gastroenterol.. 2018;9:115–121.
Xiao G, Zhu S, Xiao X, et al. Comparison of laboratory tests, ultrasound, or magnetic resonance elastography to detect fibrosis in patients with nonalcoholic fatty liver disease: a meta-analysis. Hepatology.. 2017;66:1486–1501.
Tapper EB, Sengupta N, Huniuk MGM, Afdhal NH, Lai M. Cost-effective evaluation of nonalcoholic fatty liver disease with NAFLD fibrosis score and vibration controlled transient elastography. Am J Gastroenterol.. 2015;110:1298–1304.
Armstrong MJ, Houlihan DD, Bentham L, et al. Presence and severity of non-alcoholic fatty liver disease in a large prospective primary care cohort. J Hepatol.. 2012;56:234–240.
Ekstedt M, Hagstrom H, Nasr P, et al. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology.. 2015;61:1547–1554.
Crossan C, Majumdar A, Srivastava A, et al. Referral pathways for patients with NAFLD based on non-invasive fibrosis tests: diagnostic accuracy and cost analysis. Liver Int.. 2019;39:2052–2060.
Petta S, Wong VW, Camma C, et al. Improved noninvasive prediction of liver fibrosis by liver stiffness measurement in patients with nonalcoholic fatty liver disease accounting for controlled attenuation parameter values. Hepatology.. 2017;65:1145–1155.
Acknowledgment
Study sponsors did not play any role in the study design, data collection or analysis, interpretation of data, in the writing of the report, or in the decision to submit the manuscript for publication.
Funding
This study was supported by a research Grant from Gilead Sciences Canada. GS is supported by a Junior 1 and 2 Salary Award from FRQS (Nos. 27127 and 267806) and research salary from the Department of Medicine of McGill University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
GS has acted as speaker for Merck, Gilead, Abbvie, Novonordisk, Novartis, served as an advisory board member for Merck, Novartis, Gilead and Intercept, and has received unrestricted research funding from Merck and Theratechnologies Inc. KP has the following declarations: (1) Gilead-Advisory Board, consulting, research grants; (2) Novartis-Advisory Board; (3) Intercept-Advisory Board.
Ethics and consent
This study was performed according to the protocol and guidelines of Good Clinical Practice/ICH, based upon the principles outlined in the Declaration of Helsinki and local and national guidelines governing the conduct of clinical research studies. All patient data used for the purpose of this retrospective study were de-identified and anonymized, and patient consent was not required. This study was approved by the Institutional Research Ethics Board at both the University Health Network and the McGill University Health Centre.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kosick, H.MK., Keyrouz, A., Adeyi, O. et al. A Stepwise Algorithmic Approach and External Validation Study for Noninvasive Prediction of Advanced Fibrosis in Nonalcoholic Fatty Liver Disease. Dig Dis Sci 66, 4046–4057 (2021). https://doi.org/10.1007/s10620-020-06748-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-020-06748-8