Abstract
Background
Lysyl oxidase (LOX) is frequently overexpressed in a variety of malignancies and involved in tumor invasion and metastasis. Furthermore, it has been shown that LOX is closely related to vascular endothelial growth factor (VEGF).
Aims
In this study, we aimed to investigate the exact role of LOX and the correlation between LOX and VEGF in hepatocellular carcinoma (HCC).
Methods
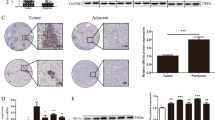
The expression levels of LOX in HCC tissue and adjacent noncancerous tissue were evaluated by quantitative reverse transcription polymerase chain reaction and immunohistochemical analysis. The effect of LOX knockdown on cell proliferation, migration, and invasion was investigated in vitro. The role of LOX in the regulation of VEGF was further characterized in HCC cells that had been treated with transforming growth factor beta (TGF-β).
Results
Our study showed that LOX was up-regulated in HCC cell lines and tissue. HCC patients with elevated expression of LOX had relatively shorter disease-free survival and overall survival. Knockdown of LOX reduced the proliferation, migration, and invasion of HCC cells. Additionally, the expression level of LOX positively correlated with that of VEGF. After treatment with TGF-β, the levels of LOX and VEGF were both up-regulated in a dose-dependent manner. In the cells treated with siRNA of LOX, levels of VEGF and phosphorylated p38 were significantly decreased and could not be up-regulated by TGF-β. Inhibition of p38 MAPK signaling abrogated TGF-β-mediated up-regulation of VGEF but did not affect LOX expression.
Conclusions
LOX appears to be a predictor of less favorable outcomes and may regulate the expression of VEGF via p38 MAPK signaling.






Similar content being viewed by others
References
Jemal A, Bray F, Center MM, et al. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
Steeg PS. Tumor metastasis: mechanistic insights and clinical challenges. Nat Med. 2006;12:895–904.
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100:57–70.
Akiri G, Sabo E, Dafni H, et al. Lysyl oxidase-related protein-1 promotes tumor fibrosis and tumor progression in vivo. Cancer Res. 2003;63:1657–1666.
Geach TJ, Dale L. Members of the lysyl oxidase family are expressed during the development of the frog Xenopus laevis. Differentiation. 2005;73:414–424.
Xu X, Wang B, Xu Y. Expression of lysyl oxidase in human osteosarcoma and its clinical significance: a tumor suppressive role of LOX in human osteosarcoma cells. Int J Oncol. 2013;43:1578–1586.
Saad FA, Torres M, Wang H, Graham L. Intracellular lysyl oxidase: effect of a specific inhibitor on nuclear mass in proliferating cells. Biochem Biophys Res Commun. 2010;396:944–949.
Nishioka T, Eustace A, West C. Lysyl oxidase: from basic science to future cancer treatment. Cell Struct Funct. 2012;37:75–80.
Osawa T, Ohga N, Akiyama K, et al. Lysyl oxidase secreted by tumour endothelial cells promotes angiogenesis and metastasis. Br J Cancer. 2013;109:2237–2247.
Liu JL, Wei W, Tang W, et al. Mechanism of lysyl oxidase (LOX) in breast cancer invasion and metastasis. Zhonghua yi xue za zhi. 2012;92:1379–1383.
Baker AM, Cox TR, Bird D, et al. The role of lysyl oxidase in SRC-dependent proliferation and metastasis of colorectal cancer. J Natl Cancer Inst. 2011;103:407–424.
Zheng Y, Wang X, Wang H, et al. Expression of the lysyl oxidase propeptide in hepatocellular carcinoma and its clinical relevance. Oncol Rep. 2014;31:1669–1676.
Kaseb AO, Hanbali A, Cotant M, et al. Vascular endothelial growth factor in the management of hepatocellular carcinoma: a review of literature. Cancer. 2009;115:4895–4906.
Dimova I, Popivanov G, Djonov V. Angiogenesis in cancer-general pathways and their therapeutic implications. J BUON. 2014;19:15–21.
Yao DF, Wu XH, Zhu Y, et al. Quantitative analysis of vascular endothelial growth factor, microvascular density and their clinicopathologic features in human hepatocellular carcinoma. Hepatobiliary Pancreat Dis Int. 2005;4:220–226.
Baker AM, Bird D, Welti JC, et al. Lysyl oxidase plays a critical role in endothelial cell stimulation to drive tumor angiogenesis. Cancer Res. 2013;73:583–594.
Dong Y, Xie X, Wang Z, et al. Increasing matrix stiffness upregulates vascular endothelial growth factor expression in hepatocellular carcinoma cells mediated by integrin beta1. Biochem Biophys Res Commun. 2014;444:427–432.
Yamazaki K, Masugi Y, Sakamoto M. Molecular pathogenesis of hepatocellular carcinoma: altering transforming growth factor-beta signaling in hepatocarcinogenesis. Dig Dis. 2011;29:284–288.
