Abstract
Background
Acute pancreatitis (AP) is a common inflammatory disease that may develop to severe AP (SAP), resulting in life-threatening complications. Impaired autophagic flux is a characteristic of early AP, and its accumulation could activate oxidative stress and nuclear factor κB (NF-κB) pathways, which aggravate the disease process.
Aim
To explore the therapeutic effects of regulating autophagy after the onset of AP.
Methods
In this study, intraperitoneal injections of 3-methyladenine (3-MA) and rapamycin (RAPA) in the l-arginine or cerulein plus lipopolysaccharide (LPS) Balb/C mouse model. At 24 h after the last injection, pulmonary, intestinal, renal and pancreatic tissues were analyzed.
Results
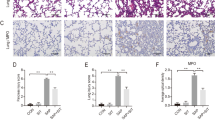
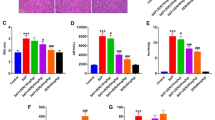
We found that 3-MA ameliorated systemic organ injury in two SAP models. 3-MA treatment impaired autophagic flux and alleviated inflammatory activation by modulating the NF-κB signaling pathway and the caspase-1-IL-1β pathway, thus decreasing the injuries to the organs and the levels of inflammatory cytokines.
Conclusion
Our study found that the regulation of autophagy could alter the progression of AP induced by l-arginine or cerulein plus LPS in mice.







Similar content being viewed by others
Abbreviations
- AP:
-
Acute pancreatitis
- SAP:
-
Severe AP
- NF-κB:
-
Nuclear factor κB
- IP:
-
Intraperitoneal
- 3-MA:
-
3-methyladenine
- RAPA:
-
Rapamycin
- LPS:
-
Lipopolysaccharide
- LC3-II:
-
Microtubule-associated protein 1 light chain 3
- ZO-1:
-
Zonula occludens-1
- IHC:
-
Immunohistochemistry
- LDH:
-
Lactate dehydrogenase
- ELISA:
-
Enzyme-linked immunosorbent assay
- BCA:
-
Bicinchoninic acid
- SDS:
-
Sodium dodecyl sulfate
- PVDF:
-
Polyvinylidene fluoride
- ECL:
-
Enhanced chemiluminescence
- JAM:
-
Junctional adhesion molecules
- ALI:
-
Acute lung injury
- PI3K:
-
Phosphatidylinositol 3-kinase
- ROS:
-
Reactive oxygen species
References
Forsmark CE, et al. Acute pancreatitis. N Engl J Med. 2016;20:1972–1981.
Lankisch PG, et al. Acute pancreatitis. Lancet. 2015;9988:85–96.
Gukovskaya AS, et al. Autophagy and pancreatitis. Am J Physiol Gastrointest Liver Physiol. 2012;9:G993–G1003.
Mareninova OA, et al. Impaired autophagic flux mediates acinar cell vacuole formation and trypsinogen activation in rodent models of acute pancreatitis. J Clin Invest. 2009;11:3340–3355.
Yang S, et al. Autophagy regulation by the nuclear factor kappaB signal axis in acute pancreatitis. Pancreas. 2012;3:367–373.
Wu D, et al. Reverse-migrated neutrophils regulated by JAM-C are involved in acute pancreatitis-associated lung injury. Sci Rep. 2016;6:20545.
Kui B, et al. New insights into the methodology of l-arginine-induced acute pancreatitis. PLoS One. 2015;2:e0117588.
Schmidt J, et al. A better model of acute pancreatitis for evaluating therapy. Ann Surg. 1992;1:44–56.
Wildi S, et al. Suppression of transforming growth factor beta signalling aborts caerulein induced pancreatitis and eliminates restricted stimulation at high caerulein concentrations. Gut. 2007;5:685–692.
He Z, et al. Intravenous hMSCs ameliorate acute pancreatitis in mice via secretion of tumor necrosis factor-alpha stimulated gene/protein. Sci Rep. 2016;6:38438.
Staubli SM, et al. Laboratory markers predicting severity of acute pancreatitis. Crit Rev Clin Lab Sci. 2015;6:273–283.
Minkov GA, et al. Pathophysiological mechanisms of acute pancreatitis define inflammatory markers of clinical prognosis. Pancreas. 2015;5:713–717.
