Abstract
Background
Vulnerable populations are disproportionately affected by hepatitis C virus (HCV) infection and experience high rates of health disparity. There are no data on real-world experience with highly efficacious direct-acting anti-HCV treatment in this population.
Aims
We aimed to evaluate the real-world experience with sofosbuvir-based regimens among a vulnerable HCV-infected population.
Methods
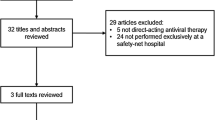
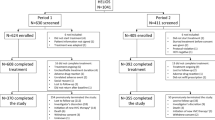
HCV treatment response was assessed among 204 patients who completed 12–24 weeks of sofosbuvir-based regimens (in combination with pegylated interferon and ribavirin, simeprevir, ledipasvir, or daclatasvir) at the San Francisco safety-net healthcare system liver specialty clinic between January 2014 and December 2015. Virologic response during therapy was assessed at weeks 4 and 8, end of therapy, and 12-week treatment discontinuation (SVR 12).
Results
Patient characteristics were median age 58 years, 60 % male, 42 % Caucasian (21 % black, 19 % Hispanic), 72 % had genotype 1 (23 % genotype 2 or 3), and the median baseline log10 HCV viral load was 6.1 IU/ml and alanine transaminase 63 U/l. Cirrhosis was present in 36 % (of whom 40 % were decompensated), and 18 % were HCV treatment-experienced. Overall, SVR 12 was achieved in 97 % (99 % genotype 1, 100 % genotype 2, 84 % genotype 3). Five of six (83 %) patients who relapsed had decompensated cirrhosis, and 67 % were also non-adherent to therapy. On-treatment virologic response did not impact SVR.
Conclusions
High rates of sustained virologic response can be achieved in safety-net HCV-infected patients. Access to DAA-based regimens is critical to addressing HCV-related health disparity in this at-risk population.

Similar content being viewed by others
References
Centers for Disease Control. Viral hepatitis. http://www.cdc.gov/hepatitis/hcv/statisticshcv.htm. Accessed December 6, 2015.
Centers for Disease Control. Notes from the field: risk factors for hepatitis C virus infections among young adults—Massachusetts, 2010. MMWR. 2011;60:1457–1458.
Davis GL, Alter MJ, El-Serag H, Poynard T, Jennings LW. Aging of hepatitis C virus infected persons in the United States: a multiple cohort model of HCV prevalence and disease progression. Gastroenterology. 2010;138:513–521.
Razavi H, ElKhoury AC, Elbasha E, et al. Chronic hepatitis C disease burden and cost in the United States. Hepatology. 2013;57:2164–2170.
Saab S, Jackson C, Nieto J, Francois F. Hepatitis C in African Americans. Am J Gastroenterol. 2014;109:1576–1584.
Singal AG, Yopp A, Skinner CS, Packer M, Lee WM, Tiro JA. Utilization of hepatocellular carcinoma surveillance among American patients: a systematic review. JGIM. 2012;27:861–867.
Nguyen GC, Segev DL, Thuluvath PJ. Racial disparities in the management of hospitalized patients with cirrhosis and complications of portal hypertension: a national study. Hepatology. 2007;45:1282–1289.
Quillin RC III, Wilson GC, Wima K, et al. Neighborhood level effects of socioeconomic status on liver transplant selection and recipient survival. Clin Gastroenterol Hepatol. 2014;12:1934–1941.
Moyer VA. U.S. Preventive Services Task Force. Screening for hepatitis C virus infection in adults: a U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2013;159:349–357.
Bumenthal D. Health care coverage under the Affordable Care Act—a progress report. N Engl J Med. 2014;17:275–281.
Centers for Disease Control and Prevention. CDC health disparities and inequalities report—United States, 2013. MMWR. 2013;62:1–186. http://www.cdc.gov/mmwr/pdf/other/su6203.pdf.
Guerra V, McMahon S. Charity care organizations as navigators: considerations for guiding consumers toward the best coverage options. Center for Health Care Strategies. February 2014. http://www.chcs.org/usr_doc/Charity_Care_as_Navigators_02414.pdf.
