Abstract
Background and Aim
Ghrelin has distinct effects on gastrointestinal motility through the vagus nerve and gastric excitatory neural plexus. The objectives of this study were to investigate the dynamics of ghrelin and expression of neuromuscular markers in a newly established surgically manipulated rat model of gastric outlet obstruction (GOO), akin to the pyloric stricture associated with duodenal ulcer, advanced gastric cancer, and other conditions, in the clinical setting.
Material and Methods
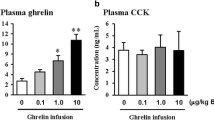
The rats were divided into two groups, a control group (sham operation) and the GOO group (proximal duodenal stricture). The animals were sacrificed 2 weeks after the operation. Plasma and gastric ghrelin were measured by radioimmunoassay. mRNA expression in the stomach of neural choline acetyltransferase (ChAT), c-kit, and membrane-bound stem cell factor (SCF) were analyzed by quantitative RT-PCR. In addition, gastric mRNA expression of the aforementioned were also evaluated 60 min after intraperitoneal administration of a synthetic GHS-R1a antagonist ([d-Lys3] GHRP-6 6.0 mg/kg).
Results
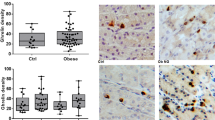
Mechanical GOO induced increases of fasting plasma ghrelin levels and hyperplasia of the gastric muscle layers, with enhanced expression of the gastric neuromuscular markers. Administration of [d-Lys3] GHRP-6 normalized the enhanced expression of c-kit and SCF.
Conclusion
GOO stimulates ghrelin dynamics and then enhances the mechanistic expression of gastric cellular communication network molecules between nerves and smooth muscle cells.






Similar content being viewed by others
References
Kojima M, Hosoda H, Date Y, et al. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature. 1999;402:656–660.
Masuda Y, Tanaka T, Inomata N, et al. Ghrelin stimulates gastric acid secretion and motility in rats. Biochem Biophys Res Commun. 2000;276:905–908.
Xu L, Depoortere I, Tomasetto C, et al. Evidence for the presence of motilin, ghrelin, and the motilin and ghrelin receptor in neurons of the myenteric plexus. Regul Pept. 2005;124:119–125.
De Winter BY, De Man JG, Seerden TC, et al. Effect of ghrelin and growth hormone-releasing peptide 6 on septic ileus in mice. Neurogastroenterol Motil. 2004;16:439–446.
Fujino K, Inui A, Asakawa A, et al. Ghrelin induces fasted motor activity of the gastrointestinal tract in conscious fed rats. J Physiol. 2003;550:227–240.
Chasen M, Bhargava R. Gastrointestinal symptoms, electrogastrography, inflammatory markers, and pg-sga in patients with advanced cancer. Support Care Cancer. 2011 [Epub ahead of print].
Nishizawa T, Suzuki H, Nomoto Y, et al. Enhanced plasma ghrelin levels in patients with functional dyspepsia. Aliment Pharmacol Ther. 2006;24(Suppl 4):104–110.
Ohnishi N, Okada K, Yoshioka M, et al. Studies on interactions between traditional herbal and western medicines. V. Effects of sho-saiko-to (xiao-cai-hu-tang) on the pharmacokinetics of carbamazepine in rats. Biol Pharm Bull. 2002;25:1461–1466.
Ariyasu H, Takaya K, Hosoda H, et al. Delayed short-term secretory regulation of ghrelin in obese animals: evidenced by a specific RIA for the active form of ghrelin. Endocrinology. 2002;143:3341–3350.
Hosoda H, Kojima M, Matsuo H, et al. Ghrelin and des-acyl ghrelin: two major forms of rat ghrelin peptide in gastrointestinal tissue. Biochem Biophys Res Commun. 2000;279:909–913.
Masaoka T, Suzuki H, Hosoda H, et al. Enhanced plasma ghrelin levels in rats with streptozotocin-induced diabetes. FEBS Lett. 2003;541:64–68.
Bisschops R. Ligand and electrically induced activation patterns in myenteric neuronal networks. Confocal calcium imaging as a bridge between basic and human physiology. Verh K Acad Geneeskd Belg. 2008;70:105–145.
Sakata I, Yamazaki M, Inoue K, et al. Growth hormone secretagogue receptor expression in the cells of the stomach-projected afferent nerve in the rat nodose ganglion. Neurosci Lett. 2003;342:183–186.
Suzuki H, Masaoka T, Hosoda H, et al. Helicobacter pylori infection modifies gastric and plasma ghrelin dynamics in mongolian gerbils. Gut. 2004;53:187–194.
Izbeki F, Wittman T, Rosztoczy A, et al. Immediate insulin treatment prevents gut motility alterations and loss of nitrergic neurons in the ileum and colon of rats with streptozotocin-induced diabetes. Diabetes Res Clin Pract. 2008;80:192–198.
Suzuki S, Suzuki H, Horiguchi K, et al. Delayed gastric emptying and disruption of the interstitial cells of cajal network after gastric ischaemia and reperfusion. Neurogastroenterol Motil. 2010;22:585–593, e126.
Tack J, Talley NJ, Camilleri M, et al. Functional gastroduodenal disorders. Gastroenterology. 2006;130:1466–1479.
Acknowledgments
This work was supported in part by a Grant-in-Aid for Young Scientists (B) from the Japan Society for the Promotion of Science (no. 18790471 to EI), a Grant-in-Aid for Scientific Research (B) from the Japan Society for the Promotion of Science (no. 22300169, to HS), a Health and Labour Sciences Research Grant for Research on Health Technology Assessment (Clinical Research Promotion no. 47 to HS), a grant from the JSPS Bilateral Joint Projects with Belgium (11035231-000061), a grant from the Smoking Research Foundation (to HS), the Keio Gijuku Academic Development Fund (to HS), and a Nateglinide Memorial Toyoshima Research and Education Fund (to HS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iwasaki, E., Suzuki, H., Masaoka, T. et al. Enhanced Gastric Ghrelin Production and Secretion in Rats with Gastric Outlet Obstruction. Dig Dis Sci 57, 858–864 (2012). https://doi.org/10.1007/s10620-011-1953-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-011-1953-y