Abstract
Background
BCCIP was originally identified as a BRCA2 interacting protein in humans and Ustilago maydis. It had low expression in some human cancer tissues. However, recent research indicated that many caretaker genes are also necessary for cell viability and their expression could contribute to tumor progression.
Aim
To characterize whether BCCIP is a caretaker gene in esophageal squamous cell carcinoma (ESCC).
Methods
Western blotting and immunohistochemistry were used to measure the expression of BCCIP β. In vitro studies were used to verify the effects of BCCIP β in Eca109 cells.
Results
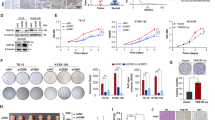
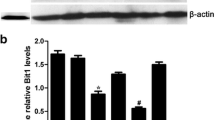
Expression of BCCIP β was notably higher in tumor tissues of ESCC and Eca 109 cells. Meanwhile, the immunohistochemistry stain revealed that BCCIP β was positively correlated with clinical pathologic variables such as tumor size and tumor grade, as well as Ki-67, and prompted poor prognosis. In vitro studies such as starvation and refeeding assay along with BCCIP β-shRNA transfection assay demonstrated that BCCIP β expression promoted proliferation of ESCC cells. In addition, BCCIP β downregulation by silencing RNA significantly decreased the rate of colony formation, alleviated cellular apoptosis and increased the chemosensitivity of cisplatin.
Conclusions
This research first put forward that BCCIP β is an oncogene in human ESCC and contributes to the poor outcome of the deadly disease.





Similar content being viewed by others
References
Rustgi AK, El-Serag HB. Esophageal carcinoma. N Engl J Med. 2014;371:2499–2509.
Orringer MB, Marshall B, Iannettoni MD. Transhiatal esophagectomy: clinical experience and refinements. Ann Surg. 1999;230:392–400. (discussion 393–400).
Liu J, Yuan Y, Huan J, Shen Z. Inhibition of breast and brain cancer cell growth by BCCIPalpha, an evolutionarily conserved nuclear protein that interacts with BRCA2. Oncogene. 2001;20:336–345.
Ono T, Kitaura H, Ugai H, et al. TOK-1, a novel p21Cip1-binding protein that cooperativ-ely enhances p21-dependent inhibitory activity toward CDK2 kinase. J Biol Chem. 2000;275:31145–31154.
Lu H, Guo X, Meng X, et al. The BRCA2-interacting protein BCCIP functions in RAD51 and BRCA2 focus formation and homologous recombinational repair. Mol Cell Biol. 2005;25:1949–1957.
Meng X, Liu J, Shen Z. Genomic structure of the human BCCIP gene and its expression in cancer. Gene. 2003;302:139–146.
Fan J, Wray J, Meng X, Shen Z. BCCIP is required for the nuclear localization of the p21 protein. Cell Cycle. 2009;8:3019–3024.
Liu X, Cao L, Ni J, et al. Differential BCCIP gene expression in primary human ovarian cancer, renal cell carcinoma and colorectal cancer tissues. Int J Oncol. 2013;43:1925–1934.
Roversi G, Pfundt R, Moroni RF, et al. Identification of novel genomic markers related to progression to glioblastoma through genomic profiling of 25 primary glioma cell lines. Oncogene. 2006;25:1571–1583.
Meng X, Liu J, Shen Z. Inhibition of G1 to S cell cycle progression by BCCIP beta. Cell Cycle. 2004;3:343–348.
Meng X, Lu H, Shen Z. BCCIP functions through p53 to regulate the expression of p21Waf1/Cip1. Cell Cycle. 2004;3:1457–1462.
Lu H, Yue J, Meng X, Nickoloff JA, Shen Z. BCCIP regulates homologous recombination by distinct domains and suppresses spontaneous DNA damage. Nucleic Acids Res. 2007;35:7160–7170.
Meng X, Fan J, Shen Z. Roles of BCCIP in chromosome stability and cytokinesis. Oncogene. 2007;26:6253–6260.
