Abstract
Background
Hovhannisyan et al. first showed evidence of plasticity between Treg and Th17 in the inflamed intestine of Crohn’s disease (CD) patients. Our previous report suggests that the inflammatory cytokine milieu generates IL-17+ Foxp3+ CD4+ T lymphocytes which is a crossover population converting Treg subset to Th17 in the peripheral blood of IBD patients. This is considered as an evidence of Treg/Th17 plasticity.
Aim
The aim of this study was to characterize a variety of helper T cell crossover population, not limited to IL-17+ Foxp3+ CD4+ T lymphocytes, in the lamina propria (LP) of IBD patients.
Methods
Fresh colonoscopic biopsies were obtained from patients with CD (n = 50) and ulcerative colitis (UC, n = 32) and from healthy controls (HC, n = 25). LP mononuclear cells were assessed for intracellular cytokines and transcription factors such as IFNγ, IL-13, IL-17, IL-22, T-bet, Gata-3, RORγt, and Foxp3 using multicolor flow cytometry to detect subsets of LP CD4+ T lymphocytes.
Results
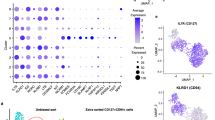
Patients with IBD demonstrated increased crossover populations in IL-17+ Foxp3+, T-bet+ Foxp3+, Gata3+ Foxp3+, RORγt+ Foxp3+ populations compared to HC. There was an inverse correlation of Harvey–Bradshaw index with Gata3+ Foxp3+ population in CD patients, while IL-13+ Foxp3+ population was directly correlated with Mayo clinical scores in UC patients. Furthermore, total IL-22 expressing cells as well as Th22 and IL-22+ Th1 populations were decreased in UC compared to CD and HC.
Conclusion
IBD patients exhibit the increased crossover populations in LP Treg cells toward Th2 and Th17 compared to HC. The prevalence of Treg/Th2 crossover populations is associated with clinical disease score of IBD.





Similar content being viewed by others
Abbreviations
- CD:
-
Crohn’s disease
- HC:
-
Healthy control
- IBD:
-
Inflammatory bowel diseases
- LP:
-
Lamina propria
- Th1:
-
T helper type 1 cells
- Th2:
-
T helper type 2 cells
- Th17:
-
T helper type 17 cells
- Th22:
-
T helper type 22 cells
- TNF:
-
Tumor necrosis factor
- Treg:
-
Regulatory T helper cells
- UC:
-
Ulcerative colitis
References
Funderburg NT, Stubblefield Park SR, Sung HC, et al. Circulating CD4(+) and CD8(+) T cells are activated in inflammatory bowel disease and are associated with plasma markers of inflammation. Immunology. 2013;140:87–97.
Baumgart DC, Sandborn WJ. Crohn’s disease. Lancet. 2012;380:1590–1605.
Ordas I, Eckmann L, Talamini M, et al. Ulcerative colitis. Lancet. 2012;380:1606–1619.
Bai A, Lu N, Guo Y, et al. All-trans retinoic acid down-regulates inflammatory responses by shifting the Treg/Th17 profile in human ulcerative and murine colitis. J Leukoc Biol. 2009;86:959–969.
Malmhall C, Bossios A, Radinger M, et al. Immunophenotyping of circulating T helper cells argues for multiple functions and plasticity of T cells in vivo in humans-possible role in asthma. PLoS ONE. 2012;7:e40012.
Li L, Boussiotis VA. The role of IL-17-producing Foxp3+ CD4+ T cells in inflammatory bowel disease and colon cancer. Clin Immunol. 2013;148:246–253.
Li J, Ueno A, Fort Gasia M, et al. Distinctive Th17 lymphocyte plasticity in intestinal lamina propria of IBD patients compared with healthy controls. Gastoroenterology. 2015;148:S-323.
Jiang W, Su J, Zhang X, et al. Elevated levels of Th17 cells and Th17-related cytokines are associated with disease activity in patients with inflammatory bowel disease. Inflamm Res. 2014;63:943–950.
Fujino S, Andoh A, Bamba S, et al. Increased expression of interleukin 17 in inflammatory bowel disease. Gut. 2003;52:65–70.
Rovedatti L, Kudo T, Biancheri P, et al. Differential regulation of interleukin 17 and interferon gamma production in inflammatory bowel disease. Gut. 2009;58:1629–1636.
Lord JD, Valliant-Saunders K, Hahn H, et al. Paradoxically increased Foxp3+ T cells in IBD do not preferentially express the isoform of Foxp3 lacking exon 2. Dig Dis Sci. 2012;57:2846–2855. doi:10.1007/s10620-012-2292-3.
Ban H, Andoh A, Shioya M, et al. Increased number of FoxP3+ CD4+ regulatory T cells in inflammatory bowel disease. Mol Med Rep. 2008;1:647–650.
