Abstract
Background
Carcinoembryonic antigen (CEA) can be used to screen for biliary tract cancer in patients with primary sclerosing cholangitis (PSC).
Aim
To study the influence of benign dominant strictures (DS), superimposed bacterial cholangitis (SBC), smoking status, and inflammatory bowel disease on CEA serum levels.
Methods
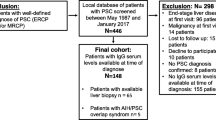
A retrospective analysis of CEA values in cancer-free PSC patients was performed. We included the maximal CEA value obtained during follow-up and information on the presence of DS and SBC at that time, and we analyzed the CEA values in the presence and absence of DS and SBC. Results are reported as medians with the interquartile range (IQR).
Results
The median maximal CEA level, which was 1.8 ng/mL (IQR 1.2–2.9) in the final 270 PSC patients included in the study, was not influenced by the presence of either DS or SBC (P = 0.320). Moreover, in 49 patients, the first CEA value available at the time of DS (1.5 ng/mL; IQR 1.2–2.1) and that at a time without DS (1.6 ng/mL; IQR 1.1–2.3) did not differ significantly (P = 0.397). Lastly, in 24 patients, the median CEA values at a time without SBC (1.8 ng/mL; IQR 1.2–2.5) and at the time of SBC (1.8 ng/mL; IQR 1.0–3.0) were comparable (P = 0.305). Smoking did not influence CEA-based cancer screening.
Conclusions
Serum CEA level is not influenced by the presence of DS or SBC and might therefore serve as a favorable parameter for improving cancer screening in PSC patients.


Similar content being viewed by others
References
Chapman R, Fevery J, Kalloo A, et al. Diagnosis and management of primary sclerosing cholangitis. Hepatology. 2010;51:660–678.
European Association for the Study of the Liver. EASL clinical practice guidelines: management of cholestatic liver diseases. J Hepatol. 2009;51:237–267.
Bergquist A, Ekbom A, Olsson R, et al. Hepatic and extrahepatic malignancies in primary sclerosing cholangitis. J Hepatol. 2002;36:321–327.
Claessen MM, Vleggaar FP, Tytgat KM, Siersema PD, van Buuren HR. High lifetime risk of cancer in primary sclerosing cholangitis. J Hepatol. 2009;50:158–164.
Boonstra K, Weersma RK, van Erpecum KJ, et al. Population-based epidemiology, malignancy risk, and outcome of primary sclerosing cholangitis. Hepatology. 2013;58:2045–2055.
Charatcharoenwitthaya P, Enders FB, Halling KC, Lindor KD. Utility of serum tumor markers, imaging, and biliary cytology for detecting cholangiocarcinoma in primary sclerosing cholangitis. Hepatology. 2008;48:1106–1117.
Wannhoff A, Rupp C, Friedrich K, et al. Inflammation but not biliary obstruction is associated with carbohydrate antigen 19-9 levels in patients with primary sclerosing cholangitis. Clin Gastroenterol Hepatol. 2015;13:2372–2379.
Ramage JK, Donaghy A, Farrant JM, Iorns R, Williams R. Serum tumor markers for the diagnosis of cholangiocarcinoma in primary sclerosing cholangitis. Gastroenterology. 1995;108:865–869.
Wannhoff A, Folseraas T, Brune M, et al. A common genetic variant of fucosyltransferase 2 correlates with serum carcinoembryonic antigen levels and affects cancer screening in patients with primary sclerosing cholangitis. United Eur Gastroenterol J. 2016;4:84–91.
Wannhoff A, Hov JR, Folseraas T, et al. FUT2 and FUT3 genotype determines CA19-9 cut-off values for detection of cholangiocarcinoma in patients with primary sclerosing cholangitis. J Hepatol. 2013;59:1278–1284.
Alexander JC, Silverman NA, Chretien PB. Effect of age and cigarette smoking on carcinoembryonic antigen levels. JAMA. 1976;235:1975–1979.
Rule AH, Goleski-Reilly C, Sachar DB, Vandevoorde J, Janowitz HD. Circulating carcinoembryonic antigen (CEA): relationship to clinical status of patients with inflammatory bowel disease. Gut. 1973;14:880–884.
Stiehl A, Rudolph G, Kloters-Plachky P, Sauer P, Walker S. Development of dominant bile duct stenoses in patients with primary sclerosing cholangitis treated with ursodeoxycholic acid: outcome after endoscopic treatment. J Hepatol. 2002;36:151–156.
Levy C, Lymp J, Angulo P, Gores GJ, Larusso N, Lindor KD. The value of serum CA 19-9 in predicting cholangiocarcinomas in patients with primary sclerosing cholangitis. Dig Dis Sci. 2005;50:1734–1740.
Chalasani N, Baluyut A, Ismail A, et al. Cholangiocarcinoma in patients with primary sclerosing cholangitis: a multicenter case-control study. Hepatology. 2000;31:7–11.
Siqueira E, Schoen RE, Silverman W, et al. Detecting cholangiocarcinoma in patients with primary sclerosing cholangitis. Gastrointest Endosc. 2002;56:40–47.
Bjornsson E, Kilander A, Olsson R. CA 19-9 and CEA are unreliable markers for cholangiocarcinoma in patients with primary sclerosing cholangitis. Liver. 1999;19:501–508.
Rupp C, Friedrich K, Folseraas T, et al. Fut2 genotype is a risk factor for dominant stenosis and biliary candida infections in primary sclerosing cholangitis Aliment. Pharmacol Ther. 2014;39:873–882.
Gotthardt D, Chahoud F, Sauer P. Primary sclerosing cholangitis: diagnostic and therapeutic problems. Dig Dis. 2011;29:41–45.
Acknowledgements
We thank Petra Klöters-Plachky and Yvonne Leopold for excellent technical assistance.
Funding
AW was funded by a grant from the Medical Faculty of the University of Heidelberg. DNG was supported by a Grant from Deutsche Forschungsgemeinschaft (DFG).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical approval
All procedures performed were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wannhoff, A., Rupp, C., Friedrich, K. et al. Carcinoembryonic Antigen Level in Primary Sclerosing Cholangitis Is Not Influenced by Dominant Strictures or Bacterial Cholangitis. Dig Dis Sci 62, 510–516 (2017). https://doi.org/10.1007/s10620-016-4370-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-016-4370-4