Dai SR, et al. Serum interleukin 17 as an early prognostic biomarker of severe acute pancreatitis receiving continuous blood purification. Int J Artif Organs. 2015;4:192–198.
Denham W, et al. Gene targeting demonstrates additive detrimental effects of interleukin 1 and tumor necrosis factor during pancreatitis. Gastroenterology. 1997;5:1741–1746.
Petrov MS, et al. Organ failure and infection of pancreatic necrosis as determinants of mortality in patients with acute pancreatitis. Gastroenterology. 2010;3:813–820.
Banks PA, et al. Classification of acute pancreatitis–2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;1:102–111.
Deretic V, et al. Autophagy in infection, inflammation and immunity. Nat Rev Immunol. 2013;10:722–737.
Mizushima N, et al. The role of Atg proteins in autophagosome formation. Annu Rev Cell Dev Biol. 2011;27:107–132.
Ichimura Y, et al. Pathophysiological role of autophagy: lesson from autophagy-deficient mouse models. Exp Anim. 2011;4:329–345.
Johansen T, et al. Selective autophagy mediated by autophagic adapter proteins. Autophagy. 2011;3:279–296.
Zeng X, et al. Mammalian target of rapamycin and S6 kinase 1 positively regulate 6-thioguanine-induced autophagy. Cancer Res. 2008;7:2384–2390.
Dai ZJ, et al. Antitumor effects of rapamycin in pancreatic cancer cells by inducing apoptosis and autophagy. Int J Mol Sci. 2012;1:273–285.
Katayama M, et al. DNA damaging agent-induced autophagy produces a cytoprotective adenosine triphosphate surge in malignant glioma cells. Cell Death Differ. 2007;3:548–558.
Gukovsky I, et al. Impaired autophagy underlies key pathological responses of acute pancreatitis. Autophagy. 2010;3:428–429.
Xiao J, et al. Spautin-1 ameliorates acute pancreatitis via inhibiting impaired autophagy and alleviating calcium overload. Mol Med. 2016;22:643.
Ji L, et al. Hydrogen sulphide exacerbates acute pancreatitis by over-activating autophagy via AMPK/mTOR pathway. J Cell Mol Med. 2016;12:2349–2361.
DiDonato JA, et al. NF-kappaB and the link between inflammation and cancer. Immunol Rev. 2012;1:379–400.
Jakkampudi A, et al. NF-kappaB in acute pancreatitis: Mechanisms and therapeutic potential. Pancreatology. 2016;4:477–488.
Rakonczay ZJ, et al. The role of NF-kappaB activation in the pathogenesis of acute pancreatitis. Gut. 2008;2:259–267.
Wang, X. et al. Acanthopanax versus 3-Methyladenine ameliorates sodium taurocholate-induced severe acute pancreatitis by inhibiting the autophagic pathway in rats. Mediators Inflamm, 8369704 (2016).
Zhang XH, et al. Caspase-1 inhibition alleviates acute renal injury in rats with severe acute pancreatitis. World J Gastroenterol. 2014;30:10457–10463.
Liu M, et al. Caspase inhibitor zVAD-fmk protects against acute pancreatitis-associated lung injury via inhibiting inflammation and apoptosis. Pancreatology. 2016;5:733–738.
Liu F, et al. MiR-155 alleviates septic lung injury by inducing autophagy via inhibition of transforming growth factor-beta-activated binding protein 2. Shock. 2017;48:61.
Acknowledgments
This work was supported in part by the National Natural Science Foundation of China (No: 81460130), the Science and Technology Plan Grant (No. 20165092) from the Health Department of Jiangxi Province, China, and the Science and Technology Plan Grant (Key project) (No. GJJ160024) from the Education Department of Jiangxi Province, China.
Author information
Authors and Affiliations
Contributions
LX and JW conceived and designed the experiments. DW, GH, YZ, YF, JW, TM, JN and GY performed the experiments. GH, XW, DW and RW analyzed the data. GH, XW and RW contributed reagents/materials/analysis tools. JW wrote the paper.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wan, J., Chen, J., Wu, D. et al. Regulation of Autophagy Affects the Prognosis of Mice with Severe Acute Pancreatitis. Dig Dis Sci 63, 2639–2650 (2018). https://doi.org/10.1007/s10620-018-5053-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-018-5053-0