Annunziato F, Romagnani S. Do studies in humans better depict Th17 cells? Blood. 2009;114:2213–2219.
Eastaff-Leung N, Mabarrack N, Barbour A, et al. Foxp3+ regulatory T cells, Th17 effector cells, and cytokine environment in inflammatory bowel disease. J Clin Immunol. 2010;30:80–89.
Kobayashi T, Okamoto S, Hisamatsu T, et al. IL23 differentially regulates the Th1/Th17 balance in ulcerative colitis and Crohn’s disease. Gut. 2008;57:1682–1689.
Himmel ME, Yao Y, Orban PC, et al. Regulatory T-cell therapy for inflammatory bowel disease: more questions than answers. Immunology. 2012;136:115–122.
Ueno A, Jijon H, Chan R, et al. Increased prevalence of circulating novel IL-17 secreting Foxp3 expressing CD4+ T cells and defective suppressive function of circulating Foxp3+ regulatory cells support plasticity between Th17 and regulatory T cells in inflammatory bowel disease patients. Inflamm Bowel Dis. 2013;19:2522–2534.
Hovhannisyan Z, Treatman J, Littman DR, et al. Characterization of interleukin-17-producing regulatory T cells in inflamed intestinal mucosa from patients with inflammatory bowel diseases. Gastroenterology. 2011;140:957–965.
Trifari S, Kaplan CD, Tran EH, et al. Identification of a human helper T cell population that has abundant production of interleukin 22 and is distinct from T(H)-17, T(H)1 and T(H)2 cells. Nat Immunol. 2009;10:864–871.
Eyerich S, Eyerich K, Pennino D, et al. Th22 cells represent a distinct human T cell subset involved in epidermal immunity and remodeling. J Clin Invest. 2009;119:3573–3585.
Duhen T, Geiger R, Jarrossay D, et al. Production of interleukin 22 but not interleukin 17 by a subset of human skin-homing memory T cells. Nat Immunol. 2009;10:857–863.
Immunology Leslie M. Cross-over immune cells blur the boundaries. Science. 2012;336:1228–1229.
Hueber W, Sands BE, Lewitzky S, et al. Secukinumab, a human anti-IL-17A monoclonal antibody, for moderate to severe Crohn’s disease: unexpected results of a randomised, double-blind placebo-controlled trial. Gut. 2012;61:1693–1700.
Nikolaus S, Schreiber S. Diagnostics of inflammatory bowel disease. Gastroenterology. 2007;133:1670–1689.
Colombel JF, Rutgeerts P, Reinisch W, et al. Early mucosal healing with infliximab is associated with improved long-term clinical outcomes in ulcerative colitis. Gastroenterology. 2011;141:1194–1201.
Daperno M, D’Haens G, Van Assche G, et al. Development and validation of a new, simplified endoscopic activity score for Crohn’s disease: the SES-CD. Gastrointest Endosc. 2004;60:505–512.
Harvey RF, Bradshaw JM. A simple index of Crohn’s-disease activity. Lancet. 1980;1:514.
Yurchenko E, Shio MT, Huang TC, et al. Inflammation-driven reprogramming of CD4+ Foxp3+ regulatory T cells into pathogenic Th1/Th17 T effectors is abrogated by mTOR inhibition in vivo. PLoS ONE. 2012;7:e35572.
Leung JM, Davenport M, Wolff MJ, et al. IL-22-producing CD4+ cells are depleted in actively inflamed colitis tissue. Mucosal Immunol. 2014;7:124–133.
Davenport M, Poles J, Leung JM, et al. Metabolic alterations to the mucosal microbiota in inflammatory bowel disease. Inflamm Bowel Dis. 2014;20:723–731.
Ylikoski E, Lund R, Kylaniemi M, et al. IL-12 up-regulates T-bet independently of IFN-gamma in human CD4+ T cells. Eur J Immunol. 2005;35:3297–3306.
Koch MA, Thomas KR, Perdue NR, et al. T-bet(+) Treg cells undergo abortive Th1 cell differentiation due to impaired expression of IL-12 receptor beta2. Immunity. 2012;37:501–510.
Mann ER, Bernardo D, English NR, et al. Compartment-specific immunity in the human gut: properties and functions of dendritic cells in the colon versus the ileum. Gut. 2016;65:256–270.
Belkaid Y, Hand TW. Role of the microbiota in immunity and inflammation. Cell. 2014;157:121–141.
Donaldson GP, Lee SM, Mazmanian SK. Gut biogeography of the bacterial microbiota. Nat Rev Microbiol. 2016;14:20–32.
Esposito M, Ruffini F, Bergami A, et al. IL-17- and IFN-gamma-secreting Foxp3+ T cells infiltrate the target tissue in experimental autoimmunity. J Immunol. 2010;185:7467–7473.