The U.S. Department of Housing and Urban Development. The 2013 Annual Homeless Assessment Report (AHAR) to congress. November 2013. http://www.onecpd.info/resources/documents/AHAR‐2013‐Part1.pdf.
Substance Abuse and Mental Health Services Administration. Current statistics on the prevalence and characteristics of people experiencing homelessness in the United States. July 2011. http://homeless.samhsa.gov/ResourceFiles/hrc_factsheet.pdf.
Sood S, Wong D, Holmes A, Everall I, Saling M, Nicoll A. Depression in a real world population of hepatitis C patients. J Gastroenterol Pancreatol Liver Disord. 2014;30:1–3.
Surjadi M, Torreullas C, Ayala C, Yee HF Jr, Khalili M. Formal patient education improves patients’ knowledge of hepatitis C in vulnerable populations. Dig Dis Sci. 2011;56:213–219.
Lubega S, Agbim U, Surjadi M, Mahoney M, Khalili M. Formal hepatitis C education enhances HCV care coordination, expedites HCV treatment and improves antiviral response. Liver Int. 2013;33:999–1007.
Bindman AB, Chen A, Fraser JS, Yee HF Jr, Ofman D. Healthcare reform with a safety net: lessons from San Francisco. Am J Manag Care. 2009;15:747–750.
Terrault N, Zeuzem S, Di Bisceglie AM, et al. Effectiveness of ledipasvir–sofosbuvir combination in patients with hepatitis C virus infection and factors associated of sustained virologic response. Gastroenterology. 2016. doi:10.1053/j.gastro.2016.08.004.
Lawitz E, Sulkowski MS, Ghalib R, et al. Simeprevir plus sofosbuvir, with or without ribavirin, to treat chronic infection with hepatitis C virus genotype 1 in non-responders to pegylated interferon and ribavirin and treatment-naive patients: the COSMOS randomised study. The Lancet. 2014;384:1756–1765.
Lawitz E, Mangia A, Wyles D, et al. Sofosbuvir for previously untreated chronic hepatitis C infection. N Engl J Med. 2013;368:1878–1887.
Afdhal N, Zeuzem S, Kwo P, et al. for the ION-1 Investigators. Ledipasvir and Sofosbuvir for untreated HCV genotype 1 infection. N Engl J Med. 2014. doi:10.1056/NEJMoa1402454.
Backus LI, Belperio PS, Shahoumian TA, Loomis TP, Mole LA. Effectiveness of sofosbuvir-based regimens in genotype 1 and 2 hepatitis C virus infection in 4026 U.S. Veterans. Aliment Pharmacol Ther. 2015;42:559–573.
Butt AA, Yan P, Shaikh OS, Chung RT, Sherman KE, ERCHIVES study. Sofosbuvir-based regimens in clinical practice achieve SVR rates closer to clinical trials: results from ERCHIVES. Liver Int. 2016;36:651–658. doi:10.1111/liv.13036.
Wu CJ, Roytman MD, Hong LK, et al. Real-world experience with sofosbuvir-based regimens for chronic hepatitis C, including patients with factors previously associated with inferior treatment response. Hawaii J Med Public Health. 2015;74:3–7.
American Association for the Study of Liver Disease/Infectious Diseases Society of America. HCV guidance: recommendations for testing, managing, and treating hepatitis C. hcvguidelines.org. Accessed January 23, 2016.
Funding
This work was supported in part by National Institutes of Health K24AA022523 (to M.K.) and P30 DK026743 (UCSF Liver Center).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest in connection with the submitted manuscript. Dr. Khalili has research grant support from Gilead Inc and Intercept Pharmaceuticals and has participated in the advisory boards of Bristol Myers Squibb, Gilead Inc, and Intercept Pharmaceuticals.
Rights and permissions
About this article
Cite this article
Beck, K.R., Kim, N. & Khalili, M. Sofosbuvir-Containing Regimens for Chronic Hepatitis C Are Successful in the Safety-Net Population: A Real-World Experience. Dig Dis Sci 61, 3602–3608 (2016). https://doi.org/10.1007/s10620-016-4340-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-016-4340-x