Abstract
Background
Genetic signatures may differ by histopathologic and anatomic subtypes of gastric cancer (GC). B-cell translocation gene 1 (BTG1) was identified as one of genes downregulated in GC tissues from our microarray data.
Aims
To evaluate the clinical implications of BTG1 expression in GC and the genetic diversity among GC subtypes.
Methods
BTG1 mRNA expression was analyzed in GC cell lines and 233 pairs of surgical specimens. The mutational and methylation status of BTG1 in GC cell lines was analyzed, and immunohistochemistry was conducted to determine the distribution of BTG1. The pattern and prognostic significance of BTG1 expression were correlated with the three proposed GC subtypes.
Results
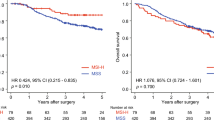
BTG1 mRNA was downregulated in 82 % of GC cell lines and in 88 % of clinical GC tissues. Promoter hypermethylation events or sequence mutations were not detected in GC cell lines. The pattern of BTG1 expression as observed by immunohistochemistry was consistent with that of its mRNA. Downregulation of BTG1 mRNA in GCs was significantly associated with shorter disease-specific and recurrence-free survival. Multivariate analysis of disease-specific survival identified downregulation of BTG1 transcription as an independent prognostic factor. BTG1 mRNA expression was more strongly suppressed in proximal nondiffuse and diffuse GC compared with distal nondiffuse GC, and subgroup analysis revealed that BTG1 downregulation led to adverse prognosis, specifically in patients with proximal nondiffuse and diffuse GC.
Conclusions
Altered expression of BTG1 is a potential biomarker for carcinogenesis and progression of GC, particularly for proximal nondiffuse and diffuse GC.




Similar content being viewed by others
References
Jang BG, Kim WH. Molecular pathology of gastric carcinoma. Pathobiology. 2011;78:302–310.
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
GLOBOCAN. Estimated cancer incidence, mortality and prevalence worldwide in 2012, stomach cancer. http://globocan.iarc.fr; 2012.
Hartgrink HH, Jansen EP, van Grieken NC, van de Velde CJ. Gastric Cancer. Lancet. 2009;374:477–490.
Gonzalez CA, Agudo A. Carcinogenesis, prevention and early detection of gastric cancer: where we are and where we should go. Int J Cancer. 2012;130:745–753.
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108.
Kanda M, Shimizu D, Nomoto S, et al. Prognostic impact of expression and methylation status of DENN/MADD domain-containing protein 2D in gastric cancer. Gastric Cancer. (Epub ahead of print). doi:10.1007/s10120-014-0372-0.
Shikata K, Doi Y, Yonemoto K, et al. Population-based prospective study of the combined influence of cigarette smoking and Helicobacter pylori infection on gastric cancer incidence: the Hisayama study. Am J Epidemiol. 2008;168:1409–1415.
Steevens J, Botterweck AA, Dirx MJ, van den Brandt PA, Schouten LJ. Trends in incidence of oesophageal and stomach cancer subtypes in Europe. Eur J Gastroenterol Hepatol. 2010;22:669–678.
Tramacere I, Negri E, Pelucchi C, et al. A meta-analysis on alcohol drinking and gastric cancer risk. Ann Oncol. 2012;23:28–36.
Janjigian YY, Kelsen DP. Genomic dysregulation in gastric tumors. J Surg Oncol. 2013;107:237–242.
Lin LL, Huang HC, Juan HF. Discovery of biomarkers for gastric cancer: a proteomics approach. J Proteomics. 2012;75:3081–3097.
Yasui W, Sentani K, Sakamoto N, Anami K, Naito Y, Oue N. Molecular pathology of gastric cancer: research and practice. Pathol Res Pract. 2011;207:608–612.
Kanda M, Nomoto S, Oya H, et al. Dihydropyrimidinase-like 3 facilitates malignant behavior of gastric cancer. J Exp Clin Cancer Res. 2014;33:66.
Shah MA, Khanin R, Tang L, et al. Molecular classification of gastric cancer: a new paradigm. Clin Cancer Res. 2011;17:2693–2701.
Kanda M, Shimizu D, Nomoto S, et al. Clinical significance of expression and epigenetic profiling of TUSC1 in gastric cancer. J Surg Oncol. 2014;110:136–144.
Kanda M, Nomoto S, Okamura Y, et al. Detection of metallothionein 1G as a methylated tumor suppressor gene in human hepatocellular carcinoma using a novel method of double combination array analysis. Int J Oncol. 2009;35:477–483.
Cho JW, Kim JJ, Park SG, et al. Identification of B-cell translocation gene 1 as a biomarker for monitoring the remission of acute myeloid leukemia. Proteomics. 2004;4:3456–3463.
Lee H, Cha S, Lee MS, Cho GJ, Choi WS, Suk K. Role of antiproliferative B cell translocation gene-1 as an apoptotic sensitizer in activation-induced cell death of brain microglia. J Immunol. 2003;171:5802–5811.
Rouault JP, Rimokh R, Tessa C, et al. BTG1, a member of a new family of antiproliferative genes. EMBO J. 1992;11:1663–1670.
Sobin LH, Gospodarowicz MK, Wittekind W. International Union Against Cancer, TNM classification of malignant tumors. 7th ed. New York: Wiley-Blackwell; 2009.