Acknowledgments
The authors thank to the Alberta IBD consortium for financial support; Ms. Gurmeet Bindra in the inflammatory intestinal tissue bank (IITB) and the staff in IBD clinic for assistance with research sample collection; Dr. Karen Poon (Nicole Perkins Microbe Core Facility, Calvin, Phoebe and Joan Snyder Institute for Chronic Disease) and Mr. Ronald Chan for assistance with data acquisition; Drs. Tie Wang, Donald Buie, Anthony MacLean, and Iwona Auer-Grzesiak for assistance with optimizing study design; and all patients and healthy individuals who participated into this study.
Author information
Authors and Affiliations
Contributions
AU, JLi, JLu, XG, SG were involved in study concept; JLi, AU, MFG were involved in data acquisition and analysis; MFG, MI, GK, PB, RP, SG were involved in patient recruitment; JQ, HB, SG were involved in project supervision; AU, HJ, MI, SG, HB, JLi were involved in manuscript preparation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Ji Li and Aito Ueno share the first authorship.
Xianyong Gui and Subrata Ghosh share the senior authorship.
Study Sponsor Alberta IBD Consortium/Alberta Innovates—Health Solutions.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
Flow cytometry plots. The gating strategy is shown in (A). First, the acquired data were gated lymphocytes in FSC-A vs. SSC-A display, followed by doublets discrimination in FSC-A vs. FSC-H display, and then live/dead discrimination. Live CD4+ T cells are further analyzed. Representative data of CD, UC, and HC are shown in B-D. The upper panels display Foxp3 (X-axis) vs. IFNγ (Y-axis) (B), the middle panels Foxp3 (X-axis) vs. IL-17 (Y-axis) display (C) and the lower panels IL-17 (X-axis) versus IFNγ (Y-axis) (D). (PPTX 113 kb)
Supplementary Figure 2
Prevalence of Th subsets in lamina propria mononuclear cells. The prevalence of expressing a Th1 signature cytokine, IFNγ (A), a Th1 transcription factor, T-bet (B), a Th2 signature cytokine, IL-13 (C), a Th2 transcription factor, Gata3 (D), a Th17 signature cytokine, IL-17A (E), a Th17 transcription factor RORγt (F), a Treg transcription factor Foxp3 (G), and a combination of CD25 and Foxp3 (H) in CD4 + T cells are shown in this figure. * p < 0.05, **p < 0.01, *** p < 0.001, **** P < 0.0001. (PPTX 99 kb)
Supplementary Figure 3
Concordance between endoscopic scores (SES-CD scores) and Foxp3+ crossover populations in lamina propria mononuclear cells of CD patients. The prevalence of IFNγ+ Foxp3+ (A), IL-13+ Foxp3+ (B), IL-17+ Foxp3+ (C), T-bet+ Foxp3+ (D), Gata3+ Foxp3+ (E), RORγt+ Foxp3+ (F), and IFNγ+ IL-17+ (G) is shown in this figure. (PPTX 146 kb)
Supplementary Figure 4
Concordance between Mayo endoscopic subscores and CD4+ T cell crossover populations in lamina propria mononuclear cells of UC patients. The prevalence of IFNγ+ Foxp3+ (A), IL-13+ Foxp3+ (B), IL-17+ Foxp3+ (C), T-bet+ Foxp3+ (D), Gata3+ Foxp3+ (E), RORγt+ Foxp3+ (F), and IFNγ+ IL-17+ (G) is shown in this figure. * p < 0.05 (PPTX 102 kb)
Supplementary Figure 5
Association between IL-22+ T helper cell subsets and clinical disease activity scores evaluated by Harvey–Bradshaw index in CD (A) and Mayo clinical index scores in UC cohort (B). Total IL-22+, IL-22+ IL-17+ (IL-22+ Th17), IL-22+ IFN+ (IL-22+ Th1) and IL-22+ IL-17-IFNγ-(Th22) cells are shown in this figure. (PPTX 81 kb)
Supplementary Figure 6
Association between IL-22+ T helper cell subsets and endoscopic disease activity evaluated by SES-CD scores in CD cohort (A) and Mayo endoscopic subscores in UC cohort (B). Total IL-22+, IL-22+ IL-17+ (IL-22+ Th17), IL-22+ IFN+ (IL-22+ Th1) and IL-22+ IL-17-IFNγ-(Th22) cells are shown in this figure. (PPTX 80 kb)
Rights and permissions
About this article
Cite this article
Li, J., Ueno, A., Iacucci, M. et al. Crossover Subsets of CD4+ T Lymphocytes in the Intestinal Lamina Propria of Patients with Crohn’s Disease and Ulcerative Colitis. Dig Dis Sci 62, 2357–2368 (2017). https://doi.org/10.1007/s10620-017-4596-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-017-4596-9