Bostrom K, Zebboudj AF, Yao Y, Lin TS, Torres A. Matrix GLA protein stimulates VEGF expression through increased transforming growth factor-beta1 activity in endothelial cells. J Biol Chem. 2004;279:52904–52913.
Taylor MA, Amin JD, Kirschmann DA, Schiemann WP. Lysyl oxidase contributes to mechanotransduction-mediated regulation of transforming growth factor-beta signaling in breast cancer cells. Neoplasia. 2011;13:406–418.
Seystahl K, Tritschler I, Szabo E, Tabatabai G, Weller M. Differential regulation of TGF-beta-induced, ALK-5-mediated VEGF release by SMAD2/3 versus SMAD1/5/8 signaling in glioblastoma. Neuro-oncology. 2014;17:254–265.
Voloshenyuk TG, Landesman ES, Khoutorova E, Hart AD, Gardner JD. Induction of cardiac fibroblast lysyl oxidase by TGF-beta1 requires PI3K/Akt, Smad3, and MAPK signaling. Cytokine. 2011;55:90–97.
Ferrari G, Terushkin V, Wolff MJ, et al. TGF-beta1 induces endothelial cell apoptosis by shifting VEGF activation of p38(MAPK) from the prosurvival p38beta to proapoptotic p38alpha. Mol Cancer Res. 2012;10:605–614.
Bruix J, Sherman M, American Association for the Study of Liver D. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020–1022.
Edmondson HA, Steiner PE. Primary carcinoma of the liver: a study of 100 cases among 48,900 necropsies. Cancer. 1954;7:462–503.
Che N, Zhao XL, Sun T, et al. The role of Twist1 in hepatocellular carcinoma angiogenesis: a clinical study. Hum Pathol. 2011;42:840–847.
Giampuzzi M, Botti G, Di Duca M, et al. Lysyl oxidase activates the transcription activity of human collagen III promoter. Possible involvement of Ku antigen. J Biol Chem. 2000;275:36341–36349.
Payne SL, Fogelgren B, Hess AR, et al. Lysyl oxidase regulates breast cancer cell migration and adhesion through a hydrogen peroxide-mediated mechanism. Cancer Res. 2005;65:11429–11436.
Wong CC, Tse AP, Huang YP, et al. Lysyl oxidase-like 2 is critical to tumor microenvironment and metastatic niche formation in hepatocellular carcinoma. Hepatology. 2014;60:1645–1658.
Wilgus ML, Borczuk AC, Stoopler M, et al. Lysyl oxidase: a lung adenocarcinoma biomarker of invasion and survival. Cancer. 2011;117:2186–2191.
Abourbih DA, Di Cesare S, Orellana ME, et al. Lysyl oxidase expression and inhibition in uveal melanoma. Melanoma Res. 2010;20:97–106.
Erler JT, Bennewith KL, Nicolau M, et al. Lysyl oxidase is essential for hypoxia-induced metastasis. Nature. 2006;440:1222–1226.
Giannelli G, Villa E, Lahn M. Transforming growth factor-beta as a therapeutic target in hepatocellular carcinoma. Cancer Res. 2014;74:1890–1894.
Wilson TJ, Nannuru KC, Futakuchi M, Singh RK. Cathepsin G-mediated enhanced TGF-beta signaling promotes angiogenesis via upregulation of VEGF and MCP-1. Cancer Lett. 2010;288:162–169.
Sidell N, Feng Y, Hao L, et al. Retinoic acid is a cofactor for translational regulation of vascular endothelial growth factor in human endometrial stromal cells. Mol Endocrinol. 2010;24:148–160.
Kagan HM, Li W. Lysyl oxidase: properties, specificity, and biological roles inside and outside of the cell. J Cell Biochem. 2003;88:660–672.
Acknowledgments
The authors wish to thank the assistance of all the staff, especially Junqi Huang, at the Department of Pathology, Guangxi Tumor Hospital. This work was supported by National Natural Science Foundation of China (No. 81360315).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jiye Zhu and Shan Huang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
(a) Efficiency of transfection in Hep-SK-1 cells; (b) LOX mRNA expression in MHCC-97-H cells was inhibited by siRNA of LOX; (c) The density of bars were measured in comparison with corresponding controls; (d) Protein level of LOX in MHCC-97-H cells was inhibited by siRNA of LOX; Experimental schematic for combination of siRNA and proliferation assay (e), or migration and invasion assay (f). (TIFF 1259 kb)
Fig. S2
(a) Representative images from migration and invasion assay of MHCC-97-H cells; Magnification, ×100 for all the pictures. (b) Representative pictures of IHC staining of LOX and VEGF from same tumor areas; Magnification, ×100 for all the pictures. (c) Correlation between total IHC scores of LOX and VEGF. (TIFF 7604 kb)
Rights and permissions
About this article
Cite this article
Zhu, J., Huang, S., Wu, G. et al. Lysyl Oxidase Is Predictive of Unfavorable Outcomes and Essential for Regulation of Vascular Endothelial Growth Factor in Hepatocellular Carcinoma. Dig Dis Sci 60, 3019–3031 (2015). https://doi.org/10.1007/s10620-015-3734-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-3734-5