Sakuramoto S, Sasako M, Yamaguchi T, et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N Engl J Med. 2007;357:1810–1820.
Kanda M, Nomoto S, Oya H, et al. Decreased expression of prenyl diphosphate synthase subunit 2 correlates with reduced survival of patients with gastric cancer. J Exp Clin Cancer Res. 2014;33:88.
Kanda M, Knight S, Topazian M, et al. Mutant GNAS detected in duodenal collections of secretin-stimulated pancreatic juice indicates the presence or emergence of pancreatic cysts. Gut. 2013;62:1024–1033.
Kanda M, Matthaei H, Wu J, et al. Presence of somatic mutations in most early-stage pancreatic intraepithelial neoplasia. Gastroenterology. 2012;142:730.e739–733.e739.
Kanda M, Sadakari Y, Borges M, et al. Mutant TP53 in duodenal samples of pancreatic juice from patients with pancreatic cancer or high-grade dysplasia. Clin Gastroenterol Hepatol. 2013;11:719.e715–730.e715.
Hibino S, Kanda M, Oya H, et al. Reduced expression of DENND2D through promoter hypermethylation is an adverse prognostic factor in squamous cell carcinoma of the esophagus. Oncol Rep. 2014;31:693–700.
Kanda M, Nomoto S, Okamura Y, et al. Promoter hypermethylation of fibulin 1 gene is associated with tumor progression in hepatocellular carcinoma. Mol Carcinog. 2011;50:571–579.
Kawamura-Tsuzuku J, Suzuki T, Yoshida Y, Yamamoto T. Nuclear localization of Tob is important for regulation of its antiproliferative activity. Oncogene. 2004;23:6630–6638.
Prevot D, Voeltzel T, Birot AM, et al. The leukemia-associated protein Btg1 and the p53-regulated protein Btg2 interact with the homeoprotein Hoxb9 and enhance its transcriptional activation. J Biol Chem. 2000;275:147–153.
Rodier A, Rochard P, Berthet C, et al. Identification of functional domains involved in BTG1 cell localization. Oncogene. 2001;20:2691–2703.
van Galen JC, Kuiper RP, van Emst L, et al. BTG1 regulates glucocorticoid receptor autoinduction in acute lymphoblastic leukemia. Blood. 2010;115:4810–4819.
Zhao Y, Gou WF, Chen S, Takano Y, Xiu YL, Zheng HC. BTG1 expression correlates with the pathogenesis and progression of ovarian carcinomas. Int J Mol Sci. 2013;14:19670–19680.
Berthet C, Guehenneux F, Revol V, et al. Interaction of PRMT1 with BTG/TOB proteins in cell signalling: molecular analysis and functional aspects. Genes Cells. 2002;7:29–39.
Yang Y, Bedford MT. Protein arginine methyltransferases and cancer. Nat Rev Cancer. 2013;13:37–50.
Waanders E, Scheijen B, van der Meer LT, et al. The origin and nature of tightly clustered BTG1 deletions in precursor B-cell acute lymphoblastic leukemia support a model of multiclonal evolution. PLoS Genet. 2012;8:e1002533.
Bang YJ, Van Cutsem E, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687–697.
Sakaguchi T, Watanabe A, Sawada H, et al. Characteristics and clinical outcome of proximal-third gastric cancer. J Am Coll Surg. 1998;187:352–357.
Tafe LJ, Janjigian YY, Zaidinski M, et al. Human epidermal growth factor receptor 2 testing in gastroesophageal cancer: correlation between immunohistochemistry and fluorescence in situ hybridization. Arch Pathol Lab Med. 2011;135:1460–1465.
Marrelli D, Roviello F, de Manzoni G, et al. Different patterns of recurrence in gastric cancer depending on Lauren’s histological type: longitudinal study. World J Surg. 2002;26:1160–1165.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10620_2014_3477_MOESM1_ESM.tif
Supplemental Fig. 1 A Mutational analysis of BTG1. Mutations were not detected using HRM analysis in BTG1 exons 1 and 2. B Methylation analysis of the BTG1 promoter in GC cell lines. Representative chromatograms of bisulfite sequence analysis, showing absence of promoter hypermethylation (TIFF 6925 kb)
10620_2014_3477_MOESM2_ESM.tif
Supplemental Fig. 2 A Even when patients were subdivided according to UICC staging (stages I–II and III–IV), BTG1 mRNA expression in GC tissues was significantly lower in proximal nondiffuse and diffuse GCs compared with distal GC. B Survival of patients with stage II/III GC. When the analysis was limited to cases with or without BTG1 mRNA downregulation in GC tissues, prognosis was similar among the three GC subtypes in patients without downregulation of BTG1 mRNA expression, whereas proximal nondiffuse and diffuse GCs were significantly associated with shorter survival in patients with downregulation of BTG1 mRNA expression (TIFF 6034 kb)
Rights and permissions
About this article
Cite this article
Kanda, M., Oya, H., Nomoto, S. et al. Diversity of Clinical Implication of B-Cell Translocation Gene 1 Expression by Histopathologic and Anatomic Subtypes of Gastric Cancer. Dig Dis Sci 60, 1256–1264 (2015). https://doi.org/10.1007/s10620-014-3477-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-014-3477-8