Lu H, Huang YY, Mehrotra S, et al. Essential roles of BCCIP in mouse embryonic development and structural stability of chromosomes. PLoS Genet. 2011;7:e1002291.
Huang YY, Dai L, Gaines D, et al. BCCIP suppresses tumor initiation but is required for tumor progression. Cancer Res. 2013;73:7122–7133.
Wang Y, Yang S, Ni Q, et al. Overexpression of forkhead box J2 can decrease the migration of breast cancer cells. J Cell Biochem. 2012;113:2729–2737.
Zhang J, Yang X, Wang Y, et al. Low expression of cyclinH and cyclin-dependent kinase 7 can decrease the proliferation of human esophageal squamous cell carcinoma. Dig Dis Sci. 2013;58:2028–2037.
Wang Y, Wang Y, Xiang J, et al. Knockdown of CRM1 inhibits the nuclear export of p27(Kip1) phosphorylated at serine 10 and plays a role in the pathogenesis of epithelial ovarian cancer. Cancer Lett. 2014;343:6–13.
Alvarez V, Vinas L, Gallego-Sanchez A, Andres S, Sacristan MP, Bueno A. Orderly progression through S-phase requires dynamic ubiquitylation and deubiquitylation of PCNA. Sci Rep. 2016;6:25513.
Liu J, Lu H, Ohgaki H, Merlo A, Shen Z. Alterations of BCCIP, a BRCA2 interacting protein, in astrocytomas. BMC Cancer. 2009;9:268.
Rewari A, Lu H, Parikh R, Yang Q, Shen Z, Haffty BG. BCCIP as a prognostic marker for radiotherapy of laryngeal cancer. Radiother Oncol. 2009;90:183–188.
Meng X, Yue J, Liu Z, Shen Z. Abrogation of the transactivation activity of p53 by BCCIP down-regulation. J Biol Chem. 2007;282:1570–1576.
Huang YY, Lu H, Liu S, Droz-Rosario R, Shen Z. Requirement of mouse BCCIP for neural development and progenitor proliferation. PLoS ONE. 2012;7:e30638.
Mao N, Zhou Q, Kojic M, Perez-Martin J, Holloman WK. Ortholog of BRCA2-interacting protein BCCIP controls morphogenetic responses during DNA replication stress in Ustilago maydis. DNA Repair. 2007;6:1651–1660.
Audhya A, Emr SD. Regulation of PI4,5P2 synthesis by nuclear-cytoplasmic shuttling of the Mss4 lipid kinase. EMBO J. 2003;22:4223–4236.
Wyler E, Wandrey F, Badertscher L, Montellese C, Alper D, Kutay U. The beta-isoform of the BRCA2 and CDKN1A(p21)-interacting protein (BCCIP) stabilizes nuclear RPL23/uL14. FEBS Lett. 2014;588:3685–3691.
Kinzler KW, Vogelstein B. Cancer-susceptibility genes. Gatekeepers and caretakers. Nature. 1997;386:761–763.
Zheng Y, Stamminger T, Hearing P. E2F/Rb family proteins mediate interferon induced repression of adenovirus immediate early transcription to promote persistent viral infection. PLoS Pathog. 2016;12:e1005415.
Acknowledgements
This work was supported by Grants from the National Natural Science Foundation of China (Nos. 81171140, 81472272).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Lingling Chen and Sujie Ni have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
BCCIP expression in ESCC tissues. Seven paired samples of ESCC tumor tissues (T) and adjacent non-tumor tissues (N). This figure with the primary data suggests that BCCIP α expression is not as evident as in BCCIP β and is even silenced in tumor tissues (TIFF 1157 kb)
Rights and permissions
About this article
Cite this article
Chen, L., Ni, S., Li, M. et al. High Expression of BCCIP β Can Promote Proliferation of Esophageal Squamous Cell Carcinoma. Dig Dis Sci 62, 387–395 (2017). https://doi.org/10.1007/s10620-016-4382-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-016-4382-0